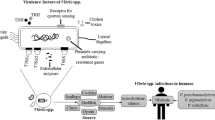
Abstract
The human bacterial pathogen, Vibrio vulnificus, is found in brackish waters and is concentrated by filter-feeding molluscan shellfish, especially oysters, which inhabit those waters. Ingestion of raw or undercooked oysters containing virulent strains of V. vulnificus can result in rapid septicemia and death in 50 % of victims. This review summarizes the current knowledge of the environmental interactions between these two organisms, including the effects of salinity and temperature on colonization, uptake, and depuration rates of various phenotypes and genotypes of the bacterium, and host–microbe immunological interactions.
Similar content being viewed by others
References
Johnston JM, Becker SF, McFarland LM (1985) Gastroenteritis in patients with stool isolates of Vibrio vulnificus. Am J Med 80(2):336–338
Oliver JD (1989) Vibrio vulnificus. In: Doyle MP (ed) Foodborne bacterial pathogens. Marcel Dekker, Inc., New York, pp 569–599
Oliver JD (2006) Vibrio vulnificus. In: Thompson FL, Austin B, Swings J (eds) The biology of vibrios. American Society of Microbiology, Washington, pp 349–366
Jones MK, Oliver JD (2009) Vibrio vulnificus: disease and pathogenesis. Infect Immun 77(5):1723–1733
Oliver J (2005) Wound infections caused by Vibrio vulnificus and other marine bacteria. Epidemiol Infect 133(03):383–391
Mead PS, Slusker L, Dietz V, McCaig LF, Bresee JS, Shapiro C, Griffin PM, Tauxe RBV (1999) Food-related illness and death in the United States. Emerg Infect Dis 5:607–625
Bisharat N, Agmon V, Finkelstein R, Raz R, Ben-Dror G, Lerner L, Soboh S, Colodner R, Cameron DN, Wykstra DL (1999) Clinical, epidemiological, and microbiological features of Vibrio vulnificus biogroup 3 causing outbreaks of wound infection and bacteraemia in Israel. Israel Vibrio Study Group. Lancet 354(9188):1421–1424
Amaro C, Biosca EG (1996) Vibrio vulnificus biotype 2, pathogenic for eels, is also an opportunistic pathogen for humans. Appl Environ Microbiol 62(4):1454
Tison DL, Nishibuchi M, Greenwood JD, Seidler RJ (1982) Vibrio vulnificus biogroup 2: new biogroup pathogenic for eels. Appl Environ Microbiol 44(3):640–646
Warner JM, Oliver JD (1999) Randomly amplified polymorphic DNA analysis of clinical and environmental isolates of Vibrio vulnificus and other Vibrio species. Appl Environ Microbiol 65:526–534
Rosche T, Yano Y, Oliver JD (2005) A rapid and simple PCR analysis indicates there are two subgroups of Vibrio vulnificus which correlate with clinical and environmental isolation. Microbiol Immunol 49(4):381–389
Warner JM, Oliver JD (1999) Randomly amplified polymorphic DNA analysis of clinical and environmental isolates of Vibrio vulnificus and other Vibrio species. Appl Environ Microbiol 65:1141–1144
Nilsson WB, Paranjype RN, DePaola A, Strom MS (2003) Sequence polymorphism of the 16S rRNA gene of Vibrio vulnificus is a possible indicator of strain virulence. J Clin Microbiol 41(1):442–446
Park JH, Cho Y-J, Chun J, Seok Y-J, Lee JK, Kim K-S, Lee K-H, Park S-J, Choi SH (2001) Complete genome sequence of Vibrio vulnificus MO6-24/O. J Bacteriol 193(8):2062–2063
Kim YR, Lee SE, Kim CM, Kim SY, Shin EK, Shin DH, Chung SS, Choy HE, Progulske-Fox A, Hillman JD, Handfield M, Rhee JH (2003) Characterization and pathogenic significance of Vibrio vulnificus antigens preferentially expressed in septicemic patients. Infect Immun 71(10):5461–5471
Froelich BA, Williams TC, Noble RT, Oliver JD (2012) Apparent loss of Vibrio vulnificus from North Carolina oysters coincides with a drought-induced increase in salinity. Appl Environ Microbiol 78(11):3885–3889. doi:10.1128/aem.07855-11
Gulig P, Vd C-L, Wright A, Walts B, Telonis-Scott M, McIntyre L (2010) SOLiD sequencing of four Vibrio vulnificus genomes enables comparative genomic analysis and identification of candidate clade-specific virulence genes. BMC Genomics 11(1):512
Morrison SS, Williams T, Cain A, Froelich B, Taylor C, Baker-Austin C, Verner-Jeffreys D, Hartnell R, Oliver JD, Gibas CJ (2012) Pyrosequencing-based comparative genome analysis of Vibrio vulnificus environmental isolates. PLoS One 7(5):e37553. doi:10.1371/journal.pone.0037553
Stanley JG, Sessers MA (1986) Species profiles: life histories of environmental requirements of coastal fishes and invertebrates (Gulf of Mexico)—American oyster. US Fish Wildl Serv Biol Rep 11.64:25
Shumway S (1996) Natural environmental factors. In: Kennedy VS, Newell RIE, Eble AF (eds) The Eastern oyster Crassostrea virginica. Maryland Sea Grant College, College Park, pp 467–513
Eastern Oyster Biological Review Team (2007) Status review of the Eastern oyster (Crassostrea virginica). Report to the National Marine Fisheries Service, Northeast Regional Office, Gloucester
Oliver JD, Warner RA, Cleland DR (1983) Distribution of Vibrio vulnificus and other lactose-fermenting vibrios in the marine environment. Appl Environ Microbiol 45(3):985–998
Warner EB, Oliver JD (2007) Population structures of two genotypes of Vibrio vulnificus in oysters (Crassostrea virginica) and seawater. Appl Environ Microbiol 74(1):80–85
Han F, Pu S, Hou A, Ge B (2009) Characterization of clinical and environmental types of Vibrio vulnificus isolates from Louisiana oysters. Foodborne Pathog Dis 6(10):1251–1258. doi:10.1089/fpd.2009.0343
Staley C, Jones MK, Wright AC, Harwood VJ (2011) Genetic and quantitative assessment of Vibrio vulnificus populations in oyster (Crassostrea virginica) tissues. Environ Microbiol Rep 3(5):543–549. doi:10.1111/j.1758-2229.2011.00256.x
Warner E, Oliver JD (2008) Population structures of two genotypes of Vibrio vulnificus in oysters (Crassostrea virginica) and seawater. Appl Environ Microbiol 74(1):80–85. doi:10.1128/aem.01434-07
Vickery M, Nilsson W, Strom M, Nordstrom J, DePaola A (2007) A real-time PCR assay for the rapid determination of 16S rRNA genotype in Vibrio vulnificus. J Microbiol Methods 68:376–384
Lin M, Payne DA, Schwarz JR (2003) Intraspecific diversity of Vibrio vulnificus in Galveston Bay water and oysters as determined by randomly amplified polymorphic DNA PCR. Appl Environ Microbiol 69(6):3170–3175
Kirs M, DePaola A, Fyfe R, Jones JL, Krantz J, Van Laanen A, Cotton D, Castle M (2011) A survey of oysters (Crassostrea gigas) in New Zealand for Vibrio parahaemolyticus and Vibrio vulnificus. Int J Food Microbiol 147(2):149–153. doi:10.1016/j.ijfoodmicro.2011.03.012
Froelich B, Ringwood A, Sokolova I, Oliver J (2010) Uptake and depuration of the C- and E-genotypes of Vibrio vulnificus by the Eastern oyster (Crassostrea virginica). Environ Microbiol Rep 2(1):112–115. doi:10.1111/j.1758-2229.2009.00112.x
Hoi L, Larsen JL, Dalsgaard I, Dalsgaard A (1998) Occurrence of Vibrio vulnificus biotypes in Danish marine environments. Appl Environ Microbiol 64(1):7–13
Johnson CN, Flowers AR, Noriea NF, Zimmerman AM, Bowers JC, DePaola A, Grimes DJ (2010) Relationships between environmental factors and pathogenic vibrios in the Northern Gulf of Mexico. Appl Environ Microbiol 76(21):7076–7084. doi:10.1128/AEM.00697-10
Lin M, Schwarz JR (2003) Seasonal shifts in population structure of Vibrio vulnificus in an estuarine environment as revealed by partial 16S ribosomal DNA sequencing. FEMS Microbiol Ecol 45(1):23–27. doi:10.1016/s0168-6496(03)00091-6
Parvathi A, Kumar HS, Karunasagar I, Karunasagar I (2004) Detection and enumeration of Vibrio vulnificus in oysters from two estuaries along the southwest coast of India, using molecular methods. Appl Environ Microbiol 70(11):6909–6913. doi:10.1128/aem.70.11.6909-6913.2004
Tamplin M, Rodrick GE, Blake NJ, Cuba T (1982) Isolation and characterization of Vibrio vulnificus from two Florida estuaries. Appl Environ Microbiol 44(6):1466–1470
Tilton RC, Ryan RW (1987) Clinical and ecological characteristics of Vibrio vulnificus in the Northeastern United States. Diagn Micr Infec Dis 6(2):109–117. doi:10.1016/0732-8893(87)90094-0
Hlady WG (1997) Vibrio infections associated with raw oyster consumption in Florida, 1981–1994. J Food Protect 60:353–357
Shapiro RL, Altekruse S, Hutwagner L, Bishop R, Hammond R, Wilson S, Ray B, Thompson S, Tauxe RV, Griffin PM (1998) The role of gulf coast oysters harvested in warmer months in Vibrio vulnificus infections in the United States, 1988–1996. J Infect Dis 178(3):752–759. doi:10.1086/515367
Fukushima H, Seki R (2004) Ecology of Vibrio vulnificus and Vibrio parahaemolyticus in brackish environments of the Sada River in Shimane Prefecture, Japan. FEMS Microbiol Ecol 48(2):221–229. doi:10.1016/j.femsec.2004.01.009
Kaspar CW, Tamplin ML (1993) Effects of temperature and salinity on the survival of Vibrio vulnificus in seawater and shellfish. Appl Environ Microbiol 59(8):2425–2429
Motes ML, DePaola A, Cook DW, Veazey JE, Hunsucker JC, Garthright WE, Blodgett RJ, Chirtel SJ (1998) Influence of water temperature and Salinity on Vibrio vulnificus in Northern Gulf and Atlantic coast oysters (Crassostrea virginica). Appl Environ Microbiol 64(4):1459–1465
Randa MA, Polz MF, Lim E (2004) Effects of temperature and salinity on Vibrio vulnificus population dynamics as assessed by quantitative PCR. Appl Environ Microbiol 70(9):5469–5476. doi:10.1128/aem.70.9.5469-5476.2004
Vanoy RW, Tamplin ML, Schwarz JR (1992) Ecology of Vibrio vulnificus in Galveston bay oysters, suspended particulate matter, sediment and seawater: detection by monoclonal antibody-immunoassay-most probable number procedures. J Ind Microbiol Biot 9(3):219–223. doi:10.1007/bf01569626
Pfeffer CS, Hite MF, Oliver JD (2003) Ecology of Vibrio vulnificus in estuarine waters of Eastern North Carolina. Appl Environ Microbiol 69(6):3526–3531. doi:10.1128/aem.69.6.3526-3531.2003
O’Neill KR, Jones SH, Grimes DJ (1992) Seasonal incidence of Vibrio vulnificus in the Great Bay estuary of New Hampshire and Maine. Appl Environ Microbiol 58(10):3257–3262
Ristori CA, Iaria ST, Gelli DS, Rivera ING (2007) Pathogenic bacteria associated with oysters (Crassostrea brasiliana) and estuarine water along the south coast of Brazil. Int J Environ Heal R 17(4):259–269. doi:10.1080/09603120701372169
Wright A, Hill R, Johnson J, Roghman M, Colwell R, Morris J Jr (1996) Distribution of Vibrio vulnificus in the Chesapeake Bay. Appl Environ Microbiol 62(2):717–724
Chae M, Cheney D, Su Y (2009) Temperature effects on the depuration of Vibrio parahaemolyticus and Vibrio vulnificus from the American oyster (Crassostrea virginica). J Food Sci 74(2):M62–66
Loosanoff V (1958) Some aspects of behavior of oysters at different temperatures. Biol Bull 114:57–70
Tamplin ML, Capers GM (1992) Persistence of Vibrio vulnificus in tissues of Gulf Coast oysters, Crassostrea virginica, exposed to seawater disinfected with UV light. Appl Environ Microbiol 58(5):1506–1510
Harris-Young L, Tamplin ML, Fisher WS, Mason JW (1993) Effects of physicochemical factors and bacterial colony morphotype on association of Vibrio vulnificus with hemocytes of Crassostrea virginica. Appl Environ Microbiol 59(4):1012–1017
Zimmerman AM, DePaola A, Bowers JC, Krantz JA, Nordstrom JL, Johnson CN, Grimes DJ (2007) Variability of total and pathogenic Vibrio parahaemolyticus densities in northern Gulf of Mexico water and oysters. Appl Environ Microbiol 73(23):7589–7596
Motes M, DePaola A (1996) Offshore suspension relaying to reduce levels of Vibrio vulnificus in oysters (Crassostrea virginica). Appl Environ Microbiol 62(10):3875–3877
Newell RIE, Langdon CJ (1996) Mechanisms and physiology of larval and adult feeding. In: Kennedy VS, Newell RIE, Eble AF (eds) The eastern oyster: Crassostrea virginica. Maryland Sea Grant College, College Park, pp 185–229
Groubert TN, Oliver JD (1994) Interaction of Vibrio vulnificus and the Eastern oyster, Crassostrea virginica. J Food Protect 57(3):224–228
Srivastava M, Tucker MS, Gulig PA, Wright AC (2009) Phase variation, capsular polysaccharide, pilus and flagella contribute to uptake of Vibrio vulnificus by the Eastern oyster (Crassostrea virginica). Environ Microbiol 11(8):1934–1944. doi:10.1111/j.1462-2920.2009.01916.x
Rodrick G, Schneider K, Steslow F, Blake N (1987) Uptake, fate and elimination by shellfish in a laboratory depuration system. In: Oceans '87, Sep. 28, 1987–Oct 1, 1987, pp 1752–1756
Kelly MT, Dinuzzo A (1985) Uptake and clearance of Vibrio vulnificus from Gulf coast oysters (Crassostrea virginica). Appl Environ Microbiol 50(6):1548–1549
Lewis M, Rikard S, Arias C (2010) Evaluation of a flow-through depuration system to eliminate the human pathogen Vibrio vulnificus from oysters. J Aquac Res Development 1(103)
Morris JG, Wright AC, Roberts DM, Wood PK, Simpson LM, Oliver JD (1987) Identification of environmental Vibrio vulnificus isolates with a DNA probe for the cytotoxin-hemolysin gene. Appl Environ Microbiol 53(1):193–195
Murphy SK, Oliver JD (1992) Effects of temperature abuse on survival of Vibrio vulnificus in oysters. Appl Environ Microbiol 58(9):2771–2775
Wright AC, Simpson LM, Oliver JD, Morris JG (1990) Phenotypic evaluation of acapsular transposon mutants of Vibrio vulnificus. Infect Immun 58(6):1769–1773
Oliver JD, Roberts DM, White VK, Dry MA, Simpson LM (1986) Bioluminescence in a strain of the human pathogenic bacterium Vibrio vulnificus. Appl Environ Microbiol 52(5):1209–1211
Oliver JD (1993) Formation of viable nut nonculturable cells. In: Kjelleberg S (ed) Starvation in bacteria. Plenum, New York, pp 239–272
Richards GP (1991) Shellfish depuration. In: Ward DR, Hackney C (eds) Microbiology of marine food products. Van Nostrand Reinhold, New York
Capers GM, Tamplin ML, Martin AL, Hopkins LH (1990) Distribution and retention of Vibrio vulnificus in tissues of the Eastern oyster, Crassostrea virginica. Abstr Annu Meating Am Soc Microbiol 305
Greenberg EP, Dubois M, Palhob B (1982) The survival of marine vibrios in Mercenaria mercenaria, the hardshell clam. J Food Safety 4:113–123
Eyles MJ, Davey GR (1984) Microbiology of commercial depuration of the Sydney rock oyster, Crassostrea commercialis. J Food Protect 47:703–706
Sokolova IM, Leamy L, Harrison M, Oliver JD (2005) Intrapopulational variation in Vibrio vulnificus levels in Crassostrea virginica (GMELIN 1971) is associated with the host size but not with disease status or developmental stability. J Shellfish Res 24(2):503–508. doi:10.2983/0730-8000(2005)24[503:ivivvl]2.0.co;2
Paranjpye RN, Johnson AB, Baxter AE, Strom MS (2007) Role of type IV pilins in persistence of Vibrio vulnificus in Crassostrea virginica oysters. Appl Environ Microbiol 73(15):5041–5044. doi:10.1128/aem.00641-07
Grau BL, Henk MC, Pettis GS (2005) High-frequency phase variation of Vibrio vulnificus 1003: isolation and characterization of rugose phenotypic variant. J Bacteriol 187(7):2519–2525
Baker-Austin C, Lemm E, Hartnell R, Lowther J, Onley R, Amaro C, Oliver JD, Lees D (2012) pilF polymorphism-based real-time PCR to distinguish Vibrio vulnificus strains of human health relevance. Food Microbiol 30(1):17–23. doi:10.1016/j.fm.2011.09.002
Roig FJ, Sanjuan E, Llorens A, Amaro C (2010) pilF Polymorphism-based PCR to distinguish Vibrio vulnificus strains potentially dangerous to public health. Appl Environ Microbiol 76(5):1328–1333. doi:10.1128/AEM.01042-09
Ward JE, Shumway SE (2004) Separating the grain from the chaff: particle selection in suspension- and deposit-feeding bivalves. J Exp Mar Biol Ecol 300:83–130
Murphree R, Tamplin M (1995) Uptake and retention of Vibrio cholerae O1 in the Eastern oyster, Crassostrea virginica. Appl Environ Microbiol 61(10):3656–3660
Sun Y, Oliver JD (1995) Hot sauce: no elimination of Vibrio vulnificus in oysters. J Food Protect 58:441–442
Aldrich H, McDowell L, Tamplin M, Frase C, Murphree R, Jackson J (1995) Detection of Vibrio vulnificus and Vibrio cholerae O1 in oyster tissue using immunoelectron microscopy. J Shellfish Res 14(2):493–499
Fisher WS (1986) Structure and functions of oyster hemocytes. In: Brehelin M (ed) Immunity in invertebrates. Springer, Berlin, pp 25–35
Genthner FJ, Volety AK, Oliver LM, Fisher WS (1999) Factors influencing in vitro killing of bacteria by hemocytes of the Eastern oyster (Crassostrea virginica). Appl Environ Microbiol 65(7):3015–3020
Tamplin ML, Fisher WS (1989) Occurance and characteristics of agglutination of Vibrio cholerae by serum from the eastern oyster, Crassostrea virginica. Appl Environ Microbiol 55:2882–2887
Tyson CJ, Jenkin CR (1974) Phagocytosis of bacteria in vitro by harmocytes from the crayfish (Parachaeraps bicarinatus). Aust J Exp Biol Med Sci 52:341–348
Harris-Young L, Tamplin M, Mason J, Aldrich H, Jackson J (1995) Viability of Vibrio vulnificus in association with hemocytes of the American oyster (Crassostrea virginica). Appl Environ Microbiol 61(1):52–57
Rodrick GE, Ulrich SA (1984) Microscopical studies on the hemocytes of bivalves and their phagocytic interaction with selected bacteria. Helgol Mar Res 37(1):167–176. doi:10.1007/bf01989301
Foley DA, Cheng TC (1975) Degranulation and other changes of molluscan granulocytes associated with phagocytosis. J Invertebr Pathol 25:189–197
McCormick-Ray MG, Howard T (1991) Morphology and mobility of oyster hemocytes: evidence for seasonal variation. J Invertebr Pathol 58:219–230
Oliver JD (1981) Lethal cold stress of Vibrio vulnificus in oysters. Appl Environ Microbiol 41(3):710–717
Pelon W, Luftig RB, Johnston KH (2005) Vibrio vulnificus load reduction in oysters after combined exposure to Vibrio vulnificus-specific bacteriophage and to an oyster extract component. J Food Protect 68(6):1188–1191
Seo JK, Crawford J, Stone K, Noga E (2005) Purification of a novel arthropod defensin from the American oyster, Crassostrea virginica. Biochem Bioph Res Co 338(4):1998–2004
Seo J-K, Stephenson J, Crawford J, Stone K, Noga E (2010) American oyster, Crassostrea virginica, expresses a potent antibacterial histone H2B protein. Mar Biotechnol 12(5):543–551. doi:10.1007/s10126-009-9240-z
Seo J-K, Stephenson J, Noga EJ (2011) Multiple antibacterial histone H2B proteins are expressed in tissues of American oyster. Comp Biochem Physiol B Biochem Mol Biol 158(3):223–229. doi:10.1016/j.cbpb.2010.11.011
Zhang G, Fang X, Guo X, Li L, Luo R, Xu F, Yang P, Zhang L, Wang X, Qi H, Xiong Z, Que H, Xie Y, Holland PWH, Paps J, Zhu Y, Wu F, Chen Y, Wang J, Peng C, Meng J, Yang L, Liu J, Wen B, Zhang N, Huang Z, Zhu Q, Feng Y, Mount A, Hedgecock D, Xu Z, Liu Y, Domazet-Lošo T, Du Y, Sun X, Zhang S, Liu B, Cheng P, Jiang X, Li J, Fan D, Wang W, Fu W, Wang T, Wang B, Zhang J, Peng Z, Li Y, Li N, Wang J, Chen M, He Y, Tan F, Song X, Zheng Q, Huang R, Yang H, Du X, Chen L, Yang M, Gaffney PM, Wang S, Luo L, She Z, Ming Y, Huang W, Zhang S, Huang B, Zhang Y, Qu T, Ni P, Miao G, Wang J, Wang Q, Steinberg CEW, Wang H, Li N, Qian L, Zhang G, Li Y, Yang H, Liu X, Wang J, Yin Y, Wang J (2012) The oyster genome reveals stress adaptation and complexity of shell formation. Nature 490:49–54
Schmitt P, Lorgeril J, Gueguen Y, Destoumieux-Garzón D, Bachère E (2012) Expression, tissue localization and synergy of antimicrobial peptides and proteins in the immune response of the oyster Crassostrea gigas. Dev Comp Immunol 37(3–4):363–370. doi:10.1016/j.dci.2012.01.004
World Health Organization (2005) Risk assessment of Vibrio vulnificus in raw oysters: interpretative summary and technical report. Food and Agriculture Organization, Rome
Ivanina AV, Froelich B, Williams T, Sokolov EP, Oliver JD, Sokolova IM (2011) Interactive effects of cadmium and hypoxia on metabolic responses and bacterial loads of eastern oysters Crassostrea virginica Gmelin. Chemosphere 82(3):377–389. doi:10.1016/j.chemosphere.2010.09.075
Bhattacharya M, Choudhury P, Kumar R (2000) Antibiotic- and metal-resistant strains of Vibrio parahaemolyticus isolated from shrimps. Microb Drug Resist 6:171–172
Fulladosa E, Murat J, Villaescusa I (2005) Effect of cadmium(II), chromium(VI), and arsenic(V) on long-term viability- and growth-inhibition assays using Vibrio fischeri marine bacteria. Arch Environ Contam Toxicol 49:299–306
El-Hendawy HH, Ali DA, Enas H, E-S H, Ghanem SM (2009) Bioaccumulation of heavy metals by Vibrio alginolyticus isolated from wastes of iron and steel factory. Acad J Biol Sci 1:23–28
Kazmi SU, Roberson BS, Stern NJ (1985) Cadmium chloride susceptibility, a characteristic of Campylobacter spp. J Clin Microbiol 21:708–710
Suzuki S, Fukagawa T, Takama K (1992) Occurence of tributyltin-tolerant bacteria in tributyltin- or cadmium-containing seawater. Appl Environ Microbiol 58:3410–3412
Auffret M, Oubella R (1997) Harmocyte aggregation in the oyster Crassostrea gigas: in vitro measurement and experimental modulation by xenobiotics. Comp Biochem Physiol 118A:705–712
Auffret M, Mujdzic N, Corporeau C, Moraga D (2002) Xenobiotic-induced immunomodulation in the European flat oyster, Ostrea edulis. Mar Environ Res 54:585–589
Brousseau P, Pellerin J, Morin Y, Cyr D, Blakely B, Boermans H, Fournier M (2000) Flow cytometry as a tool to monitor the disturbance of phagocytosis in the clam Mya arenaria haemocytes following in vitro exposure to heavy metals. Toxicology 142:145–156
Roesijadi G, Brubacher LL, Unger ME, Anderson RS (1997) Metallotheionein mRNA induction and generation of reactive oxygen species in molluscan haemocytes exposed to cadmium in vitro. Comp Biochem Physiol 118C:171–176
Sauve S, Hendawi M, Brousseau P, Fournier M (2002) Phagocytic response of terrestrial and aquatic invertebrates following in vitro exposure to trace elements. Ecotoxicol Environ Saf 52:21–29
Cherkasov AS, Grewal S, Sokolova IM (2007) Combined effects of temperature and cadmium exposure on haemocyte apoptosis and cadmium accumulation in the eastern oyster Crassostrea virginica (Gmelin). J Therm Biol 32:162–170
Acknowledgements
We would like to thank Craig Baker-Austin for his examination of the data and suggestion of the review. This material was written with the support of the Cooperative State Research, Education, and Extension Service, US Department of Agriculture, under Award No. 2007-35201-18381. Any opinions, findings, conclusions, or recommendations expressed in this publication are those of the authors and do not necessarily reflect the views of the US Department of Agriculture.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Froelich, B., Oliver, J.D. The Interactions of Vibrio vulnificus and the Oyster Crassostrea virginica . Microb Ecol 65, 807–816 (2013). https://doi.org/10.1007/s00248-012-0162-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-012-0162-3