Abstract
Purpose
To dynamically detect and characterize 18F-fluorodeoxyglucose (FDG) dose infiltrations and evaluate their effects on positron emission tomography (PET) standardized uptake values (SUV) at the injection site and in control tissue.
Methods
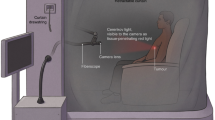
Investigational gamma scintillation sensors were topically applied to patients with locally advanced breast cancer scheduled to undergo limited whole-body FDG-PET as part of an ongoing clinical study. Relative to the affected breast, sensors were placed on the contralateral injection arm and ipsilateral control arm during the resting uptake phase prior to each patient’s PET scan. Time-activity curves (TACs) from the sensors were integrated at varying intervals (0–10, 0–20, 0–30, 0–40, and 30–40 min) post-FDG and the resulting areas under the curve (AUCs) were compared to SUVs obtained from PET.
Results
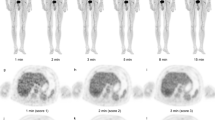
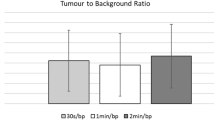
In cases of infiltration, observed in three sensor recordings (30 %), the injection arm TAC shape varied depending on the extent and severity of infiltration. In two of these cases, TAC characteristics suggested the infiltration was partially resolving prior to image acquisition, although it was still apparent on subsequent PET. Areas under the TAC 0–10 and 0–20 min post-FDG were significantly different in infiltrated versus non-infiltrated cases (Mann–Whitney, p < 0.05). When normalized to control, all TAC integration intervals from the injection arm were significantly correlated with SUVpeak and SUVmax measured over the infiltration site (Spearman ρ ≥ 0.77, p < 0.05). Receiver operating characteristic (ROC) analyses, testing the ability of the first 10 min of post-FDG sensor data to predict infiltration visibility on the ensuing PET, yielded an area under the ROC curve of 0.92.
Conclusions
Topical sensors applied near the injection site provide dynamic information from the time of FDG administration through the uptake period and may be useful in detecting infiltrations regardless of PET image field of view. This dynamic information may also complement the static PET image to better characterize the true extent of infiltrations.




Similar content being viewed by others
References
Boellaard R. Standards for PET image acquisition and quantitative data analysis. J Nucl Med. 2009;50 Suppl 1:11S–20S. doi:10.2967/jnumed.108.057182.
Hall NC, Zhang J, Reid R, Hurley D, Knopp MV. Impact of FDG extravasation on SUV measurements in clinical PET/CT. Should we routinely scan the injection site? J Nucl Med. 2006;47(Supplement 1):115P.
Helm RE, Klausner JD, Klemperer JD, Flint LM, Huang E. Accepted but unacceptable: peripheral IV catheter failure. J Infus Nurs. 2015;38(3):189–203. doi:10.1097/NAN.0000000000000100.
Osman MM, Muzaffar R, Altinyay ME, Teymouri C. FDG dose extravasations in PET/CT: frequency and impact on SUV measurements. Front Oncol. 2011;1:41. doi:10.3389/fonc.2011.00041.
Silva-Rodriguez J, Aguiar P, Sanchez M, Mosquera J, Luna-Vega V, Cortes J, et al. Correction for FDG PET dose extravasations: Monte Carlo validation and quantitative evaluation of patient studies. Med Phys. 2014;41(5):052502. doi:10.1118/1.4870979.
Delbeke D, Coleman RE, Guiberteau MJ, Brown ML, Royal HD, Siegel BA, et al. Procedure guideline for tumor imaging with 18F-FDG PET/CT 1.0. J Nucl Med. 2006;47(5):885–95.
Coleman RE, Hillner BE, Shields AF, Duan F, Merlino DA, Hanna LG, et al. PET and PET/CT reports: observations from the National Oncologic PET Registry. J Nucl Med. 2010;51(1):158–63. doi:10.2967/jnumed.109.066399.
Knowland JG, Scarantino CW, Lattanze RK. System for the detection of gamma radiation from a radioactive analyte. Google Patents. 2013; US 20130324844 A1.
Atuegwu NC, Li X, Arlinghaus LR, Abramson RG, Williams JM, Chakravarthy AB, et al. Longitudinal, intermodality registration of quantitative breast PET and MRI data acquired before and during neoadjuvant chemotherapy: preliminary results. Med Phys. 2014;41(5):052302. doi:10.1118/1.4870966.
Williams JM, Rani SD, Li X, Arlinghaus LR, Lee TC, MacDonald LR, et al. Comparison of prone versus supine 18F-FDG-PET of locally advanced breast cancer: phantom and preliminary clinical studies. Med Phys. 2015;42(7):3801. doi:10.1118/1.4921363.
QIBA FDG-PET/CT Standardized Uptake Value (SUV) Technical Subcommittee. Vendor-neutral pseudo-code for SUV calculation. QIBA/RSNA. http://qibawiki.rsna.org/index.php?title=Standardized_Uptake_Value_(SUV). Accessed 30 Aug 2015.
Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med. 2009;50 Suppl 1:122S–50S. doi:10.2967/jnumed.108.057307.
Kinahan PE, Mankoff DA, Linden HM. The value of establishing the quantitative accuracy of PET/CT imaging. J Nucl Med. 2015;56(8):1133–4. doi:10.2967/jnumed.115.159178.
Weber WA, Gatsonis CA, Mozley PD, Hanna LG, Shields AF, Aberle DR, et al. Repeatability of 18F-FDG PET/CT in advanced non-small cell lung cancer: prospective assessment in 2 multicenter trials. J Nucl Med. 2015;56(8):1137–43. doi:10.2967/jnumed.114.147728.
Bonta DV, Halkar RK, Alazraki N. Extravasation of a therapeutic dose of 131I-metaiodobenzylguanidine: prevention, dosimetry, and mitigation. J Nucl Med. 2011;52(9):1418–22. doi:10.2967/jnumed.110.083725.
Acknowledgments
Funding was provided by the National Cancer Institute (U01 CA142565, P50 CA098131, and P30 CA68485) and the Kleberg Foundation.
Author contributions
JMW, LRA, and TEY participated in study design, implementation, data analysis, and manuscript preparation. SDR, WJG, and SRP were involved in data analysis and manuscript preparation. DWT and RGA participated in study design and manuscript preparation. JGK, RKL, and CWS conceptualized and developed the investigational device and participated in manuscript preparation. MDS, VGA, PP, and ABC were involved in the clinical implementation of the study and manuscript preparation. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure of potential conflicts of interest
WJG, JGK, RKL, SRP, and CWS are employees of Lucerno Dynamics, LLC, which provided the investigational device to Vanderbilt without charge. DWT is a Scientific Advisor for Lucerno.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure 1
(PDF 550 kb)
Supplemental Table 1
(PDF 97 kb)
Rights and permissions
About this article
Cite this article
Williams, J.M., Arlinghaus, L.R., Rani, S.D. et al. Towards real-time topical detection and characterization of FDG dose infiltration prior to PET imaging. Eur J Nucl Med Mol Imaging 43, 2374–2380 (2016). https://doi.org/10.1007/s00259-016-3477-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-016-3477-3