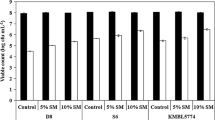
Abstract
A poorly performing industrial water kefir production process consisting of a first fermentation process, a rest period at low temperature, and a second fermentation process was characterized to elucidate the causes of its low water kefir grain growth and instability. The frozen-stored water kefir grain inoculum was thawed and reactivated during three consecutive prefermentations before the water kefir production process was started. Freezing and thawing damaged the water kefir grains irreversibly, as their structure did not restore during the prefermentations nor the production process. The viable counts of the lactic acid bacteria and yeasts on the water kefir grains and in the liquors were as expected, whereas those of the acetic acid bacteria were high, due to the aerobic fermentation conditions. Nevertheless, the fermentations progressed slowly, which was caused by excessive substrate concentrations resulting in a high osmotic stress. Lactobacillus nagelii, Lactobacillus paracasei, Lactobacillus hilgardii, Leuconostoc mesenteroides, Bifidobacterium aquikefiri, Gluconobacter roseus/oxydans, Gluconobacter cerinus, Saccharomyces cerevisiae, and Zygotorulaspora florentina were the most prevalent microorganisms. Lb. hilgardii, the microorganism thought to be responsible for water kefir grain growth, was not found culture-dependently, which could explain the low water kefir grain growth of this industrial process.




Similar content being viewed by others
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Cocolin L, Bisson LF, Mills DA (2000) Direct profiling of the yeast dynamics in wine fermentations. FEMS Microbiol Lett 189:81–87
D’Amore T, Panchal CJ, Russeil I, Stewart GG (1988) Osmotic pressure effects and intracellular accumulation of ethanol in yeast during fermentation. J Ind Microbiol Biotechnol 2:365–372
Edwards U, Rogall T, Blocker H, Emde M, Bottger EC (1989) Isolation and direct complete nucleotide determination of entire genes—characterization of a gene coding for 16S-ribosomal RNA. Nucleic Acids Res 17:7843–7853
Ercolini D, Moschetti G, Blaiotta G, Coppola S (2001) Behavior of variable V3 region from 16S rDNA of lactic acid bacteria in denaturing gradient gel electrophoresis. Curr Microbiol 42:199–202
Franzetti L, Galli A, Pagani MA, De Noni I (1998) Microbiological and chemical investigations on “sugar kefir” drink. Ann Microbiol Enzimol 48:67–80
Garcia-Armisen T, Papalexandratou Z, Hendryckx H, Camu N, Vrancken G, De Vuyst L, Cornelis P (2010) Diversity of the total bacterial community associated with Ghanaian and Brazilian cocoa bean fermentation samples as revealed by a 16S rRNA gene clone library. Appl Microbiol Biotechnol 87:2281–2292
Gulitz A, Stadie J, Ehrmann MA, Ludwig W, Vogel RF (2013) Comparative phylobiomic analysis of the bacterial community of water kefir by 16S rRNA gene amplicon sequencing and ARDRA analysis. J Appl Microbiol 114:1082–1091
Gulitz A, Stadie J, Wenning M, Ehrmann MA, Vogel RF (2011) The microbial diversity of water kefir. Int J Food Microbiol 151:284–288
Gulitz AJ (2013) Analysis of the diversity of water kefir microbiota by culture-dependent and -independent approaches. Dissertation, Technischen Universität Munchen
Hehre EJ (1946) Studies on the enzymatic synthesis of dextran from sucrose. J Biol Chem 163:221–233
Janssens M, Myter N, De Vuyst L, Leroy F (2012) Species diversity and metabolic impact of the microbiota are low in spontaneously acidified Belgian sausages with an added starter culture of Staphylococcus carnosus. Food Microbiol 29:167–177
Laureys D, Cnockaert M, De Vuyst L, Vandamme P (2016) Bifidobacterium aquikefiri sp. nov. isolated from water kefir. Int J Syst Evol Microbiol 66:1281–1286
Laureys D, De Vuyst L (2014) Microbial species diversity, community dynamics, and metabolite kinetics of water kefir fermentation. Appl Environ Microbiol 80:2564–2572
Marsh AJ, Hill C, Ross RP, Cotter PD (2014) Fermented beverages with health-promoting potential: past and future perspectives. Trends Food Sci Technol 38:113–124
Marsh AJ, O’Sullivan O, Hill C, Ross RP, Cotter PD (2013) Sequence-based analysis of the microbial composition of water kefir from multiple sources. FEMS Microbiol Lett 348:79–85
Moinas M, Horisberger M, Bauer H (1980) The structural organization of the Tibi grain as revealed by light, scanning and transmission microscopy. Arch Microbiol 128:157–161
Papalexandratou Z, Falony G, Romanens E, Jimenez JC, Amores F, Daniel HM, De Vuyst L (2011) Species diversity, community dynamics, and metabolite kinetics of the microbiota associated with traditional Ecuadorian spontaneous cocoa bean fermentations. Appl Environ Microbiol 77:7698–7714
Papalexandratou Z, Lefeber T, Bahrim B, Lee OS, Daniel HM, De Vuyst L (2013) Hanseniaspora opuntiae, Saccharomyces cerevisiae, Lactobacillus fermentum, and Acetobacter pasteurianus predominate during well-performed Malaysian cocoa bean box fermentations, underlining the importance of these microbial species for a successful cocoa bean fermentation process. Food Microbiol 35:73–85
Pidoux M (1989) The microbial flora of sugary kefir grain (the gingerbeer plant): biosynthesis of the grain from Lactobacillus hilgardii producing a polysaccharide gel. J Appl Microbiol 5:223–238
Pothakos V, Illeghems K, Laureys D, Spitaels F, Vandamme P, De Vuyst L (2016) Acetic acid bacteria in fermented food and beverage ecosystems. In: Matsushita K, Toyama H, Tonouchi N, Okamoto-Kainuma A (eds) Acetic acid bacteria: ecology and physiology. Springer, Tokyo, pp 73–100
Satokari RM, Vaughan EE, Akkermans ADL, Saarela M, de Vos WM (2001) Bifidobacterial diversity in human feces detected by genus-specific PCR and denaturing gradient gel electrophoresis. Appl Environ Microbiol 67:504–513
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172:4238–4246
White T, Buns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis N, Gelfand D, Snisky J, White T (eds) PCR protocols: a guide to methods and applications. Academic Press, NY, pp 315–322
Acknowledgements
The authors acknowledge their financial support of the Research Council of the Vrije Universiteit Brussel (SRP, IRP, and IOF projects), the Hercules Foundation, the Research Foundation-Flanders (FWO-Vlaanderen), and the KMO-portefeuille (Agentschap Ondernemen). DL was the recipient of a PhD fellowship of the Vrije Universiteit Brussel.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This paper does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Laureys, D., Van Jean, A., Dumont, J. et al. Investigation of the instability and low water kefir grain growth during an industrial water kefir fermentation process. Appl Microbiol Biotechnol 101, 2811–2819 (2017). https://doi.org/10.1007/s00253-016-8084-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-8084-5