Abstract
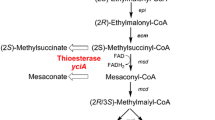
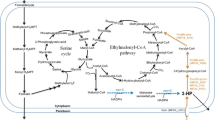
The ethylmalonyl–coenzyme A pathway (EMCP) is a recently discovered pathway present in diverse α-proteobacteria such as the well studied methylotroph Methylobacterium extorquens AM1. Its glyoxylate regeneration function is obligatory during growth on C1 carbon sources like methanol. The EMCP contains special CoA esters, of which dicarboxylic acid derivatives are of high interest as building blocks for chemical industry. The possible production of dicarboxylic acids out of the alternative, non-food competing C-source methanol could lead to sustainable and economic processes. In this work we present a testing of functional thioesterases being active towards the EMCP CoA esters including in vitro enzymatic assays and in vivo acid production. Five thioesterases including TesB from Escherichia coli and M. extorquens, YciA from E. coli, Bch from Bacillus subtilis and Acot4 from Mus musculus showed activity towards EMCP CoA esters in vitro at which YciA was most active. Expressing yciA in M. extorquens AM1 led to release of 70 mg/l mesaconic and 60 mg/l methylsuccinic acid into culture supernatant during exponential growth phase. Our data demonstrates the biotechnological applicability of the thioesterase YciA and the possibility of EMCP dicarboxylic acid production from methanol using M. extorquens AM1.



Similar content being viewed by others
References
Aitipamula S, Chow PS, Tan RBH (2010) Conformational and enantiotropic polymorphism of a 1:1 cocrystal involving ethenzamide and ethylmalonic acid. CrystEngComm 12(11):3691–3697. doi:10.1039/c004491a
Alber B (2011) Biotechnological potential of the ethylmalonyl–CoA pathway. Appl Microbiol Biotechnol 89(1):17–25. doi:10.1007/s00253-010-2873-z
Barnes EM, Wakil SJ, Swindell AC (1970) Purification and properties of a palmityl thioesterase II from Escherichia coli. J Biol Chem 245(12):3122–3128
Bertani G (1951) Studies on lysogenesis: I. The mode of phage liberation by lysogenic Escherichia coli. J Bacteriol 62(3):293–300
Bonner WM, Bloch K (1972) Purification and properties of fatty acyl thioesterase I from Escherichia coli. J Biol Chem 247(10):3123–3133
Cantu DC, Chen Y, Reilly PJ (2010) Thioesterases: a new perspective based on their primary and tertiary structures. Protein Sci 19(7):1281–1295. doi:10.1002/pro.417
Cantu DC, Chen Y, Lemons ML, Reilly PJ (2011) ThYme: a database for thioester-active enzymes. Nucleic Acids Res 39(suppl 1):D342–D346. doi:10.1093/nar/gkq1072
Chen D, Wu R, Bryan TL, Dunaway-Mariano D (2009) In vitro kinetic analysis of substrate specificity in enterobactin biosynthetic lower pathway enzymes provides insight into the biochemical function of the hot dog-fold thioesterase EntH. Biochemistry 48(3):511–513. doi:10.1021/bi802207t
Cho H, Cronan JE (1993) Escherichia coli thioesterase I, molecular cloning and sequencing of the structural gene and identification as a periplasmic enzyme. J Biol Chem 268(13):9238–45
Chubiz L, Purswani J, Carroll S, Marx C (2013) A novel pair of inducible expression vectors for use in Methylobacterium extorquens. BMC Res Notes 6(1):183
Chung A, Liu Q, Ouyang S-P, Wu Q, Chen G-Q (2009) Microbial production of 3-hydroxydodecanoic acid by pha operon and fadBA knockout mutant of Pseudomonas putida KT2442 harboring tesB gene. Appl Microbiol Biotechnol 83(3):513–519. doi:10.1007/s00253-009-1919-6
Di Giulio AV, Bauer JN (1982) Fire-retardant anhydride copolymers. US Patent US 4327197
Erb TJ, Berg IA, Brecht V, Müller M, Fuchs G, Alber BE (2007) Synthesis of C5-dicarboxylic acids from C2-units involving crotonyl-CoA carboxylase/reductase: the ethylmalonyl-CoA pathway. Proc Natl Acad Sci U S A 104(25):10631–10636. doi:10.1073/pnas.0702791104
Erb TJ, Fuchs G, Alber BE (2009) (2S)-Methylsuccinyl-CoA dehydrogenase closes the ethylmalonyl–CoA pathway for acetyl-CoA assimilation. Mol Microbiol 73(6):992–1008. doi:10.1111/j.1365-2958.2009.06837.x
Hu HY, Bailey BJ (1999) Light and thermally stable polyamide. WO Patent WO 1999/046323 A1
Janausch IG, Zientz E, Tran QH, Kröger A, Unden G (2002) C4-dicarboxylate carriers and sensors in bacteria. Biochim Biophys Acta (BBA) Bioenerg 1553(1–2):39–56. doi:10.1016/S0005-2728(01)00233-X
Kato Y, Asano Y (1997) 3-Methylaspartate ammonia-lyase as a marker enzyme of the mesaconate pathway for (S)-glutamate fermentation in Enterobacteriaceae. Arch Microbiol 168(6):457–463. doi:10.1007/s002030050522
Kiefer P, Buchhaupt M, Christen P, Kaup B, Schrader J, Vorholt JA (2009) Metabolite profiling uncovers plasmid-induced cobalt limitation under methylotrophic growth conditions. PLoS ONE 4(11):e7831. doi:10.1371/journal.pone.0007831
Kircher M (2006) White biotechnology: ready to partner and invest in. Biotechnol J 1(7–8):787–794. doi:10.1002/biot.200600087
Lee SY, Park HS, Lee Y, Lee SH (2002) Production of chiral and other valuable compounds from microbial polyesters. In: Doi Y, Steinbüchel A (eds) Biopolymers, polyesters, vol 3. Wiley-VCH, Weinheim, pp 375–387
Lee S-H, Park S, Lee S, Hong S (2008) Biosynthesis of enantiopure (S)-3-hydroxybutyric acid in metabolically engineered Escherichia coli. Appl Microbiol Biotechnol 79(4):633–641. doi:10.1007/s00253-008-1473-7
Li J, Derewenda U, Dauter Z, Smith S, Derewenda ZS (2000) Crystal structure of the Escherichia coli thioesterase II, a homolog of the human Nef binding enzyme. Nat Struct Mol Biol 7(7):555–559
Liu Q, Ouyang S-P, Chung A, Wu Q, Chen G-Q (2007) Microbial production of R-3-hydroxybutyric acid by recombinant E. coli harboring genes of phbA, phbB, and tesB. Appl Microbiol Biotechnol 76(4):811–818. doi:10.1007/s00253-007-1063-0
Liu T, Lin X, Zhou X, Deng Z, Cane DE (2008) Mechanism of thioesterase-catalyzed chain release in the biosynthesis of the polyether antibiotic nanchangmycin. Chem Biol 15(5):449–458. doi:10.1016/j.chembiol.2008.04.006
Loos RL, Mijolovic DM, Heimann JW, Szarka ZJL (2012) Polyesters based on 2-methylsuccinic acid. US Patent US 2012/0245256 A1
Martin CH, Prather KLJ (2009) High-titer production of monomeric hydroxyvalerates from levulinic acid in Pseudomonas putida. J Biotechnol 139(1):61–67. doi:10.1016/j.jbiotec.2008.09.002
Martin CH, Dhamankar H, Tseng HC, Sheppard MJ, Reisch CR, Prather KL (2013) A platform pathway for production of 3-hydroxyacids provides a biosynthetic route to 3-hydroxy-gamma-butyrolactone. Nat Commun 4:1414. doi:10.1038/ncomms2418
Marx CJ, Lidstrom ME (2001) Development of improved versatile broad-host-range vectors for use in methylotrophs and other Gram-negative bacteria. Microbiology 147:2065–2075
Muller B, Richard H (2012) Use of a 2-methylsuccinic acid diester derivative as solvent in cosmetic compositions; cosmetic compositions containing the same. World Patent WO2012119861 A2
Naggert J, Narasimhan ML, DeVeaux L, Cho H, Randhawa ZI, Cronan JE, Green BN, Smith S (1991) Cloning, sequencing, and characterization of Escherichia coli thioesterase II. J Biol Chem 266(17):11044–11050
Nie L, Ren Y, Schulz H (2008) Identification and characterization of Escherichia coli thioesterase III that functions in fatty acid β-oxidation. Biochemistry 47(29):7744–7751. doi:10.1021/bi800595f
Peel D, Quayle JR (1961) Microbial growth on C1 compounds: I. Isolation and characterization of Pseudomonas AM 1. Biochem J 81:465–9
Peyraud R, Kiefer P, Christen P, Massou S, Portais J-C, Vorholt JA (2009) Demonstration of the ethylmalonyl–CoA pathway by using 13C metabolomics. Proc Natl Acad Sci U S A 106(12):4846–4851. doi:10.1073/pnas.0810932106
Peyraud R, Kiefer P, Christen P, Portais JC, Vorholt JA (2012) Co-consumption of methanol and succinate by Methylobacterium extorquens AM1. PLoS ONE 7(11):e48271. doi:10.1371/journal.pone.0048271
Polen T, Spelberg M, Bott M (2013) Toward biotechnological production of adipic acid and precursors from biorenewables. J Biotechnol 167(2):75–84. doi:10.1016/j.jbiotec.2012.07.008
Salis HM (2011) The ribosome binding site calculator (Chapter 2). In: Christopher V (ed) Methods enzymology. vol Volume 498. Academic Press, pp 19–42
Sambrook J, Russell DW (eds) (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor
Sauer M, Porro D, Mattanovich D, Branduardi P (2008) Microbial production of organic acids: expanding the markets. Trends Biotechnol 26(2):100–108. doi:10.1016/j.tibtech.2007.11.006
Schrader J, Schilling M, Holtmann D, Sell D, Filho MV, Marx A, Vorholt JA (2009) Methanol-based industrial biotechnology: current status and future perspectives of methylotrophic bacteria. Trends Biotechnol 27(2):107–115. doi:10.1016/j.tibtech.2008.10.009
Seol W, Shatkin AJ (1991) Escherichia coli kgtP encodes an alpha-ketoglutarate transporter. Proc Natl Acad Sci U S A 88(9):3802–3806. doi:10.1073/pnas.88.9.3802
Shimomura Y, Murakami T, Fujitsuka N, Nakai N, Sato Y, Sugiyama S, Shimomura N, Irwin J, Hawes JW, Harris RA (1994) Purification and partial characterization of 3-hydroxyisobutyryl-coenzyme A hydrolase of rat liver. J Biol Chem 269(19):14248–14253
Shimomura Y, Murakami T, Nakai N, Huang B, Hawes JW, Harris RA (2000) 3-Hydroxyisobutyryl-CoA hydrolase. In: John R. Sokatch RAH (ed) Methods enzymology. vol Volume 324. Academic Press, pp 229–240
Skovran E, Crowther GJ, Guo X, Yang S, Lidstrom ME (2010) A systems biology approach uncovers cellular strategies used by Methylobacterium extorquens AM1 during the switch from multi- to single-carbon growth. PLoS ONE 5(11):e14091. doi:10.1371/journal.pone.0014091
Šmejkalová H, Erb TJ, Fuchs G (2010) Methanol assimilation in Methylobacterium extorquens AM1: demonstration of all enzymes and their regulation. PLoS ONE 5(10):e13001. doi:10.1371/journal.pone.0013001
Song F, Zhuang Z, Finci L, Dunaway-Mariano D, Kniewel R, Buglino JA, Solorzano V, Wu J, Lima CD (2006) Structure, function, and mechanism of the phenylacetate pathway hot dog-fold thioesterase PaaI. J Biol Chem 281(16):11028–11038. doi:10.1074/jbc.M513896200
Suematsu N, Okamoto K, Isohashi F (2002) Mouse cytosolic acetyl-CoA hydrolase, a novel candidate for a key enzyme involved in fat metabolism: cDNA cloning, sequencing and functional expression. Acta Biochim Pol 49(4):937–945
Thakker C, Martínez I, San K-Y, Bennett GN (2012) Succinate production in Escherichia coli. Biotechnol J 7(2):213–224. doi:10.1002/biot.201100061
Toyama H, Anthony C, Lidstrom ME (1998) Construction of insertion and deletion mxa mutants of Methylobacterium extorquens AM1 by electroporation. FEMS Microbiol Lett 166:1–7
Tseng HC, Prather KL (2012) Controlled biosynthesis of odd-chain fuels and chemicals via engineered modular metabolic pathways. Proc Natl Acad Sci U S A 109(44):17925–30. doi:10.1073/pnas.1209002109
Van Dien SJ, Okubo Y, Hough MT, Korotkova N, Taitano T, Lidstrom ME (2003) Reconstruction of C3 and C4 metabolism in Methylobacterium extorquens AM1 using transposon mutagenesis. Microbiology 149(3):601–609. doi:10.1099/mic.0.25955-0
Vuilleumier S, Chistoserdova L, Lee M-C, Bringel F, Lajus A, Zhou Y, Gourion B, Barbe V, Chang J, Cruveiller S, Dossat C, Gillett W, Gruffaz C, Haugen E, Hourcade E, Levy R, Mangenot S, Muller E, Nadalig T, Pagni M, Penny C, Peyraud R, Robinson DG, Roche D, Rouy Z, Saenampechek C, Salvignol G, Vallenet D, Wu Z, Marx CJ, Vorholt JA, Olson MV, Kaul R, Weissenbach J, Médigue C, Lidstrom ME (2009) Methylobacterium genome sequences: a reference blueprint to investigate microbial metabolism of c1 compounds from natural and industrial sources. PLoS ONE 4(5):e5584. doi:10.1371/journal.pone.0005584
Werpy T, Petersen G (2004) Top value added chemicals from biomass: Volume I — Results of screening for potential candidates from sugars and synthesis gas other information: PBD: 1 Aug 2004. p Medium: ED; Size: 76 pp
Westin MAK, Hunt MC, Alexson SEH (2005) The identification of a succinyl-CoA thioesterase suggests a novel pathway for succinate production in peroxisomes. J Biol Chem 280(46):38125–38132. doi:10.1074/jbc.M508479200
Zheng Z, Gong Q, Liu T, Deng Y, Chen J-C, Chen G-Q (2004) Thioesterase II of Escherichia coli plays an important role in 3-hydroxydecanoic acid production. Appl Environ Microbiol 70(7):3807–3813. doi:10.1128/aem.70.7.3807-3813.2004
Zhuang Z, Song F, Martin BM, Dunaway-Mariano D (2002) The YbgC protein encoded by the ybgC gene of the tol–pal gene cluster of Haemophilus influenzae catalyzes acyl-coenzyme A thioester hydrolysis. FEBS Lett 516(1–3):161–163. doi:10.1016/S0014-5793(02)02533-4
Zhuang Z, Song F, Zhao H, Li L, Cao J, Eisenstein E, Herzberg O, Dunaway-Mariano D (2008) Divergence of function in the hot dog fold enzyme superfamily: the bacterial thioesterase YciA†. Biochemistry 47(9):2789–2796. doi:10.1021/bi702334h
Acknowledgments
This work was funded by the European Union in the context of PROMYSE research project (FP7-KBBE.2011.3.6-04). We thank Dr. Tobias Jürgen Erb (Swiss Federal Institute of Technology Zurich, Institute of Microbiology) for his very useful advices on the synthesis and analytics of CoA esters.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sonntag, F., Buchhaupt, M. & Schrader, J. Thioesterases for ethylmalonyl–CoA pathway derived dicarboxylic acid production in Methylobacterium extorquens AM1. Appl Microbiol Biotechnol 98, 4533–4544 (2014). https://doi.org/10.1007/s00253-013-5456-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-013-5456-y