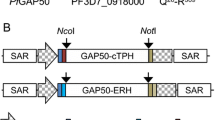
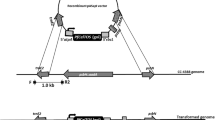
Abstract
Malaria is a widespread and infectious disease that is a leading cause of death in many parts of the world. Eradication of malaria has been a major world health goal for decades, but one that still remains elusive. Other diseases have been eradicated using vaccination, but traditional vaccination methods have thus far been unsuccessful for malaria. Infection by Plasmodium species, the causative agent of malaria, is currently treated with drug-based therapies, but an increase in drug resistance has led to the need for new methods of treatment. A promising strategy for malaria treatment is to combine transmission blocking vaccines (TBVs) that prevent spread of disease with drug-based therapies to treat infected individuals. TBVs can be developed against surface protein antigens that are expressed during parasite reproduction in the mosquito. When the mosquito ingests blood from a vaccinated individual harboring the Plasmodium parasite, the antibodies generated by vaccination prevent completion of the parasites life-cycle. Animal studies have shown that immunization with Pfs48/45 results in the production of malaria transmission blocking antibodies; however, the development of this vaccine candidate has been hindered by poor expression in both prokaryotic and eukaryotic hosts. Recently, the chloroplast of Chlamydomonas reinhardtii has been used to express complex recombinant proteins. In this study, we show that the C-terminal antigenic region of the Pfs48/45 antigen can be expressed in the chloroplast of the green algae C. reinhardtii and that this recombinant protein has a conformation recognized by known transmission blocking antibodies. Production of this protein in algae has the potential to scale to the very large volumes required to meet the needs of millions at risk for contracting malaria.




Similar content being viewed by others
References
Barnes D, Franklin S, Schultz J, Henry R, Brown E, Coragliotti A, Mayfield SP (2005) Contribution of the 5’ and 3’-untranslated regions of the plastid mRNAs to expression of Chlamydomonas reinhardtii chloroplast genes. Mol Gen Genom 274:625–636. doi:10.1007/s00438-005-0055-y
Berrang-Ford I, MacLean JD, Gyorkos TW, Ford JD, Ogden NH (2009) Climate change and malaria in Canada: a systems approach. Interdiscip Perspect Infect Dis 1–13. doi:10.1155/2009/385487
Bousema T, Roeffen W, Meijerink H, Mwerinde H, Mwakalinga S, van Gemert GJ, van de Vegte-Bolmer M, Mosha F, Targett G, Riley EM, Sauerwein R, Drakeley C (2010) The dynamics of naturally acquired immune responses to Plasmodium falciparum sexual stage antigens Pfs230 & Pfs48/45 in a low endemic area in Tanzania. PLoS One 5:e14114. doi:10.1371/journal.pone.0014114
Carter R, Graves PM, Keister DB, Quakyi IA (1990) Properties of epitopes of Pfs 48/45, a target of transmission blocking monoclonal antibodies, on gametes of different isolates of Plasmodium falciparum. Parasite Immunol 12:587–603. doi:10.1111/j.1365-3024.1990.tb00990.x
Chowdhury DR, Angov E, Kariuki T, Kumar N (2009) A potent malaria transmission blocking vaccine based on codon harmonized full length Pfs48/45 expressed in Escherichia coli. PLoS One 4:e6352. doi:10.1371/journal.pone.0006352
Davoodi-Semiromi A, Schreiber M, Nalapalli S, Verma D, Singh ND, Chakrabarti D, Daniell H (2010) Chloroplast-derived vaccine antigens confer dual immunity against cholera and malaria by oral or injectable delivery. Plant Biotech J 8:223–242. doi:10.1111/j.1467-7652.2009.00479.x
Graves PM, Doubrovsky A, Sattabongkot J, Battistutta D (1992) Human antibody responses to epitopes on the Plasmodium falciparum gametocyte antigen Pfs 48/45 and their relationship to infectivity of gametocyte carriers. AmJTrop Med Hyg 46:711–719
Greenwood BM, Targett GAT (2011) Malaria vaccines and the new malaria agenda. Clin Microbiol Infect 17:1600–1607. doi:10.1111/j.1469-0691.2011.03612.x
Greenwood BM, Fidock DA, Kyle DE, Kappe SHI, Alonso PL, Collins FH, Duffy PE (2008) Malaria: progress, perils and prospects for eradication. J Clin Invest 4:1266–1276. doi:10.1172/JCI33996
Kocken CHM, Jansen J, Kaan AM, Beckers PJA, Ponnudurai T, Kaslow DC, Konings RNH, Schoenmakers JGG (1993) Cloning and expression of the gene coding for the transmission blocking target antigen Pfs48/45 of Plasmodium falciparum. Mol Biochem Parasitol 61:59–68. doi:10.1016/0166-6851(93)90158-T
Manuell AL, Beligni MV, Elder JH, Siefker DT, Tran M, Weber A, McDonald TL, Mayfield SP (2007) Robust expression of a bioactive mammalian protein in Chlamydomonas chloroplast. Plant Biotech J 5:402–412. doi:10.1111/j.1467-7652.2007.00249.x
Mayfield SP, Franklin SE, Lerner RA (2003) Expression and assembly of a fully active antibody in algae. Proc Nat Acad Sci 100:438–442. doi:10.1073/pnas.0237108100
Milek RLB, DeVries AAF, Roeffen WFG, Stunnenberg H, Rottier PJM, Konings NH (1998) Plasmodium falciparum: heterologous synthesis of the transmission-blocking vaccine candidate Pfs48/45 in recombinant Vaccinia virus-infected cells. Exper Parasitol 90:165–174. doi:10.1006/expr.1998.4315
Milek RLB, Stunnenberg HG, Konings RNH (2000) Assembly and expression of a synthetic gene encoding the antigen Pfs48/45 of the human malaria parasite Plasmodium falciparum in yeast. Vaccine 18:1402–1411. doi:10.1016/S0264-410X(99)00392-8
Ouédraogo AL, Roeffen W, Luty AJF, de Vlas SJ, Nebie I, Edith IS, Cuzin-Ouattara N, Teleen K, Tiono K, Tiono AB, Sirima SB, Verhave JP, Bousema T, Sauerwein R (2011) Naturally acquired immune responses to Plasmodium falciparum sexual stage antigens Pfs48/45 and Pfs230 in an area of seasonal transmission. Infect Immun 79:4957–4964. doi:10.1128/IAI.05288-11
Outchkourov N, Vermunt A, Jansen J, Kaan A, Roeffen W, Teelen K, Lasonder E, Braks A, van de Vegte-Bolmer M, Qiu LY, Sauerwein R, Stunnenberg HG (2007) Epitope analysis of the malaria surface antigen Pfs48/45 identifies a subdomain that elicits transmission blocking antibodies. J Biol Chem 282:17148–17156. doi:10.1074/jbc.M700948200
Outchkourov NS, Roeffen W, Kaan A, Jansen J, Luty A, Schuiffel D, van Gemert GJ, van de Vegte-Bolmer M, Sauerwein RW, Stunnenberg HG (2008) Correctly folded Pfs48/45 protein of Plasmodium falciparum elicits malaria transmission-blocking immunity in mice. Proc Nat Acad Sci 105:4301–4305. doi:10.1073/pnas.0800459105
Rasala BA, Muto M, Lee PA, Jager M, Cardoso RMF, Behnke CA, Kirk P, Hokanson CA, Crea R, Mendez M, Mayfield SP (2010) Production of therapeutic proteins in algae, analysis of expression of seven human proteins in the chloroplast of Chlamydomonas reinhardtii. Plant Biotech J 8:719–733. doi:10.1111/j.1467-7652.2010.00503.x
Rasala BA, Muto M, Sullivan J, Mayfield SP (2011) Improved heterologous expression in the chloroplast of Chlamydomonas reinhardtii through promoter and 5’ untranslated region optimization. Plant Biotech J 9:674–683. doi:10.1111/j.1467-7652.2011.00620.x
Rener J, Graves PM, Carter R, Williams JL, Burkot TR (1983) Target antigens of transmission-blocking immunity on gametes of Plasmodium falciparum. J Exp Med 158:976–981. doi:10.1084/jem.158.3.976
Saul A (2007) Mosquito stage, transmission blocking vaccines for malaria. Curr Opin Infect Dis 20:476–481. doi:10.1097/QCO.0b013e3282a95e12
Schwartz L, Brown GV, Genton B, Moorthy VS (2012) A review of malaria vaccine clinical projects based on the WHO rainbow table. Malaria J 11:11. doi:10.1186/1475-2875-11-11
Sutherland CJ (2009) Surface antigens of Plasmodium falciparum gametocytes—a new class of transmission-blocking vaccine targets? Mol Biochem Parasitol 166:93–98. doi:10.1016/j.molbiopara.2009.03.007
Targett GA, Greenwood B (2008) Malaria vaccines and their potential role in the elimination of malaria. Malaria J 7:S10. doi:10.1186/1475-2875-7-S1-S10
Targett GAT, Harte PG, Eida S, Rogers NC, Ong CSL (1990) Plasmodium falciparum sexual stage antigens: immunogenicity and cell-mediated responses. Immunol Let 25:77–82. doi:10.1016/0165-2478(90)90095-8
Todryk SM, Hill AVS (2007) Malaria vaccines: the stage we are at. Nat Rev Microbiol 5:487–489. doi:10.1038/nrmicro1712
Tran M, Zhou B, Pettersson PL, Gonzalez MJ, Mayfield SP (2009) Synthesis and assembly of a full-length human monoclonal antibody in algal chloroplasts. Biotech Bioeng 104:663–673. doi:10.1002/bit.22446
Van Dijk MR, Janse CJ, Thompson J, Waters AP, Braks JAM, Dodemont HJ, Stunnenberg HG, van Gemert GJ, Sauerwein RW, Eling W (2001) A central role for Pfs48/45 in malaria parasite male gamete fertility. Cell 104:153–164. doi:10.1016/S0092-8674(01)00199-4
Van Dijk MR, van Schaijk BCL, Khan SM, van Dooren MW, Ramesar J, Kaczanowski S, van Gemert GJ, Kroeze H, Stunnenberg HG, Eling WM, Sauerwein RW, Waters AP, Janse CJ (2010) Three members of the 6-Cys protein family of Plasmodium play a role in gamete fertility. PLOS Pathogens 6:e1000853. doi:10.1371/journal.ppat.1000853
Vermeulen AN, Ponnudurai T, Beckers PJA, Verhave JP, Smits MA, Meuwissen JHE (1985) Sequential expression of antigens on sexual stages of Plasmodium falciparum accessible to transmission-blocking antibodies in the mosquito. J Exp Med 162:1460–1476. doi:10.1084/jem.162.5.1460
Webster DE, Wang L, Mulcair M, Ma C, Santi L, Mason HS, Wesselingh SL, Coppel RL (2009) Production and characterization of an orally immunogenic Plasmodium antigen in plants using a virus-based expression system. Plant Biotech J 7:846–855. doi:10.1111/j.1467-7652.2009.00447.x
Acknowledgments
We acknowledge SAIC subcontract P010020844 and Gabe Gutierrez for initial support for this project. This work was also supported by the San Diego IRACDA Postdoctoral Fellowship from the NIH grant GM06852 and the San Diego Foundation Blasker Science and Technology Grant C-2011-00208. We would also like to acknowledge Joseph Vinetz and laboratory at the University of California, San Diego for the IIC5-10 antibody.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jones, C.S., Luong, T., Hannon, M. et al. Heterologous expression of the C-terminal antigenic domain of the malaria vaccine candidate Pfs48/45 in the green algae Chlamydomonas reinhardtii . Appl Microbiol Biotechnol 97, 1987–1995 (2013). https://doi.org/10.1007/s00253-012-4071-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4071-7