Abstract
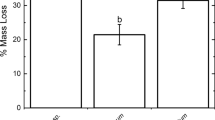
Foliar chemistry influences leaf decomposition, but little is known about how litter chemistry affects the assemblage of bacterial communities during decomposition. Here we examined relationships between initial litter chemistry and the composition of the bacterial community in a stream ecosystem. We incubated replicated genotypes of Populus fremontii and P. angustifolia leaf litter that differ in percent tannin and lignin, then followed changes in bacterial community composition during 28 days of decomposition using 16S rRNA gene-based pyrosequencing. Using a nested experimental design, the majority of variation in bacterial community composition was explained by time (i.e., harvest day) (R 2 = 0.50). Plant species, nested within harvest date, explained a significant but smaller proportion of the variation (R 2 = 0.03). Significant differences in community composition between leaf species were apparent at day 14, but no significant differences existed among genotypes. Foliar chemistry correlated significantly with community composition at day 14 (r = 0.46) indicating that leaf litter with more similar phytochemistry harbor bacterial communities that are alike. Bacteroidetes and β-proteobacteria dominated the bacterial assemblage on decomposing leaves, and Verrucomicrobia and α- and δ-proteobacteria became more abundant over time. After 14 days, bacterial diversity diverged significantly between leaf litter types with fast-decomposing P. fremontii hosting greater richness than slowly decomposing P. angustifolia; however, differences were no longer present after 28 days in the stream. Leaf litter tannin, lignin, and lignin: N ratios all correlated negatively with diversity. This work shows that the bacterial community on decomposing leaves in streams changes rapidly over time, influenced by leaf species via differences in genotype-level foliar chemistry.


Similar content being viewed by others
Reference
Fisher SG, Likens GE (1973) Energy flow in Bear Brook. New Hampshire: an integrative approach to stream ecosystem metabolism. Ecol Monogr 43:421–439
Hieber M, Gessner MO (2002) Contribution of stream detrivores, fungi, and bacteria to leaf breakdown based on biomass estimates. Ecology 83:1026–1038
Suberkropp K, Klug MJ (1976) Fungi and bacteria associated with leaves during processing in a woodland stream. Ecology 57:707–719
Gessner MO, Chauvet E (1994) Importance of stream microfungi in controlling breakdown rates of leaf litter. Ecology 75:1807–1817
Cornwell WK, Cornelissen JHC, Amatangelo K, Dorrepaal E, Eviner VT, Godoy O, Hobbie SE, Hoorens B, Kurokawa H, Pérez-Harguindeguy N, Quested HM, Santiago LS, Wardle DA, Wright IJ, Aerts R, Allison SD, Van Bodegom P, Brovkin V, Chatain A, Callaghan TV, Díaz S, Garnier E, Gurvich DE, Kazakou E, Klein JA, Read J, Reich PB, Soudzilovskaia NA, Vaieretti MV, Westoby M (2008) Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol Lett 11:1065–1107
LeRoy CJ, Whitham TG, Wooley SC, Marks JC (2007) Within species variation in foliar chemistry influences leaf-litter decomposition in a Utah River. J N Am Benthol Soc 26:426–438
Das M, Royer TV, Leff LG (2007) Diversity of fungi, bacteria, and actinomycetes on leaves decomposing in a stream. Appl Environ Microbiol 73:756–767
Fernandes I, Duarte S, Cassio F, Pascoal C (2013) Effects of riparian plant diversity loss on aquatic microbial decomposers become more pronounced with time. Microb Ecol 66:763–772
Harrop BL, Marks JC, Watwood ME (2009) Early bacterial and fungal colonization of leaf litter in Fossil Creek, Arizona. J N Am Benthol Soc 28:383–396
Kominoski JS, Marczak LB, Richardson JS (2011) Riparian forest composition affects stream litter decomposition despite similar microbial and invertebrate communities. Ecology 92:151–159
Kominoski JS, Hoellin TJ, Kelly JJ, Pringle CM (2009) Does mixing litter of different qualities alter stream microbial diversity and functioning on individual litter species? Oikos 118:457–463
Dilly O, Bloem J, Vos A, Munch JC (2004) Bacterial diversity in agricultural soils during litter decomposition. Appl Environ Microbiol 70:468–474
Kelly JJ, Bansal A, Winkelman J, Janus LR, Hell S, Wencel M, Belt P, Kuehn KA, Rier ST, Tuchman NC (2010) Alteration of microbial communities colonizing leaf litter in a temperate woodland stream by growth of trees under condition of elevated atmospheric CO2. Appl Environ Microbiol 76:4950–4959
Marks JC, Haden GA, Harrop BL, Reese EG, Keams JL, Watwood ME, Whitham TG (2009) Genetic and environmental controls of microbial communities on leaf litter in streams. Freshw Biol 54:2616–2627
Duarte S, Pascoal C, Alves A, Correia A, Cassio F (2010) Assessing the dynamic of microbial communities during leaf litter decomposition in a low-order stream by microscopic and molecular techniques. Microbiol Res 165:351–362
Besemer K, Peter H, Logue JB, Langenheder S, Lindström ES, Tranvik LJ, Battin TJ (2012) Unraveling assembly of stream biofilm communities. ISME J 6:1459–1468
Portillo MC, Anderson SP, Fierer N (2012) Temporal variability in the diversity and composition of stream bacterioplankton communities. Environ Microbiol 14:2417–2428
Driebe EM, Whitham TG (2000) Cottonwood hybridization affects tannin and nitrogen content of leaf litter and alters decomposition. Oecologia 123:99–107
Whitham TG, Bailey JK, Schweitzer JA, Shuster SM, Bangert RK, LeRoy CJ, Lonsdorf EV, Allan GJ, DiFazio SP, Potts BM, Fischer DG, Gehring CA, Lindroth RL, Marks JC, Hart SC, Wimp GM, Wooley SC (2006) A framework for community and ecosystem genetics: from genes to ecosystems. Nat Rev Genet 7:510–523
Wymore AS, Compson ZG, Liu CM, Price LB, Whitham TG, Keim P, Marks JC (2013) Contrasting rRNA gene abundance patterns for aquatic fungi and bacteria in response to leaf litter chemistry. Freshw Sci 32:663–672
Wymore AS, Compson ZG, McDowell WH, Potter JD, Hungate BA, Whitham TG, Marks JC (2015) Leaf litter leachate is distinct in optical properties and bioavailability to stream heterotrophs. Freshwater Sci 34:doi:10.1086/682000.
Ellison AM, Bank MS, Clinton BD, Colburn EA, Elliott K, Ford CR, Foster DR, Kloeppel BD, Knoepp JD, Lovett GM et al (2005) Loss of foundation species: consequences for the structure and dynamics of forested ecosystems. Front Ecol Environ 3:479–486
Schweitzer JA, Bailey JK, Rehill BJ, Martinsen GD, Hart SC, Lindroth RL, Keim P, Whitham TG (2004) Genetically based trait in a dominant tree affects ecosystem processes. Ecol Lett 7:127–134
Fierer N, Bradford MA, Jackson RB (2007) Toward an ecological classification of soil bacteria. Ecology 88:1354–1364
Fierer N, Nemergut D, Knight R, Craine JM (2010) Changes through time: integrating microorganisms into the study of succession. Res Microbiol 161:635–642
Moorhead DL, Sinsabaugh RL (2006) A theoretical model of litter decay and microbial interaction. Ecol Monogr 76:151–174
Findlay S, Tank J, Dye S, Valett HM, Mulholland PJ, McDowell WH, Johnson SL, Hamilton SK, Edmonds J, Dodds WK, Bowden WB (2002) A cross-system comparison of bacterial and fungal biomass in detritus pools of headwater streams. Microb Ecol 43:55–66
Heino J, Tolkkinen M, Pirttilla AM, Aisala H, Mykra H (2014) Microbial diversity and community-environment relationships in boreal streams. J Biogeogr 41:2234–2244
Shade A, Caporaso JG, Handelsman J, Knight R, Fierer N (2013) A meta-analysis of changes in bacterial and archaeal communities with time. ISME J. doi:10.1038/ismej.2013.54
McIntosh ACS, Macdonald SE, Quideau SA (2013) Linkages between the forest floor microbial community and resource heterogeneity within mature lodgepole pine forests. Soil Biol Biochem 63:61–72
Moore JC, Berlow EL, Coleman DC, de Ruiter PC, Dong Q, Hastings A, Johnson NC, McCann KS, Melville K, Morin PJ, Nadelhoffer K, Rosemond AD, Post DM, Sabo JL, Scow KM, Vanni MJ, Wall DH (2004) Detritus, trophic dynamics, and biodiversity. Ecol Lett 7:584–600
Keim P, Paige N, Whitham TG, Lark KG (1989) Genetic analysis of an interspecific hybrid swarm of Populus: occurrence of unidirectional introgression. Genetics 123:557–565
Martinsen GD, Whitham TG, Turek RJ, Keim P (2001) Hybrid populations selectively filter gene introgression between species. Evolution 55:1325–1335
Porter LJ, Hrstich LN, Chan BC (1986) The conversion of procyanidins and prodelphinidins to cyaniding and delphinidin. Pythochemistry 25:223–230
LeRoy CJ, Marks JC (2006) Litter quality, stream characteristics and litter diversity influence decomposition rates and macroinvertebrates. Freshw Biol 51:605–617
Liu CM, Hungate BA, Tobian AAR, Serwadda D, Ravel J, Lester R, Kigozi G, Aziz M, Galiwango RM, Nalugoda F, Contente-Cuomo TL, Wawer MJ, Keim P, Gray RH, Price LB (2013) Male circumcision significantly reduces prevalence and load of genital anaerobic bacterial. mBio 4:doi: 10.1128/mBio.00076-13
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pena AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods. doi:10.1038/nmeth.f.303
Cole JR, Wang Q, Cardenas E, Fish J, Chai B, Farris RJ, Kulum-Syed-Mohideen AS, McGarrell DM, Marsh T, Garrity GM, Tiedje JM (2009) The ribosomal database project: improved alignments and new tools for rRNA analysis. Nucleic Acids Res 37(Supplement 1):D141–D145
Gotelli NJ, Ellison AM (2004) A primer of ecological statistics. Sinauer Associates, Inc, Sunderland, MA. USA
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46
Mantel NA (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
R Development Core Team (2009) R: a language and environment for statistical computing. doi:10.1159/000115429
SPSS. IBM Corporation (2011) IBM SPSS Statistics for Windows, Version 19.0. Armonk, NY.
Besemer K, Singer GA, Limberger R, Chlup A-K, Hochedlinger G, Hödl I-A, Baranyi C, Battin TJ (2007) Biophysical controls on community succession in stream biofilms. Appl Environ Microbiol 73:4966–4974
Jackson CR, Churchill PF, Roden EE (2001) Successional changes in bacterial assemblage structure during epilithic biofilm development. Ecology 82:555–566
Langenheder S, Székely AJ (2011) Species sorting and neutral processes are both important during the initial assembly of bacterial communities. ISME J 5:1086–1094
Lyautey E, Jackson CR, Cayrou J, Rols J-L, Garabétian F (2005) Bacterial community succession in natural river biofilm assemblages. Microb Ecol 50:589–601
Rui JP, Peng JJ, Lu YH (2009) Succession of bacteria populations during plant residue decomposition in rice field soil. Appl Environ Microbiol 75:4879–4886
Padmanabhan P, Padmanabhan S, DeRito C, Gray A, Gannon D, Snape JR, Tsai CS, Park W, Jeon C, Madsen EL (2003) Respiration of 13C-labeled substrates added to soil in the field and subsequent 16S rRNA gene analysis of 13C-labeled soil DNA. Appl Environ Microbiol 69:1614–1622
Fazi S, Amalfitano S, Pernthaler J, Puddu A (2005) Bacterial communities associated with benthic organic matter in headwater stream microhabitats. Environ Microbiol 7:1633–1640
Simon M, Grossart H-P, Schweitzer B, Ploug H (2002) Microbial ecology of organic aggregates in aquatic ecosystems. Aquat Microb Ecol 28:175–211
Stevenson B, Eichorst S, Wertz J, Schmidt T, Breznak J (2004) New strategies for cultivation and detection of previously uncultured microbes. Appl Environ Microbiol 70:4748–4755
Gessner MO, Swan CM, Dang CK, McKie BG, Bardgett RD, Wall DH, Hättenschwiler S (2010) Diversity meets decomposition. Trends Ecol Evol 25:372–380
Rinkes ZL, DeForest JL, Grandy AS, Moorhead DL, Weintraub MN (2014) Interactions between leaf litter quality, particle size, and microbial community composition during the earliest stage of decay. Biogeochemistry 117:153–168
Snajdr J, Cajthaml T, Valaskova V, Merhautova V, Petrankova M, Spetz P, Leppanen K, Baldrian P (2011) Transformation of Quercus petraea litter: successive changes in litter chemistry are reflected in differential enzyme activity and changes in the microbial community composition. FEMS Microbiol Ecol 75:291–303
van Hees PAW, Jones DL, Finlay R, Godbold DL, Lundström US (2005) The carbon we don’t see—the impact of low molecular weight compounds on carbon dynamics and respiration in forest soils: a review. Soil Biol Biochem 37:1–13
Berg B (2000) Litter decomposition and organic matter turnover in northern forest soils. For Ecol Manag 133:13–22
McGuire KL, Fierer N, Bateman C, Treseder KK, Turner BL (2012) Fungal community composition in neotropical rain forests: the influence of tree diversity and precipitation. Environ Microbiol 63:804–812
Nikolcheva LG, Cockshutt AM, Barlocher F (2003) Determining diversity of freshwater fungi on decaying leaves: comparison of traditional and molecular approaches. Appl Environ Microbiol 69:2548–2554
Melillo JM, Aber JD, Muratore JF (1982) Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 63:621–626
Enriquez S, Duarte CM, Sand-Jensen K (1993) Patterns in decomposition rates among photosynthetic organisms: the importance of detritus C:N:P content. Oecologia (Berlin) 94:457–471
Gulis V, Suberkropp K (2003) Leaf litter decomposition and microbial activity in nutrient-enriched and unaltered reaches of a headwater stream. Freshw Biol 48:123–134
Pastor A, Compson ZG, Dijkstra P, Rivera JL, Marti E, Sabater F, Hungate BA, Marks JC (2014) Stream carbon and nitrogen supplements during leaf litter decomposition; contrasting patterns for two foundation species. Oecologia 176:1111–1121
Holeski LM, Hillstrom ML, Whitham TG, Lindroth RL (2012) Relative importance of genetic, ontogenetic, induction, and seasonal variation in producing a multivariate defense phenotype in a foundation tree species. Oecologia 170:695–707
Schweitzer JA, Madritch MD, Bailey JK, LeRoy CJ, Fisher DG, Rehill BJ, Hagerman AE, Wooley SC, Hart SC, Whitham TG (2008) From genes to ecosystems: the genetic basis of condensed tannins and their role in nutrient regulation in Populus model system. Ecosystems 11:1005–1020
Acknowledgments
This research was funded by the National Science Foundation Ecosystems grants (DEB-1120343 and DEB-1119843). ASW was funded by the National Science Foundation IGERT and GK-12 programs. Assistance from J. Buchhagen, Z. Compson, T. Contente, L. Holeski, B. Moan, and S. Owen was appreciated. We greatly appreciate the feedback and constructive criticism from the anonymous referees who reviewed this paper.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 361 kb)
Rights and permissions
About this article
Cite this article
Wymore, A.S., Liu, C.M., Hungate, B.A. et al. The Influence of Time and Plant Species on the Composition of the Decomposing Bacterial Community in a Stream Ecosystem. Microb Ecol 71, 825–834 (2016). https://doi.org/10.1007/s00248-016-0735-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-016-0735-7