Abstract
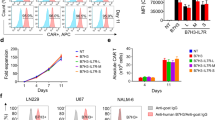
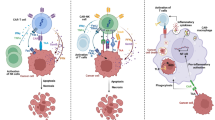
Non-Hodgkin lymphoma (NHL) is an incurable lymphoproliferative cancer, and patients with NHL have a poor prognosis. The present study explored the regulatory mechanism of expression and possible roles of the immunosuppressive B7-H4 molecule in human NHL. For functional studies, NHL-reactive T cell lines were generated via the isolation of allogeneic CD3+ T cells from healthy donors and repeated in vitro stimulation with irradiated NHL cells isolated from patients. B7-H4 was found to be distributed in NHL cells and tissues, and its surface protein expression levels were further upregulated by the incubation of NHL cells with interleukin (IL)-6, IL-10, or interferon-γ. Additionally, the supernatants of tumor-associated macrophages (tMφs) upregulated B7-H4 surface expression by producing IL-6 and IL-10. B7-H4 expressed in NHL cells inhibited the cytotoxic activity of NHL-reactive T cells. Conversely, the inhibition of B7-H4 in NHL cells promoted T cell immunity and sensitized NHL cells to cytolysis. Furthermore, tMφs induced B7-H4 promoted NHL cell evasion of the T cell immune response. In conclusion, this study shows that NHL-expressed B7-H4 is an important immunosuppressive factor that inhibits host anti-tumor immunity to NHL. Targeting tumor-expressed B7-H4 may thus provide a new treatment strategy for NHL patients.







Similar content being viewed by others
Abbreviations
- CTL:
-
Cytotoxic T lymphocyte
- DLBCL:
-
Diffuse large B cell lymphoma
- FasL:
-
Fas ligand
- FL:
-
Follicular lymphoma
- IFN:
-
Interferon
- IL:
-
Interleukin
- IgG:
-
Immunoglobulin G
- LPS:
-
Lipopolysaccharide
- Ly10:
-
OCI-Ly10
- mAb:
-
Monoclonal antibody
- M-CSF:
-
Macrophage colony-stimulating factor
- MHC:
-
Major histocompatibility complex
- Nal:
-
Nalmawa
- NHL:
-
Non-Hodgkin lymphoma
- nMφs:
-
Normal macrophages
- PBMCs:
-
Peripheral blood mononuclear cells
- PD:
-
Programmed death protein
- PD-L:
-
Programmed death ligand
- TGF:
-
Transforming growth factor
- tMφs:
-
Tumor-associated macrophages
- TNF:
-
Tumor necrosis factor
- Tregs:
-
Regulatory T cells
- VEGF:
-
Vascular endothelial growth factor
References
Havranek O, Kleiblova P, Hojny J, Lhota F, Soucek P, Trneny M, Kleibl Z (2015) Association of Germline CHEK2 gene variants with risk and prognosis of Non-Hodgkin lymphoma. PLoS One 10:e0140819. doi:10.1371/journal.pone.0140819
Glass S, Phan A, Williams JN, Flowers CR, Koff JL (2016) Integrating understanding of epidemiology and genomics in B-cell non-Hodgkin lymphoma as a pathway to novel management strategies. Discov Med 21:181–188
Grover NS, Park SI (2015) Novel targeted agents in Hodgkin and non-Hodgkin lymphoma therapy. Pharmaceuticals (Basel) 8:607–636. doi:10.3390/ph8030607
Stienen JJ, Hermens RP, Wennekes L et al (2015) Variation in guideline adherence in non-Hodgkin’s lymphoma care: impact of patient and hospital characteristics. BMC Cancer 15:578. doi:10.1186/s12885-015-1547-8
Zappasodi R, de Braud F, Di Nicola M (2015) Lymphoma immunotherapy: current status. Front Immunol 6:448. doi:10.3389/fimmu.2015.00448
Germain C, Guillaudeux T, Galsgaard ED et al. (2015) Lectin-like transcript 1 is a marker of germinal center-derived B-cell non-Hodgkin’s lymphomas dampening natural killer cell functions. Oncoimmunology 4:e1026503. doi:10.1080/2162402X.2015.1026503
Gabellier L, Cartron G (2016) Obinutuzumab for relapsed or refractory indolent non-Hodgkin’s lymphomas. Ther Adv Hematol 7:85–93. doi:10.1177/2040620715622613
Wang L, Qian J, Lu Y et al (2013) Immune evasion of mantle cell lymphoma: expression of B7-H1 leads to inhibited T-cell response to and killing of tumor cells. Haematologica 98:1458–1466. doi:10.3324/haematol.2012.071340
Hollander N (2012) Immunotherapy for B-cell lymphoma: current status and prospective advances. Front Immunol 3:3. doi:10.3389/fimmu.2012.00003
Sakamaki I, Qin H, Kwak LW (2011) Translational development of vaccination strategies in follicular NHL. Best Pract Res Clin Haematol 24:295–304. doi:10.1016/j.beha.2011.03.007
Schuster SJ, Neelapu SS, Gause BL et al (2011) Vaccination with patient-specific tumor-derived antigen in first remission improves disease-free survival in follicular lymphoma. J Clin Oncol 29:2787–2794. doi:10.1200/JCO.2010.33.3005
Kline J, Bishop MR (2015) Update on checkpoint blockade therapy for lymphoma. J Immunother Cancer 3:33. doi:10.1186/s40425-015-0079-8
Bagley SJ, Bauml JM, Langer CJ (2015) PD-1/PD-L1 immune checkpoint blockade in non-small cell lung cancer. Clin Adv Hematol Oncol 13:676–683
Westin JR, Chu F, Zhang M et al (2014) Safety and activity of PD1 blockade by pidilizumab in combination with rituximab in patients with relapsed follicular lymphoma: a single group, open-label, phase 2 trial. Lancet Oncol 15:69–77. doi:10.1016/S1470-2045(13)70551-5
Choi IH, Zhu G, Sica GL et al (2003) Genomic organization and expression analysis of B7-H4, an immune inhibitory molecule of the B7 family. J Immunol 171:4650–4654
Sica GL, Choi IH, Zhu G et al (2003) B7-H4, a molecule of the B7 family, negatively regulates T cell immunity. Immunity 18:849–861
Salceda S, Tang T, Kmet M, Munteanu A, Ghosh M, Macina R, Liu W, Pilkington G, Papkoff J (2005) The immunomodulatory protein B7-H4 is overexpressed in breast and ovarian cancers and promotes epithelial cell transformation. Exp Cell Res 306:128–141. doi:10.1016/j.yexcr.2005.01.018
Hansen JD, Du Pasquier L, Lefranc MP, Lopez V, Benmansour A, Boudinot P (2009) The B7 family of immunoregulatory receptors: a comparative and evolutionary perspective. Mol Immunol 46:457–472. doi:10.1016/j.molimm.2008.10.007
Wang L, Heng X, Lu Y, Cai Z, Yi Q, Che F (2016) Could B7-H4 serve as a target to activate anti-cancer immunity? Int Immunopharmacol 38:97–103. doi:10.1016/j.intimp.2016.05.020
Cheng C, Qu QX, Shen Y, Lv YT, Zhu YB, Zhang XG, Huang JA (2011) Overexpression of B7-H4 in tumor infiltrated dendritic cells. J Immunoassay Immunochem 32:353–364. doi:10.1080/15321819.2011.578190
Seliger B, Quandt D (2012) The expression, function, and clinical relevance of B7 family members in cancer. Cancer Immunol Immunother 61:1327–1341. doi:10.1007/s00262-012-1293-6
Jeon H, Ohaegbulam KC, Abadi YM, Zang X (2013) B7x and myeloid-derived suppressor cells in the tumor microenvironment: a tale of two cities. Oncoimmunology 2:e24744. doi:10.4161/onci.24744
Arigami T, Uenosono Y, Ishigami S, Hagihara T, Haraguchi N, Natsugoe S (2011) Clinical significance of the B7-H4 coregulatory molecule as a novel prognostic marker in gastric cancer. World J Surg 35:2051–2057. doi:10.1007/s00268-011-1186-4
Shi H, Ji M, Wu J et al (2014) Serum B7-H4 expression is a significant prognostic indicator for patients with gastric cancer. World J Surg Oncol 12:188. doi:10.1186/1477-7819-12-188
Geng Y, Wang H, Lu C, Li Q, Xu B, Jiang J, Wu C (2015) Expression of costimulatory molecules B7-H1, B7-H4 and Foxp3(+) Tregs in gastric cancer and its clinical significance. Int J Clin Oncol 20:273–281. doi:10.1007/s10147-014-0701-7
Matsunaga T, Saito H, Ikeguchi M (2011) Increased B7-H1 and B7-H4 expressions on circulating monocytes and tumor-associated macrophages are involved in immune evasion in patients with gastric cancer. Yonago Acta Med 54:1–10.
Jiang J, Zhu Y, Wu C et al (2010) Tumor expression of B7-H4 predicts poor survival of patients suffering from gastric cancer. Cancer Immunol Immunother 59:1707–1714. doi:10.1007/s00262-010-0900-7
Zhao LW, Li C, Zhang RL, Xue HG, Zhang FX, Zhang F, Gai XD (2014) B7-H1 and B7-H4 expression in colorectal carcinoma: correlation with tumor FOXP3(+) regulatory T-cell infiltration. Acta Histochem 116:1163–1168. doi:10.1016/j.acthis.2014.06.003
Chen Y, Sun J, Zhao H et al. (2014) The coexpression and clinical significance of costimulatory molecules B7-H1, B7-H3, and B7-H4 in human pancreatic cancer. Onco Targets Ther 7:1465–1472. doi:10.2147/OTT.S66809
Mugler KC, Singh M, Tringler B, Torkko KC, Liu W, Papkoff J, Shroyer KR (2007) B7-h4 expression in a range of breast pathology: correlation with tumor T-cell infiltration. Appl Immunohistochem Mol Morphol 15:363–370. doi:10.1097/01.pai.0000213159.79557.71
Li ZY, Zhang XH, Chen Y, Guo JG, Sai K, Yang QY, Chen ZP, Mou YG (2013) Clinical significance of B7-H4 expression in matched non-small cell lung cancer brain metastases and primary tumors. Oncol Targets Ther 6:869–875. doi:10.2147/OTT.S48085
Sun Y, Wang Y, Zhao J, Gu M, Giscombe R, Lefvert AK, Wang X (2006) B7-H3 and B7-H4 expression in non-small-cell lung cancer. Lung Cancer 53:143–151. doi:10.1016/j.lungcan.2006.05.012
Zhu J, Chu BF, Yang YP, Zhang SL, Zhuang M, Lu WJ, Liu YB (2013) B7-H4 expression is associated with cancer progression and predicts patient survival in human thyroid cancer. Asian Pac J Cancer Prev 14:3011–3015
Xu Y, Zhu S, Song M, Liu W, Liu C, Li Y, Wang M (2014) B7-H4 expression and its role in interleukin-2/interferon treatment of clear cell renal cell carcinoma. Oncol Lett 7:1474–1478. doi:10.3892/ol.2014.1961
Jung SG, Choi KU, Lee SD, Lee ZZ, Chung MK (2011) The relationship between B7-H4 expression and clinicopathological characteristics in clinical stage T1 conventional renal cell carcinoma. Korean J Urol 52:90–95. doi:10.4111/kju.2011.52.2.90
Krambeck AE, Thompson RH, Dong H et al (2006) B7-H4 expression in renal cell carcinoma and tumor vasculature: associations with cancer progression and survival. Proc Natl Acad Sci USA 103:10391–10396. doi:10.1073/pnas.0600937103
Zhang L, Wu H, Lu D et al (2013) The costimulatory molecule B7-H4 promote tumor progression and cell proliferation through translocating into nucleus. Oncogene 32:5347–5358. doi:10.1038/onc.2012.600
Quandt D, Fiedler E, Boettcher D, Marsch W, Seliger B (2011) B7-h4 expression in human melanoma: its association with patients’ survival and antitumor immune response. Clin Cancer Res 17:3100–3111. doi:10.1158/1078-0432.CCR-10-2268
Zhang C, Li Y, Wang Y (2015) Diagnostic value of serum B7-H4 for hepatocellular carcinoma. J Surg Res 197:301–306. doi:10.1016/j.jss.2015.04.034
Wang X, Wang T, Xu M, Xiao L, Luo Y, Huang W, Zhang Y, Geng W (2014) B7-H4 overexpression impairs the immune response of T cells in human cervical carcinomas. Hum Immunol 75:1203–1209. doi:10.1016/j.humimm.2014.10.002
Maskey N, Li K, Hu M et al (2014) Impact of neoadjuvant chemotherapy on lymphocytes and co-inhibitory B7-H4 molecule in gastric cancer: low B7-H4 expression associates with favorable prognosis. Tumour Biol 35:11837–11843. doi:10.1007/s13277-014-2410-2
Xu H, Chen X, Tao M, Chen K, Chen C, Xu G, Li W, Yuan S, Mao Y (2016) B7-H3 and B7-H4 are independent predictors of a poor prognosis in patients with pancreatic cancer. Oncol Lett 11:1841–1846. doi:10.3892/ol.2016.4128
Chen X, Wang L, Wang W, Zhao L, Shan B (2016) B7-H4 facilitates proliferation of esophageal squamous cell carcinoma cells through promoting interleukin-6/signal transducer and activator of transcriotion 3 pathway activation. Cancer Sci 107:944–954. doi:10.1111/cas.12949
Abadi YM, Jeon H, Ohaegbulam KC et al (2013) Host b7x promotes pulmonary metastasis of breast cancer. J Immunol 190:3806–3814. doi:10.4049/jimmunol.1202439
Sabattini E, Bacci F, Sagramoso C, Pileri SA (2010) WHO classification of tumours of haematopoietic and lymphoid tissues in 2008: an overview. Pathologica 102:83–87
Wang L, Zhao Y, Qian J et al (2013) Toll-like receptor-4 signaling in mantle cell lymphoma: effects on tumor growth and immune evasion. Cancer 119:782–791. doi:10.1002/cncr.27792
Kikuchi T, Crystal RG (2001) Antigen-pulsed dendritic cells expressing macrophage-derived chemokine elicit Th2 responses and promote specific humoral immunity. J Clin Invest 108:917–927. doi:10.1172/JCI11564
Zheng Y, Cai Z, Wang S et al (2009) Macrophages are an abundant component of myeloma microenvironment and protect myeloma cells from chemotherapy drug-induced apoptosis. Blood 114:3625–3628. doi:10.1182/blood-2009-05-220285
Zindl CL, Chaplin DD (2010) Immunology. Tumor immune evasion. Science 328:697–698. doi:10.1126/science.1190310
Wang S, Yang J, Qian J, Wezeman M, Kwak LW, Yi Q (2006) Tumor evasion of the immune system: inhibiting p38 MAPK signaling restores the function of dendritic cells in multiple myeloma. Blood 107:2432–2439. doi:10.1182/blood-2005-06-2486
Smyth MJ, Godfrey DI, Trapani JA (2001) A fresh look at tumor immunosurveillance and immunotherapy. Nat Immunol 2:293–299. doi:10.1038/86297
Freedman A, Neelapu SS, Nichols C et al (2009) Placebo-controlled phase III trial of patient-specific immunotherapy with mitumprotimut-T and granulocyte-macrophage colony-stimulating factor after rituximab in patients with follicular lymphoma. J Clin Oncol 27:3036–3043. doi:10.1200/JCO.2008.19.8903
Laurent C, Charmpi K, Gravelle P et al. (2015) Several immune escape patterns in non-Hodgkin’s lymphomas. Oncoimmunology 4:e1026530. doi:10.1080/2162402X.2015.1026530
Chen C, Qu QX, Shen Y, Mu CY, Zhu YB, Zhang XG, Huang JA (2012) Induced expression of B7-H4 on the surface of lung cancer cell by the tumor-associated macrophages: a potential mechanism of immune escape. Cancer Lett 317:99–105. doi:10.1016/j.canlet.2011.11.017
Kryczek I, Wei S, Zou L, Zhu G, Mottram P, Xu H, Chen L, Zou W (2006) Cutting edge: induction of B7-H4 on APCs through IL-10: novel suppressive mode for regulatory T cells. J Immunol 177:40–44
Kryczek I, Zou L, Rodriguez P et al (2006) B7-H4 expression identifies a novel suppressive macrophage population in human ovarian carcinoma. J Exp Med 203:871–881. doi:10.1084/jem.20050930
Mirza N, Gabrilovich D (2007) Comment on “Cutting edge: induction of B7-H4 on APCs through IL-10: novel suppressive mode for regulatory T cells”. J Immunol 178:4705–4706 (author reply 4706)
Kagi D, Vignaux F, Ledermann B, Burki K, Depraetere V, Nagata S, Hengartner H, Golstein P (1994) Fas and perforin pathways as major mechanisms of T cell-mediated cytotoxicity. Science 265:528–530
Kojima H, Shinohara N, Hanaoka S et al (1994) Two distinct pathways of specific killing revealed by perforin mutant cytotoxic T lymphocytes. Immunity 1:357–364
Rook GA, Dalgleish A (2011) Infection, immunoregulation, and cancer. Immunol Rev 240:141–159. doi:10.1111/j.1600-065X.2010.00987.x
Kryczek I, Wei S, Zhu G, Myers L, Mottram P, Cheng P, Chen L, Coukos G, Zou W (2007) Relationship between B7-H4, regulatory T cells, and patient outcome in human ovarian carcinoma. Cancer Res 67:8900–8905. doi:10.1158/0008-5472.CAN-07-1866
Morantz RA, Wood GW, Foster M, Clark M, Gollahon K (1979) Macrophages in experimental and human brain tumors. Part 2: studies of the macrophage content of human brain tumors. J Neurosurg 50:305–311. doi:10.3171/jns.1979.50.3.0305
Farinha P, Masoudi H, Skinnider BF et al (2005) Analysis of multiple biomarkers shows that lymphoma-associated macrophage (LAM) content is an independent predictor of survival in follicular lymphoma (FL). Blood 106:2169–2174. doi:10.1182/blood-2005-04-1565
Leich E, Hartmann EM, Burek C, Ott G, Rosenwald A (2007) Diagnostic and prognostic significance of gene expression profiling in lymphomas. APMIS 115:1135–1146. doi:10.1111/j.1600-0463.2007.apm_867.xml.x
Alvaro T, Lejeune M, Camacho FI et al (2006) The presence of STAT1-positive tumor-associated macrophages and their relation to outcome in patients with follicular lymphoma. Haematologica 91:1605–1612
Assaf C, Hwang ST (2016) Mac attack: macrophages as key drivers of cutaneous T-cell lymphoma pathogenesis. Exp Dermatol 25:105–106. doi:10.1111/exd.12894
Shen L, Li H, Shi Y, Wang D, Gong J, Xun J, Zhou S, Xiang R, Tan X (2016) M2 tumour-associated macrophages contribute to tumour progression via legumain remodelling the extracellular matrix in diffuse large B-cell lymphoma. Sci Rep 6:30347. doi:10.1038/srep30347
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This work was supported by Funds for Young Scholars of the National Natural Science Foundation of China (Grant No. 81402353), the Medical Health Science and Technology Development Plan of Shandong Province (Grant No. 2014WS0287), the China Postdoctoral Science Foundation (Grant No. 2015M580594), the Postdoctoral Innovation Foundation of Shandong Province (Grant No. 201502008), the Key Research Project program of Shandong Province (Grant No. 2016GSF201056), and the Natural Science Foundation of Shandong Province (Grant No. ZR2014HM077).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Che, F., Heng, X., Zhang, H. et al. Novel B7-H4-mediated crosstalk between human non-Hodgkin lymphoma cells and tumor-associated macrophages leads to immune evasion via secretion of IL-6 and IL-10. Cancer Immunol Immunother 66, 717–729 (2017). https://doi.org/10.1007/s00262-017-1961-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-017-1961-7