Abstract
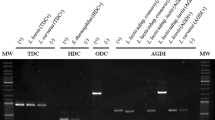
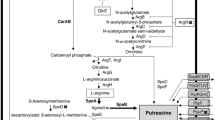
Lactococcus lactis is the most important starter culture organism used in the dairy industry. Although L. lactis species have been awarded Qualified Presumption of Safety status by the European Food Safety Authority, and Generally Regarded as Safe status by the US Food and Drug Administration, some strains can produce the biogenic amine putrescine. One such strain is L. lactis subsp. cremoris CECT 8666 (formerly L. lactis subsp. cremoris GE2-14), which was isolated from Genestoso cheese. This strain catabolizes agmatine to putrescine via the agmatine deiminase (AGDI) pathway, which involves the production of ATP and two ammonium ions. The present work shows that the availability of agmatine and its metabolization to putrescine allows for greater bacterial growth (in a biphasic pattern) and causes the alkalinization of the culture medium in a dose-dependent manner. The construction of a mutant lacking the AGDI cluster (L. lactis CECT 8666 Δagdi) confirmed the latter’s direct role in putrescine production, growth, and medium alkalinization. Alkalinization did not affect the putrescine production pattern and was not essential for increased bacterial growth.





Similar content being viewed by others
References
Bearson S, Bearson B, Foster JW (1997) Acid stress responses in enterobacteria. FEMS Microbiol Lett 147:173–180
Beresford TP, Fitzsimons NA, Brennan NL, Cogan TM (2001) Recent advances in cheese microbiology. Int Dairy J 11:259–274
Cai J, Tong HC, Qi FX, Dong XZ (2012) CcpA-dependent carbohydrate catabolite repression regulates galactose metabolism in Streptococcus oligofermentans. J Bacteriol 194:3824–3832
Chou HT, Kwon DH, Hegazy M, Lu CD (2008) Transcriptome analysis of agmatine and putrescine catabolism in Pseudomonas aeruginosa PAO1. J Bacteriol 190:1966–1975
de Vos WM, Vos P, de Haard H, Boerrigter I (1989) Cloning and expression of the Lactococcus lactis subsp. cremoris SK11 gene encoding an extracellular serine proteinase. Gene 85:169–176
Fernandez M, Linares DM, del Rio B, Ladero V, Alvarez MA (2007) HPLC quantification of biogenic amines in cheeses: correlation with PCR-detection of tyramine-producing microorganisms. J Dairy Res 74:276–282
Fernandez E, Alegria A, Delgado S, Martin MC, Mayo B (2011) Comparative phenotypic and molecular genetic profiling of wild Lactococcus lactis subsp. lactis strains of the L. lactis subsp. lactis and L. lactis subsp. cremoris genotypes, isolated from starter-free cheeses made of raw milk. Appl Environ Microbiol 77:5324–5335
Galgano F, Caruso M, Condelli N, Favati F (2012) Focused review: agmatine in fermented foods. Front Microbiol 3:199
Garcia-Villar N, Hernandez-Cassou S, Saurina J (2009) Determination of biogenic amines in wines by pre-column derivatization and high-performance liquid chromatography coupled to mass spectrometry. J Chromatogr A 1216:6387–6393
Gerner EW, Meyskens FL Jr (2004) Polyamines and cancer: old molecules, new understanding. Nat Rev Cancer 4:781–792
Green MR, Sambrook J (2012) Molecular cloning: a laboratory manual, 4th edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Griswold AR, Jameson-Lee M, Burne RA (2006) Regulation and physiologic significance of the agmatine deiminase system of Streptococcus mutans UA159. J Bacteriol 188:834–841
Griswold AR, Nascimento MM, Burne RA (2009) Distribution, regulation and role of the agmatine deiminase system in mutans streptococci. Oral Microbiol Immunol 24:79–82
Hopwood DA, Bibb MJ, Chater KF, Kieser T, Bruton CJ, Kieser HM, Lydiate DJ, Smith CP, Ward JM, Schrempf H (1985) Genetic manipulation of Streptomyces: a laboratory manual. In: The John Innes Foundation, N., UK and Cold Spring Harbor Laboratory (ed)
Ignatenko NA, Besselsen DG, Roy UK, Stringer DE, Blohm-Mangone KA, Padilla-Torres JL, Guillen RJ, Gerner EW (2006) Dietary putrescine reduces the intestinal anticarcinogenic activity of sulindac in a murine model of familial adenomatous polyposis. Nutr Cancer 56:172–181
Kelly WJ, Ward LJ, Leahy SC (2010) Chromosomal diversity in Lactococcus lactis and the origin of dairy starter cultures. Genome Biol Evol 2:729–744
Kuipers OP (2001) Complete DNA sequence of Lactococcus lactis adds flavor to genomics. Genome Res 11:673–674
Ladero V, Calles-Enriquez M, Fernandez M, Alvarez MA (2010a) Toxicological effects of dietary biogenic amines. Curr Nutr Food Sci 6:145–156
Ladero V, Coton M, Fernandez M, Buron N, Martin MC, Guichard H, Coton E, Alvarez MA (2011a) Biogenic amines content in Spanish and French natural ciders: application of qPCR for quantitative detection of biogenic amine-producers. Food Microbiol 28:554–561
Ladero V, Rattray FP, Mayo B, Martin MC, Fernandez M, Alvarez MA (2011b) Sequencing and transcriptional analysis of the biosynthesis gene cluster of putrescine-producing Lactococcus lactis. Appl Environ Microbiol 77:6409–6418
Ladero V, Cañedo E, Perez M, Martin MC, Fernandez M, Alvarez MA (2012a) Multiplex qPCR for the detection and quantification of putrescine-producing lactic acid bacteria in dairy products. Food Control 27:307–313
Ladero V, Fernandez M, Calles-Enriquez M, Sanchez-Llana E, Cañedo E, Martin MC, Alvarez MA (2012b) Is the production of the biogenic amines tyramine and putrescine a species-level trait in enterococci? Food Microbiol 30:132–138
Ladero V, Fernandez M, Cuesta I, Alvarez MA (2010b) Quantitative detection and identification of tyramine-producing enterococci and lactobacilli in cheese by multiplex qPCR. Food Microbiol 27:933–939
Larsen R, Buist G, Kuipers OP, Kok J (2004) ArgR and AhrC are both required for regulation of arginine metabolism in Lactococcus lactis. J Bacteriol 186:1147–1157
Linares DM, Martin MC, Ladero V, Alvarez MA, Fernandez M (2011) Biogenic amines in dairy products. Crit Rev Food Sci Nutr 51:691–703
Linares DM, del Rio B, Ladero V, Redruello B, Martin MC, Fernandez M, Alvarez MA (2013) The putrescine biosynthesis pathway in Lactococcus lactis is transcriptionally regulated by carbon catabolic repression, mediated by CcpA. Int J Food Microbiol 165:43–50
Llacer JL, Polo LM, Tavarez S, Alarcon B, Hilario R, Rubio V (2007) The gene cluster for agmatine catabolism of Enterococcus faecalis: study of recombinant putrescine transcarbamylase and agmatine deiminase and a snapshot of agmatine deiminase catalyzing its reaction. J Bacteriol 189:1254–1265
Lucas PM, Blancato VS, Claisse O, Magni C, Lolkema JS, Lonvaud-Funel A (2007) Agmatine deiminase pathway genes in Lactobacillus brevis are linked to the tyrosine decarboxylation operon in a putative acid resistance locus. Microbiology 153:2221–2230
Okada T, Ueyama K, Niiya S, Kanazawa H, Futai M, Tsuchiya T (1981) Role of inducer exclusion in preferential utilization of glucose over melibiose in diauxic growth of Escherichia coli. J Bacteriol 146:1030–1037
Redruello B, Ladero V, Cuesta I, Alvarez-Buylla JR, Martin MC, Fernandez M, Alvarez MA (2013) A fast, reliable, ultra high performance liquid chromatography method for the simultaneous determination of amino acids, biogenic amines and ammonium ions in cheese, using diethyl ethoxymethylenemalonate as a derivatising agent. Food Chem 139:1029–1035
Romano A, Trip H, Lonvaud-Funel A, Lolkema JS, Lucas PM (2012) Evidence of two functionally distinct ornithine decarboxylation systems in lactic acid bacteria. Appl Environ Microbiol 78:1953–1961
Seiler N, Atanassov CL, Raul F (1998) Polyamine metabolism as target for cancer chemoprevention (review). Int J Oncol 13:993–1006
Simon JP, Stalon V (1982) Enzymes of agmatine degradation and the control of their synthesis in Streptococcus faecalis. J Bacteriol 152:676–681
Solem C, Defoor E, Jensen PR, Martinussen J (2008) Plasmid pCS1966, a new selection/counterselection tool for lactic acid bacterium strain construction based on the oroP gene, encoding an orotate transporter from Lactococcus lactis. Appl Environ Microbiol 74:4772–4775
Solopova A, van Gestel J, Weissing FJ, Bachmann H, Teusink B, Kok J, Kuipers OP (2014) Bet-hedging during bacterial diauxic shift. Proc Natl Acad Sci U S A 111:7427–7432
Spano G, Russo P, Lonvaud-Funel A, Lucas P, Alexandre H, Grandvalet C, Coton E, Coton M, Barnavon L, Bach B, Rattray F, Bunte A, Magni C, Ladero V, Alvarez M, Fernandez M, Lopez P, de Palencia PF, Corbi A, Trip H, Lolkema JS (2010) Biogenic amines in fermented foods. Eur J Clin Nutr 64(Suppl 3):S95–S100
Suarez C, Espariz M, Blancato VS, Magni C (2013) Expression of the agmatine deiminase pathway in Enterococcus faecalis is activated by the AguR regulator and repressed by CcpA and PTS(Man) systems. PLoS One 8:e76170
Wolken WA, Lucas PM, Lonvaud-Funel A, Lolkema JS (2006) The mechanism of the tyrosine transporter TyrP supports a proton motive tyrosine decarboxylation pathway in Lactobacillus brevis. J Bacteriol 188:2198–2206
Acknowledgments
This work was performed with the financial support of the Spanish Ministry of Economy and Competitiveness (AGL2013-45431-R) and the CSIC (201270E144). B. del Rio and D. Linares were beneficiaries of a JAE DOC contract (CSIC). The authors thank Adrian Burton for the linguistic assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
del Rio, B., Linares, D.M., Ladero, V. et al. Putrescine production via the agmatine deiminase pathway increases the growth of Lactococcus lactis and causes the alkalinization of the culture medium. Appl Microbiol Biotechnol 99, 897–905 (2015). https://doi.org/10.1007/s00253-014-6130-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-6130-8