Abstract
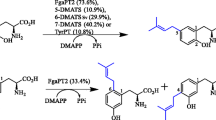
Prenyltransferases of the dimethylallyltryptophan synthase (DMATS) superfamily are involved in the biosynthesis of secondary metabolites and contribute as modification enzymes significantly to structural diversity of natural products. They show usually broad specificity toward their aromatic substrates with regiospecific prenylations on aromatic rings. However, most members of this superfamily exhibit a high specificity toward their prenyl donors and usually accept exclusively dimethylallyl diphosphate (DMAPP). Recently, several indole prenyltransferases from this family were also demonstrated to accept unnatural DMAPP analogs such as methylallyl, 2-pentenyl and benzyl diphosphate for alkylation, or benzylation of the indole ring. Partial or complete shift of the substitution position was observed for these enzymes. In this study, we report the acceptance of these DMAPP analogs by two tyrosine O-prenyltransferases TyrPT from Aspergillus niger and SirD from Leptosphaeria maculans for alkylation or benzylation of tyrosine and derivatives. NMR and mass spectrometry (MS) analyses of nine isolated enzyme products confirmed the regiospecific O- or N-alkylation or benzylation at position C-4 of the aromatic ring, which is the same prenylation position of these enzymes in the presence of DMAPP.




Similar content being viewed by others
References
Ahmed F, Ohtsuki T, Aida W, Ishibashi M (2008) Tyrosine derivatives isolated from Streptomyces sp. IFM 10937 in a screening program for TRAIL-resistance-overcoming activity. J Nat Prod 71:1963–1966
Chooi YH, Cacho R, Tang Y (2010) Identification of the viridicatumtoxin and griseofulvin gene clusters from Penicillium aethiopicum. Chem Biol 17:483–494
Chooi YH, Wang P, Fang J, Li Y, Wu K, Wang P, Tang Y (2012) Discovery and characterization of a group of fungal polycyclic polyketide prenyltransferases. J Am Chem Soc 134:9428–9437
Fan A, Li S-M (2014) Prenylation of tyrosine and derivatives by a tryptophan C7-prenyltransferase. Tetrahedron Lett 55:5199–5202
Fan A, Chen H, Wu R, Xu H, Li S-M (2014) A new member of the DMATS superfamily from Aspergillus niger catalyzes prenylations of both tyrosine and tryptophan derivatives. Appl Microbiol Biotechnol 98:10119–10129
Fan A, Zocher G, Stec E, Stehle T, Li S-M (2015) Site-directed mutagenesis switching a dimethylallyl tryptophan synthase to a specific tyrosine C3-prenylating enzyme. J Biol Chem 290:1364–1373
Gardiner DM, Cozijnsen AJ, Wilson LM, Pedras MS, Howlett BJ (2004) The sirodesmin biosynthetic gene cluster of the plant pathogenic fungus Leptosphaeria maculans. Mol Microbiol 53:1307–1318
Grundmann A, Li S-M (2005) Overproduction, purification and characterization of FtmPT1, a brevianamide F prenyltransferase from Aspergillus fumigatus. Microbiology 151:2199–2207
Heide L (2009) Prenyl transfer to aromatic substrates: genetics and enzymology. Curr Opin Chem Biol 13:171–179
Kremer A, Li S-M (2010) A tyrosine O-prenyltransferase catalyses the first pathway-specific step in the biosynthesis of sirodesmin PL. Microbiology 156:278–286
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Li S-M (2010) Prenylated indole derivatives from fungi: structure diversity, biological activities, biosynthesis and chemoenzymatic synthesis. Nat Prod Rep 27:57–78
Liebhold M, Li S-M (2013) Regiospecific benzylation of tryptophan and derivatives catalyzed by a fungal dimethylallyl transferase. Org Lett 15:5834–5837
Liebhold M, Xie X, Li S-M (2012) Expansion of enzymatic Friedel-Crafts alkylation on indoles: acceptance of unnatural beta-unsaturated allyl diphospates by dimethylallyl-tryptophan synthases. Org Lett 14:4884–4885
Liebhold M, Xie X, Li S-M (2013) Breaking cyclic dipeptide prenyltransferase regioselectivity by unnatural alkyl donors. Org Lett 15:3062–3065
Pockrandt D, Li S-M (2013) Geranylation of cyclic dipeptides by the dimethylallyl transferase AnaPT resulting in a shift of prenylation position on the indole ring. Chembiochem 14:2023–2028
Pockrandt D, Ludwig L, Fan A, König GM, Li S-M (2012) New insights into the biosynthesis of prenylated xanthones: XptB from Aspergillus nidulans catalyses an O-prenylation of xanthones. Chembiochem 13:2764–2771
Pockrandt D, Sack C, Kosiol T, Li S-M (2014) A promiscuous prenyltransferase from Aspergillus oryzae catalyses C-prenylations of hydroxynaphthalenes in the presence of different prenyl donors. Appl Microbiol Biotechnol 98:4987–4994
Rudolf JD, Poulter CD (2013) Tyrosine O-prenyltransferase SirD catalyzes S-, C-, and N-prenylations on tyrosine and tryptophan derivatives. ACS Chem Biol 8:2707–2714
Tarcz S, Xie X, Li S-M (2014) Substrate and catalytic promiscuity of secondary metabolite enzymes: O-prenylation of hydroxyxanthones with different prenyl donors by a bisindolyl benzoquinone C- and N-prenyltransferase. RSC Adv 4:17986–17992
Unsöld IA, Li S-M (2005) Overproduction, purification and characterization of FgaPT2, a dimethylallyltryptophan synthase from Aspergillus fumigatus. Microbiology 151:1499–1505
Wollinsky B, Ludwig L, Hamacher A, Yu X, Kassack MU, Li S-M (2012) Prenylation at the indole ring leads to a significant increase of cytotoxicity of tryptophan-containing cyclic dipeptides. Bioorg Med Chem Lett 22:3866–3869
Woodside AB, Huang Z, Poulter CD (1988) Trisammonium geranyl diphosphate. Org Synth 66:211–215
Wunsch C, Zou HX, Linne U, Li S-M (2015) C7-prenylation of tryptophanyl and O-prenylation of tyrosyl residues in dipeptides by an Aspergillus terreus prenyltransferase. Appl Microbiol Biotechnol 99:1719–1730
Yazaki K, Sasaki K, Tsurumaru Y (2009) Prenylation of aromatic compounds, a key diversification of plant secondary metabolites. Phytochemistry 70:1739–1745
Yin W-B, Grundmann A, Cheng J, Li S-M (2009) Acetylaszonalenin biosynthesis in Neosartorya fischeri: identification of the biosynthetic gene cluster by genomic mining and functional proof of the genes by biochemical investigation. J Biol Chem 284:100–109
Yin W-B, Yu X, Xie X-L, Li S-M (2010) Preparation of pyrrolo[2,3-b]indoles carrying a ß-configured reverse C3-dimethylallyl moiety by using a recombinant prenyltransferase CdpC3PT. Org Biomol Chem 8:2430–2438
Yin S, Yu X, Wang Q, Liu XQ, Li S-M (2013) Identification of a brevianamide F reverse prenyltransferase BrePT from Aspergillus versicolor with a broad substrate specificity towards tryptophan-containing cyclic dipeptides. Appl Microbiol Biotechnol 97:1649–1660
Yu X, Li S-M (2011) Prenylation of flavonoids by using a dimethylallyltryptophan synthase 7-DMATS from Aspergillus fumigatus. Chembiochem 12:2280–2283
Yu X, Li S-M (2012) Prenyltransferases of the dimethylallyltryptophan synthase superfamily. Methods Enzymol 516:259–278
Yu X, Xie X, Li S-M (2011) Substrate promiscuity of secondary metabolite enzymes: prenylation of hydroxynaphthalenes by fungal indole prenyltransferases. Appl Microbiol Biotechnol 92:737–748
Zou H-X, Xie X, Zheng X-D, Li S-M (2011) The tyrosine O-prenyltransferase SirD catalyzes O-, N-, and C-prenylations. Appl Microbiol Biotechnol 89:1443–1451
Acknowledgments
We thank Nina Zitzer and Stefan Newel for taking MS and NMR spectra, respectively. This work was supported in part by grants from the Deutsche Forschungsgemeinschaft (Li844/4-1 to S.-M L.). Huili Yu is a recipient of a scholarship from the China Scholarship Council.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 761 kb)
Rights and permissions
About this article
Cite this article
Yu, H., Liebhold, M., Xie, X. et al. Tyrosine O-prenyltransferases TyrPT and SirD displaying similar behavior toward unnatural alkyl or benzyl diphosphate as their natural prenyl donor dimethylallyl diphosphate. Appl Microbiol Biotechnol 99, 7115–7124 (2015). https://doi.org/10.1007/s00253-015-6452-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6452-1