Abstract
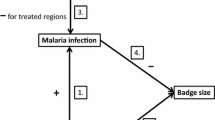
Parasites can impose heavy costs on their hosts, and females may benefit from selecting mates that are parasite resistant and/or have a stronger immune response. Trade-offs between immune response and sexual signaling have been proposed as a mechanism to ensure signal honesty. Much of the work on sexual signaling and immune response does not consider parasites directly and thus cannot confirm whether a stronger immune response necessarily results in lower parasite burdens. Also, immunity is costly, and these costs can lower the overall fitness of individuals with too strong of an immune response. Males with immune responses of intermediate strength are therefore expected to be preferred by females and have the highest reproductive success. We tested whether immune response and blood parasite loads relate to sexual signaling and mating preferences in western bluebirds (Sialia mexicana). Immunity did not predict male reproductive success when considering either within- or extra-pair offspring, although a stronger immune response was correlated with lower parasite loads. However, uninfected males were more likely to sire extra-pair offspring than males infected with avian malaria. Thus, females were more likely to mate with uninfected males but not necessarily males with a stronger immune response. Our results may indicate that females select parasite-resistant males as mates to gain resistance genes for their offspring or that infected males are less likely to pursue extra-pair copulations.


Similar content being viewed by others
References
Able DJ (1996) The contagion indicator hypothesis for parasite-mediated sexual selection. Proc Natl Acad Sci U S A 93:2229–2233
Adamo SA (2004) Estimating disease resistance in insects: phenoloxidase and lysozyme-like activity and disease resistance in the cricket Gryllus texensis. J Insect Physiol 50:209–216
Akçay E, Roughgarden J (2007) Extra-pair paternity in birds: review of the genetic benefits. Evol Ecol Res 9:855–868
Akçay Ç, Searcy WA, Campbell E, Reed VA, Templeton CN, Hardwick KM, Beecher MD (2012) Who initiates extrapair mating in song sparrows? Behav Ecol 23:44–50
Albrecht T, Vinkler M, Schnitzer J, Poláková R, Munclinger P, Bryja J (2009) Extra-pair fertilizations contribute to selection on secondary male ornamentation in a socially monogamous passerine. J Evol Biol 22:2020–2030
Asghar M, Hasselquist D, Bensch S (2011) Are chronic avian haemosporidian infections costly in wild birds? J Avian Biol 42:530–537
Asghar M, Westerdahl H, Zehtindjiev P, Ilieva M, Hasselquist D, Bensch S (2012) Primary peak and chronic malaria infection levels are correlated in experimentally infected great reed warblers. Parasitology 139:1246–1252
Atkinson CT, van Riper C (1991) Pathogenicity and epizootiology of avian haematozoa: Plasmodium, Leucocytozoon, and Haemoproteus. In: Loye JE, Zuk M (eds) Bird–parasite interactions: ecology, evolution, and behaviour. Oxford University Press, London, pp 19–48
Balenger SL, Johnson LS, Mays HL, Masters BS (2009a) Extra-pair paternity in the socially monogamous mountain bluebird Sialia currucoides and its effect on the potential for sexual selection. J Avian Biol 40:173–180
Balenger SL, Johnson LS, Masters BS (2009b) Sexual selection in a socially monogamous bird: male color predicts paternity success in the mountain bluebird. Behav Ecol Sociobiol 63:403–411
Bensch S, Hellgren O, Pérez-Tris J (2009) MalAvi: a public database of malaria parasites and related haemosporidians in avian hosts based on mitochondrial cytochrome b lineages. Mol Ecol Resour 9:1353–1358
Chiver I, Stutchbury BJM, Morton ES (2008) Do male plumage and song characteristics influence female off-territory forays and paternity in the hooded warbler? Behav Ecol Sociobiol 62:1981–1990
Clayton DH (1991) The influence of parasites on host sexual selection. Parasitol Today 7:329–334
Dein FJ (1984) Laboratory manual of avian hematology. Association of Avian Veterinarians, East Northport
Dickinson JL (2001) Extrapair copulations in western bluebirds (Sialia mexicana): female receptivity favors older males. Behav Ecol Sociobiol 50:423–429
Dickinson JL (2003) Male share of provisioning is not influenced by actual or apparent loss of paternity in western bluebirds. Behav Ecol 14:360–366
Dickinson JL, Kraaijeveld K, Smit-Kraaijeveld F (2000) Specialized extrapair mating display in western bluebirds. Auk 117:1078–1080
Duckworth RA (2006) Behavioral correlations across breeding contexts provide a mechanism for a cost of aggression. Behav Ecol 17:1011–1019
Fair JM, Myers OB (2002a) Early reproductive success of western bluebirds and ash-throated flycatchers: a contaminated landscape perspective. Environ Pollut 118:321–330
Fair JM, Myers OB (2002b) The ecological and physiological costs of lead shot and immunological challenge to developing western bluebirds. Ecotoxicology 11:199–208
Fair JM, Whitaker SJ (2008) Avian cell-mediated immune response to drought. Wilson J Ornithol 120:813–819
Fair JM, Myers OB, Ricklefs RE (2003) Immune and growth response of western bluebirds and ash-throated flycatchers to soil contaminants. Ecol Appl 13:1817–1829
Fair JM, Paul E, Jones J, Clark AB, Davie C, Kaiser G (2010) Guidelines to the use of wild birds in research. The Ornithological Council, Washington, DC
Faircloth BC, Keller GP, Nairn J, Palmer WE, Carroll JP, Gowaty PA (2006) Tetranucleotide microsatellite loci from eastern bluebirds Sialia sialis. Mol Ecol Notes 6:646–649
Ferree ED, Dickinson JL, Kleiber D, Stern CA, Haydock J, Stanback MT, Schmidt V, Eisenberg L, Stolzenburg C (2008) Development and cross-species testing of western bluebird (Sialia mexicana) microsatellite primers. Mol Ecol Resour 8:1348–1350
Folstad I, Karter AJ (1992) Parasites, bright males, and the immunocompetence handicap hypothesis. Am Nat 139:603–622
Forsman AM, Vogel LA, Sakaluk SK, Thompson CF (2008) Immune-challenged house wren broods differ in the relative strengths of their responses among different axes of the immune system. J Evol Biol 21:873–878
Fox A, Hudson PJ (2001) Parasites reduce territorial behaviour in red grouse (Lagopus lagopus scoticus). Ecol Lett 4:139–143
French SS, Moore MC, Demas GE (2009) Ecological immunology: the organism in context. Integr Comp Biol 49:246–253
Getty T (2002) Signaling health versus parasites. Am Nat 159:363–371
Girard J, Goldberg T, Hamer GL (2011) Field investigation of innate immunity in passerine birds in suburban Chicago, Illinois, USA. J Wildl Dis 47:603–611
Graham AL, Allen JE, Read AF (2005) Evolutionary causes and consequences of immunopathology. Annu Rev Ecol Evol Syst 36:373–397
Graham AL, Shuker DM, Pollitt LC, Auld SKJR, Wilson AJ, Little TJ (2011) Fitness consequences of immune responses: strengthening the empirical framework for ecoimmunology. Funct Ecol 25:5–17
Griffith SC, Owens IPF, Thuman KA (2002) Extra-pair paternity in birds: a review of interspecific variation and adaptive function. Mol Ecol 11:2195–2212
Guinan JA, Gowaty PA, Eltzroth EK (2008) Western bluebird (Sialia mexicana). In: Poole A (ed) The birds of North America online. Cornell Lab of Ornithology, Ithaca. doi:10.2173/bna.510, Retrieved from the Birds of North America Online: http://bna.birds.cornell.edu/bna/species/510
Hamilton WD, Zuk M (1982) Heritable true fitness and bright birds: a role for parasites? Science 218:384–387
Hanssen SA, Hasselquist D, Folstad I, Erikstad KE (2004) Costs of immunity: immune responsiveness reduces survival in a vertebrate. Proc R Soc Lond B 271:925–930
Jacobs AC (2013) Parasites, immune response, and sexual selection in western bluebirds. Dissertation, University of California
Jacobs AC, Zuk M (2012) Parasites and sexual selection: do mechanisms matter? In: Demas GE, Nelson RJ (eds) Eco-immunology. Oxford University Press, New York, pp 497–529
Jacobs AC, Reader LL, Fair JM (2013) What determines the rates of double brooding in the western bluebird? Condor 115:386–393
Kalinowski ST, Taper ML, Marshall TC (2007) Revising how the computer program CERVUS accommodates genotyping error increases success in paternity assignment. Mol Ecol 16:1099–1106
Kim S-Y, Fargallo JA, Vergara P, Martinez-Padilla J (2013) Multivariate heredity of melanin-based coloration, body mass and immunity. Heredity 111:139–146
Kozma JM, Kroll AJ (2010) Nest survival of western bluebirds using tree cavities in managed ponderosa pine forests of central Washington. Condor 112:87–95
Lessells CM, Boag PT (1987) Unrepeatable repeatabilities: a common mistake. Auk 104:116–121
Liebl AL, Martin LB (2009) Simple quantification of blood and plasma antimicrobial capacity using spectrophotometry. Funct Ecol 23:1091–1096
Lindström K, Lundström J (2000) Male greenfinches (Carduelis chloris) with brighter ornaments have higher virus infection clearance rate. Behav Ecol Sociobiol 48:44–51
Martinsen ES, Perkins SL, Schall JJ (2008) A three-genome phylogeny of malaria parasites (Plasmodium and closely related genera): evolution of life-history traits and host switches. Mol Phylogenet Evol 47:261–273
Merino S, Moreno J, Sanz JJ, Arriero E (2000) Are avian blood parasites pathogenic in the wild? A medication experiment in blue tits (Parus caeruleus). Proc R Soc Lond B 267:2507–2510
Millet S, Bennett J, Lee KA, Hau M, Klasing KC (2007) Quantifying and comparing constitutive immunity across avian species. Dev Comp Immunol 31:188–201
Møller AP (1990) Effects of a haematophagous mite on the barn swallow (Hurundo rustica). Evolution 44:771–784
Møller AP (1991) Parasite load reduces song output in a passerine bird. Anim Behav 41:723–730
Møller AP, Christe P, Lux E (1999) Parasitism, host immune function, and sexual selection. Q Rev Biol 74:3–20
Mougeot F, Evans SA, Redpath SM (2005) Interactions between population processes in a cyclic species: parasites reduce autumnal territorial behaviour of male red grouse. Oecologia 144:289–298
New Mexico Incident Information System [online] (2011) Las Conchas. http://www.inciweb.org/incident/2385/ (May 11 2012)
Nolan PM, Hill GE, Stoehr AM (1998) Sex, size, and plumage redness predict house finch survival in an epidemic. Proc R Soc Lond B 265:961–965
O’Brien EL, Dawson RD (2011) Plumage color and food availability affect male reproductive success in a socially monogamous bird. Behav Ecol 22:66–72
Palacios MG, Cunnick JE, Winkler DW, Vleck CM (2012) Interrelations among immune defense indices reflect major components of the immune system in a free-living vertebrate. Physiol Biochem Zool 85:1–10
Pérez-Tris J, Hasselquist D, Hellgren O, Krizanauskiene A, Waldenström J, Bensch S (2005) What are malaria parasites? Trends Parasitol 21:209–211
Råberg L, Stjernman M (2003) Natural selection on immune responsiveness in blue tits Parus caeruleus. Evolution 57:1670–1678
Raouf SA, Parker PG, Ketterson ED, Nolan V Jr, Ziegenfus C (1997) Testosterone affects reproductive success by influencing extra-pair fertilizations in male dark-eyed juncos (Aves: Junco hyemalis). Proc R Soc Lond B 264:1599–1603
Ricklefs RE, Fallon SM (2002) Diversification and host switching in avian malaria parasites. Proc R Soc Lond B 269:885–892
Salvante KG (2006) Techniques for studying integrated immune function in birds. Auk 123:575–586
SAS Institute (2014) JMP version 11. SAS Institute Inc, Cary
Schmid-Hempel P (2003) Variation in immune defense as a question of evolutionary ecology. Proc R Soc Lond B 270:357–366
Schmid-Hempel P (2008) Parasite immune evasion: a momentous molecular war. Trends Ecol Evol 23:318–326
Sheldon BC, Verhulst S (1996) Ecological immunity: costly parasite defense and trade-offs in evolutionary ecology. Trends Ecol Evol 11:317–321
Shizuka D, Dickinson JL (2005) Using molt limits to age western bluebirds. J Field Ornithol 76:193–196
Siefferman L, Hill GE (2003) Structural and melanin coloration indicate parental effort and reproductive success in male eastern bluebirds. Behav Ecol 14:855–861
Siefferman LM, Hill GE (2005) Blue structural coloration of male eastern bluebirds Sialia sialis predicts incubation provisioning to females. J Avian Biol 36:488–493
Stjernman M, Råberg L, Nilsson J (2008) Maximum host survival at intermediate parasite infection intensities. PLoS ONE 3:e2463
Viney ME, Riley EM, Buchanan KL (2005) Optimal immune responses: immunocompetence revisited. Trends Ecol Evol 20:665–669
Waldenström J, Bensch S, Hasselquist D, Östman Ö (2004) A new nested polymerase chain reaction method very efficient in detecting Plasmodium and Haemoproteus infections from avian blood. J Parasitol 90:191–194
Wilcoxen TE, Boughton RK, Schoech SJ (2010) Selection on innate immunity and body condition in Florida scrub-jays throughout an epidemic. Biol Lett 6:552–554
Zahavi A (1975) Mate selection: a selection for a handicap. J Theor Biol 53:205–214
Zahavi A (1977) The cost of honesty (further remarks on the handicap principle). J Theor Biol 67:603–605
Zehtindjiev P, Ilieva M, Westerdahl H, Hansson B, Valkiūnas G, Bensch S (2008) Dynamics of parasitemia of malaria parasites in a naturally and experimentally infected migratory songbird, the great reed warbler Acrocephalus arundinaceus. Exp Parasitol 119:99–110
Zuk M, Stoehr AM (2002) Immune defense and host life history. Am Nat 160:S9–S22
Zuk M, Thornhill R, Ligon JD, Johnson K (1990) Parasites and mate choice in red jungle fowl. Am Zool 30:235–244
Acknowledgments
We are grateful to K. Burnett, C. Hathcock, D. Keller, L. Maestas, R. Robinson, L. Reader, and C. Lemanski for assistance in the field. We thank B. Faircloth, J. Burnette, L. Enders, L. Nunney, and A. Cortez for invaluable advice and assistance regarding the molecular analyses. We thank Y. Shou, K. McCabe, E. Hong-Geller, and the Bradbury lab for assistance with the immune assays and M. Venesky for help with the statistical analyses. This project was funded by the University of California, Riverside and Los Alamos National Security, LLC, operator of the Los Alamos National Laboratory under contract no. DE-AC52-06NA25396 with the US Department of Energy. Additional funding for molecular work was provided by a Sigma Xi Grant-in-Aid-of-Research, a Ralph Schreiber Memorial Award from the Los Angeles Audubon’s Society, and a National Science Foundation Doctoral Dissertation Improvement Grant awarded to ACJ.
Ethical standards
This experiment was conducted in compliance with the current laws of the USA. Our research protocols were approved by the Animal Care and Use Committee at the Los Alamos National Laboratory (protocol no. 10-60).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by K. McGraw
Rights and permissions
About this article
Cite this article
Jacobs, A.C., Fair, J.M. & Zuk, M. Parasite infection, but not immune response, influences paternity in western bluebirds. Behav Ecol Sociobiol 69, 193–203 (2015). https://doi.org/10.1007/s00265-014-1832-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-014-1832-6