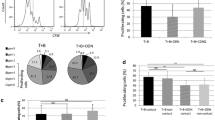
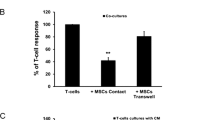
Abstract
Activated T cells from patients with chronic lymphocytic leukemia (CLL) provide survival and proliferative signals to the leukemic clone within lymphoid tissues. Recruitment of both, CLL cells and T lymphocytes, to this supportive microenvironment greatly depends on CXCL12 production by stromal and myeloid cells. CXCL12 also supplies survival stimuli to leukemic B cells, but whether it exerts stimulatory effects on T lymphocytes from CLL patients is unknown. In order to evaluate the capacity of CXCL12 to increase CD4+ T cell activation and proliferation in CLL patients, peripheral blood mononuclear cells were cultured with or without recombinant human CXCL12 or autologous nurse-like cells, and then T cell activation was induced by anti-CD3 mAb. CXCL12 increases the proliferation and the expression of CD25, CD69, CD154, and IFNγ on CD3-stimulated CD4+ T cells from CLL patients, similarly in T cells from ZAP-70+ to ZAP-70− patients. Autologous nurse-like cells establish a close contact with CD4+ T cells and increase their activation and proliferation partially through a CXCR4-dependent mechanism. In addition, we found that activated T cells in the presence of CXCL12 enhance the activation and proliferation of the leukemic clone. In conclusion, CXCL12 production by lymphoid tissue microenvironment in CLL patients might play a key dual role on T cell physiology, functioning not only as a chemoattractant but also as a costimulatory factor for activated T cells.





Similar content being viewed by others
References
Rosati S, Kluin PM (2005) Chronic lymphocytic leukaemia: a review of the immuno-architecture. Curr Top Microbiol Immunol 294:91–107
Soma LA, Craig FE, Swerdlow SH (2006) The proliferation center microenvironment and prognostic markers in chronic lymphocytic leukemia/small lymphocytic lymphoma. Hum Pathol 37(2):152–159
Ghia P, Circosta P, Scielzo C, Vallario A, Camporeale A, Granziero L, Caligaris-Cappio F (2005) Differential effects on CLL cell survival exerted by different microenvironmental elements. Curr Top Microbiol Immunol 294:135–145
Caligaris-Cappio F (2003) Role of the microenvironment in chronic lymphocytic leukaemia. Br J Haematol 123(3):380–388
Shanafelt T, Kay N (2006) T-cell abnormalities in patients with chronic lymphocytic leukemia. Leuk Lymphoma 47(7):1197–1198
Scrivener S, Goddard RV, Kaminski ER, Prentice AG (2003) Abnormal T-cell function in B-cell chronic lymphocytic leukaemia. Leuk Lymphoma 44(3):383–389
Ravandi F, O’Brien S (2006) Immune defects in patients with chronic lymphocytic leukemia. Cancer Immunol Immunother 55(2):197–209
Gorgun G, Holderried TA, Zahrieh D, Neuberg D, Gribben JG (2005) Chronic lymphocytic leukemia cells induce changes in gene expression of CD4 and CD8 T cells. J Clin Invest 115(7):1797–1805
Fluckiger AC, Rossi JF, Bussel A, Bryon P, Banchereau J, Defrance T (1992) Responsiveness of chronic lymphocytic leukemia B cells activated via surface Igs or CD40 to B-cell tropic factors. Blood 80(12):3173–3181
Buske C, Gogowski G, Schreiber K, Rave-Frank M, Hiddemann W, Wormann B (1997) Stimulation of B-chronic lymphocytic leukemia cells by murine fibroblasts, IL-4, anti-CD40 antibodies, and the soluble CD40 ligand. Exp Hematol 25(4):329–337
Bagnara D, Kaufman MS, Calissano C, Marsilio S, Patten PE, Simone R, Chum P, Yan XJ, Allen SL, Kolitz JE, Baskar S, Rader C, Mellstedt H, Rabbani H, Lee A, Gregersen PK, Rai KR, Chiorazzi N (2011) A novel adoptive transfer model of chronic lymphocytic leukemia suggests a key role for T lymphocytes in the disease. Blood 117(20):5463–5472
Nishio M, Endo T, Tsukada N, Ohata J, Kitada S, Reed JC, Zvaifler NJ, Kipps TJ (2005) Nurselike cells express BAFF and APRIL, which can promote survival of chronic lymphocytic leukemia cells via a paracrine pathway distinct from that of SDF-1alpha. Blood 106(3):1012–1020
Burger JA, Tsukada N, Burger M, Zvaifler NJ, Dell’Aquila M, Kipps TJ (2000) Blood-derived nurse-like cells protect chronic lymphocytic leukemia B cells from spontaneous apoptosis through stromal cell-derived factor-1. Blood 96(8):2655–2663
Caligaris-Cappio F, Ghia P (2008) Novel insights in chronic lymphocytic leukemia: are we getting closer to understanding the pathogenesis of the disease? J Clin Oncol 26(27):4497–4503
Burkle A, Niedermeier M, Schmitt-Graff A, Wierda WG, Keating MJ, Burger JA (2007) Overexpression of the CXCR5 chemokine receptor, and its ligand, CXCL13 in B-cell chronic lymphocytic leukemia. Blood 110(9):3316–3325
Burger JA, Kipps TJ (2006) CXCR4: a key receptor in the crosstalk between tumor cells and their microenvironment. Blood 107(5):1761–1767
Tsukada N, Burger JA, Zvaifler NJ, Kipps TJ (2002) Distinctive features of “nurselike” cells that differentiate in the context of chronic lymphocytic leukemia. Blood 99(3):1030–1037
O’Hayre M, Salanga CL, Kipps TJ, Messmer D, Dorrestein PC, Handel TM (2010) Elucidating the CXCL12/CXCR4 signaling network in chronic lymphocytic leukemia through phosphoproteomics analysis. PLoS One 5(7):e11716
Borge M, Nannini PR, Galletti JG, Morande PE, Avalos JS, Bezares RF, Giordano M, Gamberale R (2010) CXCL12-induced chemotaxis is impaired in T cells from patients with ZAP-70-negative chronic lymphocytic leukemia. Haematologica 95(5):768–775
Nanki T, Lipsky PE (2000) Cutting edge: stromal cell-derived factor-1 is a costimulator for CD4+ T cell activation. J Immunol 164(10):5010–5014
Gamberale R, Fernandez-Calotti P, Sanjurjo J, Arrossagaray G, Avalos JS, Geffner J, Giordano M (2005) Signaling capacity of FcgammaRII isoforms in B-CLL cells. Leuk Res 29(11):1277–1284
Crespo M, Bosch F, Villamor N, Bellosillo B, Colomer D, Rozman M, Marce S, Lopez-Guillermo A, Campo E, Montserrat E (2003) ZAP-70 expression as a surrogate for immunoglobulin-variable-region mutations in chronic lymphocytic leukemia. N Engl J Med 348(18):1764–1775
Chattopadhyay PK, Yu J, Roederer M (2006) Live-cell assay to detect antigen-specific CD4+ T-cell responses by CD154 expression. Nat Protoc 1(1):1–6
Crawford MP, Yan SX, Ortega SB, Mehta RS, Hewitt RE, Price DA, Stastny P, Douek DC, Koup RA, Racke MK, Karandikar NJ (2004) High prevalence of autoreactive, neuroantigen-specific CD8+ T cells in multiple sclerosis revealed by novel flow cytometric assay. Blood 103(11):4222–4231
Galletti J, Canones C, Morande P, Borge M, Oppezzo P, Geffner J, Bezares R, Gamberale R, Giordano M (2008) Chronic lymphocytic leukemia cells bind and present the erythrocyte protein band 3: possible role as initiators of autoimmune hemolytic anemia. J Immunol 181(5):3674–3683
D’Arena G, Tarnani M, Rumi C, Vaisitti T, Aydin S, De Filippi R, Perrone F, Pinto A, Chiusolo P, Deaglio S, Malavasi F, Laurenti L (2007) Prognostic significance of combined analysis of ZAP-70 and CD38 in chronic lymphocytic leukemia. Am J Hematol 82(9):787–791
Del Giudice I, Morilla A, Osuji N, Matutes E, Morilla R, Burford A, Maravelaki S, Owusu-Ankomah K, Swansbury J, A’Hern R, Brito-Babapulle V, Catovsky D (2005) Zeta-chain associated protein 70 and CD38 combined predict the time to first treatment in patients with chronic lymphocytic leukemia. Cancer 104(10):2124–2132
Schroers R, Griesinger F, Trumper L, Haase D, Kulle B, Klein-Hitpass L, Sellmann L, Duhrsen U, Durig J (2005) Combined analysis of ZAP-70 and CD38 expression as a predictor of disease progression in B-cell chronic lymphocytic leukemia. Leukemia 19(5):750–758
Hus I, Podhorecka M, Bojarska-Junak A, Rolinski J, Schmitt M, Sieklucka M, Wasik-Szczepanek E, Dmoszynska A (2006) The clinical significance of ZAP-70 and CD38 expression in B-cell chronic lymphocytic leukaemia. Ann Oncol 17(4):683–690
Dighiero G, Binet JL (2000) When and how to treat chronic lymphocytic leukemia. N Engl J Med 343(24):1799–1801
Tangye SG, Weston KM, Raison RL (1997) Cytokines and cross-linking of sIgM augment PMA-induced activation of human leukaemic CD5+ B cells. Immunol Cell Biol 75(6):561–567
Buschle M, Campana D, Carding SR, Richard C, Hoffbrand AV, Brenner MK (1993) Interferon gamma inhibits apoptotic cell death in B cell chronic lymphocytic leukemia. J Exp Med 177(1):213–218
Gamberale R, Geffner JR, Giordano M (2002) Immune complexes and apoptosis in B-cell chronic lymphocytic leukemia. Leuk Lymphoma 43(2):251–255
Ghia P, Strola G, Granziero L, Geuna M, Guida G, Sallusto F, Ruffing N, Montagna L, Piccoli P, Chilosi M, Caligaris-Cappio F (2002) Chronic lymphocytic leukemia B cells are endowed with the capacity to attract CD4+, CD40L+ T cells by producing CCL22. Eur J Immunol 32(5):1403–1413
Patten PE, Buggins AG, Richards J, Wotherspoon A, Salisbury J, Mufti GJ, Hamblin TJ, Devereux S (2008) CD38 expression in chronic lymphocytic leukemia is regulated by the tumor microenvironment. Blood 111(10):5173–5181
Murdoch C (2000) CXCR4: chemokine receptor extraordinaire. Immunol Rev 177:175–184
Molon B, Gri G, Bettella M, Gomez-Mouton C, Lanzavecchia A, Martinez AC, Manes S, Viola A (2005) T cell costimulation by chemokine receptors. Nat Immunol 6(5):465–471
Suzuki Y, Rahman M, Mitsuya H (2001) Diverse transcriptional response of CD4(+) T cells to stromal cell-derived factor (SDF)-1: cell survival promotion and priming effects of SDF-1 on CD4(+) T cells. J Immunol 167(6):3064–3073
Kumar A, Humphreys TD, Kremer KN, Bramati PS, Bradfield L, Edgar CE, Hedin KE (2006) CXCR4 physically associates with the T cell receptor to signal in T cells. Immunity 25(2):213–224
Amara A, Lorthioir O, Valenzuela A, Magerus A, Thelen M, Montes M, Virelizier JL, Delepierre M, Baleux F, Lortat-Jacob H, Arenzana-Seisdedos F (1999) Stromal cell-derived factor-1alpha associates with heparan sulfates through the first beta-strand of the chemokine. J Biol Chem 274(34):23916–23925
Balabanian K, Lagane B, Infantino S, Chow KY, Harriague J, Moepps B, Arenzana-Seisdedos F, Thelen M, Bachelerie F (2005) The chemokine SDF-1/CXCL12 binds to and signals through the orphan receptor RDC1 in T lymphocytes. J Biol Chem 280(42):35760–35766
Granziero L, Ghia P, Circosta P, Gottardi D, Strola G, Geuna M, Montagna L, Piccoli P, Chilosi M, Caligaris-Cappio F (2001) Survivin is expressed on CD40 stimulation and interfaces proliferation and apoptosis in B-cell chronic lymphocytic leukemia. Blood 97(9):2777–2783
Gamberale R, Geffner J, Arrosagaray G, Scolnik M, Salamone G, Trevani A, Vermeulen M, Giordano M (2001) Non-malignant leukocytes delay spontaneous B-CLL cell apoptosis. Leukemia 15(12):1860–1867
Acknowledgments
The authors would like to thank Beatriz Loria and Edit Mabel Horvat for technical assistance. We also thank CLL patients who participated in the study. This work was supported by grants from Agencia Nacional de Promoción Científica y Tecnológica, CONICET and Fundación Roemmers. M. B. designed and did experiments and created the figures; P. R. N. and P. E. M contributed in the analysis and interpretation of the data; C. J. contributed in the analysis and interpretation of the data and collaborated in confocal microscopy assay; R. F. B. and A. B. provided patients samples and advice; M. G. participated in the project conception and critically reviewed the manuscript, which was written by M. B. and R. G.; R. G. designed and supervised the study.
Conflict of interest
The authors reported no potential conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Borge, M., Nannini, P.R., Morande, P.E. et al. CXCL12 is a costimulator for CD4+ T cell activation and proliferation in chronic lymphocytic leukemia patients. Cancer Immunol Immunother 62, 113–124 (2013). https://doi.org/10.1007/s00262-012-1320-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-012-1320-7