Abstract
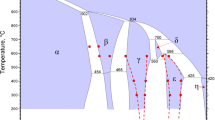
We present an ultrasonic and neutron powder diffraction study of crystalline MgSO4·7H2O (synthetic epsomite) and MgSO4·7D2O under pressure up to ~3 GPa near room temperature and up to ~2 GPa at lower temperatures. Both methods provide complementary data on the phase transitions and elasticity of magnesium sulphate heptahydrate, where protonated and deuterated counterparts exhibit very similar behaviour and properties. Under compression in the declared pressure intervals, we observed three different sequences of phase transitions: between 280 and 295 K, phase transitions occurred at approximately 1.4, 1.6, and 2.5 GPa; between 240 and 280 K, only a single phase transition occurred; below 240 K, there were no phase transformations. Overall, we have identified four new phase fields at high pressure, in addition to that of the room-pressure orthorhombic structure. Of these, we present neutron powder diffraction data obtained in situ in the three phase fields observed near room temperature. We present evidence that these high-pressure phase fields correspond to regions where MgSO4·7H2O decomposes to a lower hydrate by exsolving water. Upon cooling to liquid nitrogen temperatures, the ratio of shear modulus G to bulk modulus B increases and we observe elastic softening of both moduli with pressure, which may be a precursor to pressure-induced amorphization. These observations may have important consequences for modelling the interiors of icy planetary bodies in which hydrated sulphates are important rock-forming minerals, such as the large icy moons of Jupiter, influencing their internal structure, dynamics, and potential for supporting life.












Similar content being viewed by others
References
Alexandrov KS, Rhyzhova TV, Rostuntseva AI (1963) Elastic properties of some sulfate heptahydrate crystals. Sov Phys Crystallogr 7:753–755
Brazhkin VV, Lyapin AG (1996) Lattice instability approach to the problem of high-pressure solid-state amorphization. High Press Res 15:9–30
Brazhkin VV, Lyapin AG, Stalgorova OV, Gromnitskaya EL, Popova SV, Tsiok OB (1997) On the nature of amorphous-to-amorphous and crystal-to-amorphous transitions under high pressure. J Non Cryst Solids 212:49–54
Bridgman PW (1948a) Rough compression of 177 substances to 40,000 kg/cm2. Proc Am Acad Arts Sci 76:71–87
Bridgman PW (1948b) The linear compression of various single crystals to 30,000 kg/cm2. Proc Am Acad Arts Sci 76:89–99
Chou I, Seal RR (2007) Magnesium and calcium sulfate stabilities and the water budget on Mars. J Geophys Res Planets 112:E11004
Churagulov BR (1987) Some characteristics of the effect of a pressure (up to 1000 MPa) on temperatures of phase-transformations and concentration of saturated solutions on lines of the phase-equilibria on R-T-diagrams of salt-water binary-systems. Zh Neorg Khim 32(10):2527–2536
Churagulov BR, Kalashnikov YA (1969) Effect of pressure on the partial dehydration of zinc sulphate heptahydrate. Russ J Phys Chem 43(2):258–262
Dalton JB (2010) Spectroscopy of icy moon surface materials. Space Sci Rev 153(1–4):219–247
Dalton JB, Prieto-Ballesteros O, Kargel JS, Jamieson CS, Jolivet J, Quinn R (2005) Spectral comparison of heavily hydrated salts with disrupted terrains on Europa. Icarus 177(2):472–490
Dalton JB, Shirley JH, Kamp LW (2012) Europa’s icy bright plains and dark linea: exogenic and endogenic contributions to composition and surface properties. J Geophys Res 117:E03003
Dougherty AJ, Hogenboom DL, Kargel JS (2007) Volumetric and optical studies of high pressure phases of MgSO4-H2O with applications to Europa. Lunar Planet Sci Conf 38:2275
Fateev EG (2012) Cвepxнизкaя yпpyгaя cтaбильнocть MgSO4 − H2O льдa. JETP Lett 38(8):1–9
Feldman WC, Mellon MT, Maurice S, Prettyman TH, Carey JW, Vaniman DT, Fialips CI, Kargel JS, Elphic RC, Funsten HO, Laurence DJ, Tokar RL (2004) Contributions from hydrated states of MgSO4 to the reservoir of hydrogen at equatorial latitudes on Mars. Lunar Planet Sci Conf 35:2035
Ferraris G, Jones DW, Yerkess J (1973) Refinement of the crystal structure of magnesium sulphate heptahydrate (Epsomite) by neutron diffraction. J Chem Soc, Dalton Trans 1973:816–821
Fortes AD, Choukroun M (2010) Phase behaviour of ices and hydrates. Space Sci Rev 153(1–4):185–218
Fortes AD, Wood IG, Alfredsson M, Vočadlo L, Knight KS (2006a) The thermoelastic properties of MgSO4·7D2O (epsomite) from powder neutron diffraction and ab initio simulation. Eur J Miner 18(4):449–462
Fortes AD, Wood IG, Vočadlo L, Brand HEA, Grindrod PM, Joy KH, Tucker MG (2006b) The phase behaviour of epsomite (MgSO4·7H2O) to 50 kbar: planetary implications. Lunar Planet Sci Conf 37:1029
Fortes AD, Wood IG, Alfredsson M, Vočadlo L, Knight KS, Marshall WG, Tucker MG, Fernandez-Alonso F (2007) The high-pressure phase diagram of ammonia dihydrate. High Press Res 27(2):201–212
Fortes AD, Wood IG, Brand HEA, Tucker MG (2008) The effect of pressure on the structure of meridianiite (MgSO4·11D2O). ISIS Experimental Report RB 820064, Rutherford Appleton Laboratory
Fortes AD, Wood IG, Knight KS (2008b) The crystal structure and thermal expansion tensor of MgSO4·11D2O (meridianiite) determined by neutron powder diffraction. Phys Chem Miner 35(4):207–221
Fortes AD, Wood IG, Tucker MG (2009) The effect of pressure on the structure of meridianiite (MgSO4·11D2O). ISIS Experimental Report RB 910226, Rutherford Appleton Laboratory
Fortes AD, Lemée-Cailleau M–H, Knight KS, Jura M (2010) Magnesium sulfate trihydrate: an elusive mineral on Earth and Mars? International Mineralogical Association, 20th General Meeting, Budapest, August 21–27th 2010. See also, Institut Laue Langevin experimental report 5-11-360 (2009), and ISIS Experimental Report RB1010078, Rutherford Appleton Laboratory
Fortes AD, Browning F, Wood IG (2012a) Cation substitution in synthetic meridianiite (MgSO4·11H2O) I: X-ray powder diffraction analysis of quenched polycrystalline aggregates. Phys Chem Miner 39(5):419–441
Fortes AD, Browning F, Wood IG (2012b) Cation substitution in synthetic meridianiite (MgSO4·11H2O) II: variation in unit-cell parameters determined from X-ray powder diffraction data. Phys Chem Miner 39(6):443–454
Fortes AD, Wood IG, Alfredsson M, Vočadlo L, Knight KS, Marshall WG, Tucker MG, Fernandez-Alonso F (2012c) Corrigendum. High Press Res 32:337. doi:10.1080/08957959.2012.673603
Fredericksson K, Kerridge JF (1988) Carbonates and sulfates in CI chondrites–formation by aqueous alteration on the parent body. Meteoritics 23:35–44
Genceli FE, Horikawa S, Iizuka Y, Sakurai T, Hondoh T, Kawamura T, Witkamp G (2009) Meridianiite detected in ice. J Glaciol 55:117–122
Grasset O, Sotin C, Mousis O, Mevel L (2000) High pressure experiments in the system MgSO4–H2O: implications for Europa. Lunar Planet Sci Conf 31:1386
Grasset O, Mevel L, Mousis O, Sotin C (2001) The pressure dependence of the eutectic composition in the system MgSO4-H2O: implications for the deep liquid layer of icy satellites. Lunar Planet Sci Conf 32:1524
Gromnitskaya EL, Stal’gorova OV, Brazhkin VV, Lyapin AG (2001) Ultrasonic study of the nonequilibrium pressure-temperature diagram of H2O. Phys Rev B 64:094205–094221
Gromnitskaya EL, Yagafarov OF, Lyapin AG, Brazhkin VV, Fortes AD (2010) Ultrasonic study of epsomite (MgSO4·7H2O) under pressure. High Press Res 30(1):51–54
Hodenburg RF, Kühn R (1967) Zur Kenntnis der Magnesiumsulfathydrate und der Effloreszenzen des Kieserits von Hartsalzen. Kali und Steinsalz 4(10):326–340
Hogenboom DL, Kargel JS, Ganasan JP, Lee L (1995) Magnesium sulfate-water to 400 MPa using a novel piezometer: densities, phase equilibria, and planetological implications. Icarus 115(2):258–277
Jahn S, Schmidt C (2010) Speciation in aqueous MgSO4 fluids at high pressures and high temperatures from ab initio molecular dynamics and Raman spectroscopy. J Phys Chem B 114(47):15565–15572
Jambor JL, Nordstrom DK, Alpers CN (2000) Metal-sulfate salts from sulfide mineral oxidation. Rev Mineral 40:303–350
Kargel JS (1991) Brine volcanism and the interior structures of asteroids and icy satellites. Icarus 94(2):369–390
Larsen AC, Von Dreele RB (2000) General Structure Analysis System (GSAS). Los Alamos National Laboratory Report LAUR 86–748, Los Alamos, New Mexico http://www.ncnr.nist.gov/xtal/software/gsas.html
Le Bail A (2005) Whole powder pattern decomposition methods and applications–a retrospection. Powder Diffr 20:316–326
Livshits LD, Genshaft YS, Ryabin YN (1963) Equilibrium diagram of the crystal hydrates of MgSO4 at high pressures. Russ J Inorg Chem 8:676–678
Lyapin AG, Brazhkin VV (1996) Pressure-induced lattice instability and solid-state amorphization. Phys Rev B 54:12036–12048
Ma H, Bish DL, Wang H-W, Chipera SJ (2009a) Determination of the crystal structure of sanderite, MgSO4·2H2O, by X-ray powder diffraction and the charge flipping method. Am Mineral 94:622–625
Ma H, Bish DL, Wang H-W, Chipera SJ (2009b) Structure determination of the 2.5 hydrate MgSO4 by simulated annealing. Am Mineral 94:1071–1074
Marshall WG, Francis DJ (2002) Attainment of near hydrostatic conditions using the Paris-Edinburgh cell. J Appl Cryst 35(1):122–125
McCord TB, Hansen GB, Matson DL, Johnson TV, Crowley JK, Fanale FP, Carlson RW, Smythe WD, Martin PD, Hibbitts CA, Granahan JC, Ocampo A, the NIMS Team (1999) Hydrated salt minerals on Europa’s surface from the Galileo Near-Infrared Mapping Spectrometer (NIMS) investigation. J Geophys Res Planets 104(E5):11827–11851
McCord TB, Hansen GB, Hibbitts CA (2001) Hydrated salts on Ganymede’s surface: evidence of an ocean below. Science 292:1523–1525
McSkimin HJ (1964) Ultrasound methods of measurements of mechanical characteristics in liquids and solids. In: Mason WP (ed) Physical Acoustics, Principles and Methods, vol I, Methods and devices, Part A. Academic press, New York
Milliken RE, Mustard JF, Poulet F, Jouglet D, Bibring J-P, Gondet B, Langevin Y (2007) Hydration state of the martian surface as seen by Mars Express OMEGA:2. H2O content of the surface. J Geophys Res 112, E08S07
Mishima O, Calvert LD, Whalley E (1984) ‘Melting ice’ I at 77 K and 10 kbar: a new method of making amorphous solids. Nature 310:393–395
Murnaghan FD (1944) The compressibility of media under extreme pressures. Proc Natl Acad Sci USA 30(9):244–247
Nakamura RS (2003) High pressure sulfate-water system in the large icy satellites. Eos Trans AGU 84(46): Fall Meeting Suppl, Abstract P51B-0449
Nakamura R, Ohtani E (2011) The high-pressure phase relation of the MgSO4-H2O system and its implications for the internal structure of Ganymede. Icarus 211(1):648–654
Pantea C, Rickel DG, Migliori A, Leisure RG, Jianzhong Zhang J, Zhao Y, El-Khatib S, Li B (2005) Digital ultrasonic pulse-echo overlap system and algorithm for unambiguous determination of pulse transit time. Rev Sci Instr 76:114902
Peterson RC, Wang R (2006) Crystal molds on Mars: melting of a possible new mineral species to create Martian chaotic terrain. Geology 34(11):957–960
Peterson RC, Nelson W, Madu B, Shurvell HF (2007) Meridianiite: a new species observed on Earth and predicted to exist on Mars. Am Mineral 92(10):1756–1759
Solomonidou A, Coustenis A, Bampasidis G, Kyriakopoulos K, Moussas X, Bratsolis E, Hirtzig M (2011) Water oceans of Europa and other moons: implications for life in other solar systems. J Cosmol 13:4191–4211
Sood RR, Stager RA (1966) Pressure-induced dehydration reactions and transitions in inorganic hydrates. Science 154:388–390
Spencer RJ (2000) Sulfate minerals in evaporite deposits. Rev Min Geochem 40(1):173–192
Stal’gorova OV, Gromnitskaya EL, Dmitriev DR, Voronov FF (1996) Ultrasonic piezometer for the 0–2.0 GPa pressure and 77–300 K temperature range. Instr Exp Tech 39:880–884
Stal’gorova OV, Gromnitskaya EL, Brazhkin VV (1995) Experimental confirmation of the instability of the crystal structure of Ih ice prior to amorphization under pressure. JETP Lett 62:356–360
Strässle T, Saitta AM, Klotz S, Braden M (2004) Phonon dispersion of ice under pressure. Phys Rev Lett 93:225901
Sundara Rao RVG (1950) Elastic constants of the heptahydrates of magnesium and zinc sulphates. Proc Indian Acad Sci A 31:365–370
Toby BH (2001) EXPGUI a graphical user interface for GSAS. J Appl Cryst 34(2):210–213
Vance S (2007) High pressure and low temperature equations of state for aqueous magnesium sulfate: applications to the search for life in extraterrestrial oceans, with particular reference to Europa. PhD Thesis, University of Washington
Voronkov AA (1958) The piezoelectric, elastic and dielectric properties of crystals of MgSO4·7H2O. Sov Phys–Crystallogr 3:722–725
Voronov FF, Chernysheva EV (1999) Anomalies in the elastic properties of silicious iron single crystals at pressures of up to 9 GPa and the α–ε phase transformation. Phys Solid State (St. Petersburg) 41:462–467
Wang A, Freeman JJ, Chou I-M, Joliff BL (2011) Stability of Mg-sulfates at −10 °C and the rates of dehydration/rehydration processes under conditions relevant to Mars. J Geophys Res 116:E120006
Yakuschenko AN, Churagulov BR (1984) Pressure effect on temperatures of phase transitions in the zinc-sulfate water system. Zh Fiz Khim 58(2):311–314
Zolotov MY, Kuzmin RO, Shock EL (2004) Mineralogy, abundance, and hydration state of sulfates and chlorides at the Mars Pathfinder landing site. Lunar Planet Sci Conf 35:1465
Acknowledgments
The authors wish to thank S.G. Lyapin for valuable discussions. The work was partially supported by the Russian Foundation for Basic Research (11-02-00303, 10-02-01407) and by the Programs of the Presidium of the Russian Academy of Sciences. ADF was supported by Fellowships from the U.K. Science and Technology Facilities Council (PPA/P/S/2003/00247, PP/E006515/1). The authors thank the ISIS facility for beam time to conduct the neutron diffraction experiments and recognise the substantial contributions of ISIS technical support staff to the success of those studies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gromnitskaya, E.L., Yagafarov, O.F., Lyapin, A.G. et al. The high-pressure phase diagram of synthetic epsomite (MgSO4·7H2O and MgSO4·7D2O) from ultrasonic and neutron powder diffraction measurements. Phys Chem Minerals 40, 271–285 (2013). https://doi.org/10.1007/s00269-013-0567-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00269-013-0567-7