Abstract
Purpose
The relationship between tumor metabolism and stage, subcutaneous and visceral fat thickness, and their glucose metabolism and overall survival in patients recently diagnosed with pancreatic carcinoma was assessed.
Methods
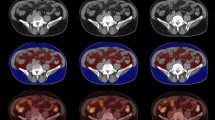
Thirty-eight consecutive patients were studied. Subcutaneous fat thickness (SFT), visceral fat thickness (VFT), and their corresponding FDG SUVmean, as well as SUVmean and SUVmax values of the primary tumor (PT) were derived from FDG-PET CT imaging. Results obtained as well as clinical variables obtained, including gender and BMI, were related to patient outcome. Median follow-up was 382 days (range: 36–917 days).
Results
Median age was 66 years (13 women). Mean BMI was 24.6 (SD: 4.5). Lymph node (LN) involvement was diagnosed in 17 patients and 14 patients presented with distant metastases. Mean SUV max and SUVmean values of the PT were 9.0 (SD 5.9) and 4.2 (SD 2.1). Mean values of SFT and VFT were, respectively, 11.9 mm (range 1–31.7 mm) and 11.5 mm (range 0–49.8 mm). The corresponding SUVmean values were 0.4 (range 0–1.0) and 0.6 (range 0.0–1.6). SUVmean values of SFT proved significantly lower in LNpositive versus LNnegative patients (p = 0.021), in patients with and without metastatic disease (p = 0.017) and in stage III+IV patients versus stage I+II patients (p = 0.03). An inverse logarithmic relationship was found between SUVmean values of subcutaneous fat and SUVmean values of the PT (p = 0.02). Only disease stage dichotomized according to stage I+IIA versus stage IIB+III+IV was predictive of overall survival (p = 0.05).
Conclusion
Glucose metabolism of subcutaneous fat in de novo diagnosed pancreas carcinoma patients presenting with lymph node involvement and metastatic disease is significantly reduced and inversely correlated to the primary tumor metabolism. Of the various fat-related variables studied, none proved significantly related to outcome.




Similar content being viewed by others
References
Fearon KC. Cancer cachexia: developing multimodal therapy for a multidimensional problem. Eur J Cancer. 2008;44:1124–32.
Murphy R, Wilke M, Perrine M, Pawlowicz M, Mourtzakis M, Lieffers J, et al. Loss of adipose tissue and plasma phospholipids: relationship to survival in advanced cancer patients. Clin Nutr. 2010;29:482–7.
Fouladiun M, Korner U, Bosaeus I, Daneryd P, Hyltlander A, Lundholm K. Body composition and time course changes in regional distribution of fat and lean tiussue in unselected cancer patients on palliative care; correlation with food intake, metabolism, exercise capacity and hormones. Cancer. 2005;103:2189–98.
Tisdale M. Cahcexia in cancer patients. Nat Rev Cancer. 2002;2:862–71.
Ebadi M, Mazurak V. Evidence and mechanisms of fat depletion in cancer. Nutrients. 2014;6:5280–97.
Aisenberg A. The glycolysis and respiration of tumors. New York: Academic; 1961. p. 12–3.
Zaidi N, Lupien L, Kuemmerle N, Kinlaw W, Swinnen J, Smans K. Lipogenesis and lipolysis: the pathways exploited by the cancer cells to acquire fatty acids. Prog Lipid Res. 2013;52:585–9.
Kuemmerle N et al. Lipoprotein lipase links dietary fat to solid tumor cell proliferation. Mol Cancer Ther. 2011;10:427–36.
Carpelan-Holmstrom M, Nordling S, Pukkala E, Sankila R, Luttges J, Kloppel G, et al. Does anyone survive pancreatic ductal adenocarcinoma? A nationwide study re-evaluating the data of the Finnish Cancer registry. Gut. 2005;54:385–7.
Ockenga J, Pirlich M, Gastell S, Lochs H. Tumour anorexia-tumour cachexia in case of gastrointestinal tumours: standards and visions. Z Gastroenterol. 2002;40:929–36.
Oliveira A, Azevedo D, Bredella M, Stankey T, Torriani M. Visceral and subcutaneous adipose tissue FDG uptake by PET/CT in metabolically healthy obese subjects. Obesity. 2015;23:286–9.
Christen T, Sheikine Y, Rocha V, Hurwitz S, Goldfine A, Di Carli M, et al. Increased glucose uptake in visceral versus subcutaneous adipose tissue revealed by PET imaging. JACC Cardiovasc Imaging. 2010;3:843–51.
Tahara N, Yamagishi S, Kodama N, Tahara A, Honda A, Nitta Y, et al. Clinical and biochemical factors associated with area and metabolic activity in the visceral and subcutaneous adipose tissues by FDG-PET/CT. J Clin Endocrinol Metab. 2015;100:E739–47.
Rebuffe-Scrive M, Guy-Grand B. Lipogenesis in human adipose tissue in vitro: effect of fat cell size on some enzymatic activities. Diabete Metab. 1979;5:129–33.
Ryden M, Agustsson T, Lauernckinie J, Britton T, Sjoil E, Isaksson B, et al. Lipolysis – Not inflammation, cell death, or lipogenesis- is involved in adipose tissue loss in cancer cahchexia. Cancer. 2008;113:1695–704.
Dahlman I, Mejhert N, Linder K, Agustsson T, Mutch D, Kulyte A, et al. Adipose tissue pathways involved in weight loss of cancer cachexia. Br J Cancer. 2010;102:1541–8.
Mracek T, Stephens N, Gao D, Bao Y, Ross J, Ryden M, et al. Enhanced ZAG production by subcutaneous adipose tissue is linked to weight loss in gastrointestinal cancer patients. Br J Cancer. 2011;104:441–7.
Swinnen J, Brusselmans K, Verhoeven G. Increased lipogenesis in cancer cells: new players, novel targets. Curr Opin Clin Nutr Metab Care. 2006;9:358–65.
Rysman E, Brusselmans K, Scheys K, Timmermans L, Derua R, Munck S, et al. Cancer Res. 2010;70:8117–26.
Mori M, Yamaguchi K, Honda S, Nagasaki K, Ueda M, Abe O, et al. Cancer cachexia syndrome developped in nude mice bearing melanoma cells producing leukemia-inhibitory factor. Cancer Res. 1991;51:6656–9.
Fried S, Zechner R. Cachectin/tumor necrosis factor decreases human adipose tissue lipoprotein lipase mRNA levels, synthesis, and activity. J Lipid Res. 1989;30:1917–23.
Karayiannakis A, Syrigos K, Polychronidis A, Pitiakoudis M, Bounovas A, Simopoulos K. Serum levels of tumor necrosis factor-alpha and nutritional status in pancreatic cancer patients. Anticancer Res. 2001;21:1355–8.
Fleming J, Gonzalez R, Petzel M, Lin E, Morris J, Gomez H, et al. Influence of obesity on cancer-related outcomes after pancreatectomy to treat pancreatic adenocarcinoma. Arch Surg. 2009;144:216–21.
Donohoe C, O’Farelle N, Doyle S, Reynolds J. The role of obesity in gastrointestinal cancer: evidence and opinion. Ther Adv Gastroenterol. 2014;7:38–50.
Gaujoux S, Torres J, Olson S, Winston C, Gonen M, Brennan M, et al. Impact of obesity and body fat on survival after pancreaticoduodenectomy for pancreatic adenocarcinoma. Ann Surg Oncol. 2012;19:2908–16.
Bucerius J, Venkatesh M, Wong S, Moncrieff C, Izquierdo-Garcia D, Machac J, et al. Arterial and fat tissue inflammation are highly correlated – a prospective 18F-FDG PET/CT study. Eur J Nucl Med Mol Imaging. 2014;41:934–45.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in this study including patients were in accordance with the Ethical Standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
No funding was received for this study.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Van de Wiele, C., Van Vlaenderen, M., D’Hulst, L. et al. Metabolic and morphological measurements of subcutaneous and visceral fat and their relationship with disease stage and overall survival in newly diagnosed pancreatic adenocarcinoma. Eur J Nucl Med Mol Imaging 44, 110–116 (2017). https://doi.org/10.1007/s00259-016-3525-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-016-3525-z