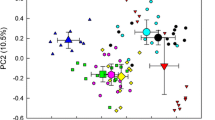
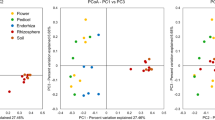
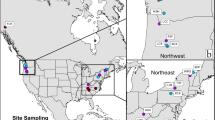
Abstract
Carnivorous pitcher plants host diverse microbial communities. This plant–microbe association provides a unique opportunity to investigate the evolutionary processes that influence the spatial diversity of microbial communities. Using next-generation sequencing of environmental samples, we surveyed microbial communities from 29 pitcher plants (Sarracenia alata) and compare community composition with plant genetic diversity in order to explore the influence of historical processes on the population structure of each lineage. Analyses reveal that there is a core S. alata microbiome, and that it is similar in composition to animal gut microfaunas. The spatial structure of community composition in S. alata (phyllogeography) is congruent at the deepest level with the dominant features of the landscape, including the Mississippi river and the discrete habitat boundaries that the plants occupy. Intriguingly, the microbial community structure reflects the phylogeographic structure of the host plant, suggesting that the phylogenetic structure of bacterial communities and population genetic structure of their host plant are influenced by similar historical processes.


Similar content being viewed by others
References
Whitham TG, Bailey JK, Schweitzer JA, Shuster SM, Bangert RK, LeRoy CJ, Lonsdorf E, Allan GJ, DiFazio SP, Potts BM, Fischer DG, Gehring CA, Lindroth RL, Marks J, Hart SC, Wimp GM, Wooley SC (2006) A framework for community and ecosystem genetics: from genes to ecosystems. Nat Rev Genet 7:510–523
Whitham TG, DiFazio SP, Schweitzer JA, Shuster SM, Allan GJ, Bailey JK, Woolbright SA (2008) Extending genomics to natural communities and ecosystems. Science 320:492–495
Johnson MTJ, Stinchcombe JR (2007) An emerging synthesis between community ecology and evolutionary biology. Trends Ecol Evol 22:250–257
Cavender-Bares J, Kozak KH, Fine PVA, Kembel SW (2009) The merging of community ecology and phylogenetic biology. Ecol Lett 12:693–715
Crutsinger GM, Cadotte MW, Sanders NJ (2009) Plant genetics shapes inquiline community structure across spatial scales. Ecol Lett 12:285–292
Ellison AM, Bank MS, Clinton BD, Colburn EA, Elliot K, Ford CR, Foster DR, Kloeppel BD, Knoepp JD, Lovett GM, Mohan J, Orwig DA, Rodenhouse NL, Sobczak WV, Stinson KA, Stone JK, Swan CM, Thomspon J, Von HOlle B, Webster JR (2005) Loss of foundation species: consequences for the structure and dynamics of forested ecosystems. Front Ecol 3:479–486
Delmotte N, Knief C, Chaffron S, Innerebner G, Roschitzki B, Schlapback R, von Mering C, Vorholt JA (2009) Community proteogenomics reveals insights into the physiology of phyllospher bacteria. Proc Natl Acad Sci USA 106:16428–16433
Schweitzer JA, Bailey JK, Bangert RK, Hart SC, Whitham TG (2007) The role of plant genetic variation in determining above- and belowground microbial communities. In: Bailey MJ, Lilley AK, Timms-Wilson TM, Spencer-Phillips PTN (eds) Microbial Ecology of Arial Plant Surfaces. CABI Publishing, Wallingford pp 109–119
Crutsinger GM, Souza L, Sanders NJ (2008) Intraspecific diversity and dominant genotypes resist plant invasions. Ecol Lett 11:16–23
Lankau RA, Strauss SY (2007) Mutual feebacks maintain both genetic and species diversity in a plant community. Science 317:1561–1563
Johnson MTJ, Agrawal AA (2005) Plant genotype and environment interact to shape a diverse arthropod community on evening primrose (Oenothera biennis). Ecology 86:874–885
Lindow SE, Brandl MT (2003) Microbiology of the phyllosphere. Appl Environ Microbiol 69:1875–1883
Bailey JK, Lilley AK, Timms-Wilson TM, Spencer-Phillips PTN (2006) Microbial ecology of aerial plant surfaces. CABI, Wallingford
Falkowski PG, Fenchel T, Delong EF (2008) The microbial engines that drive Earth’s biogeochemical cycles. Science 320:1034–1039
van der Heijden MGA, Bardgett RD, van Straalen NM (2008) The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol Lett 11:296–310
Jessup CM, Kassen R, Forde SE, Kerr B, Buckling A, Rainey PB, Bohannan BJM (2004) Big questions, small worlds: microbial model systems in ecology. Trends Ecol Evol 19:189–197
Brock TD (1987) The study of microorganism in situ: progress and problems. Symp Soc Gen Microbiol 41:1–17
Achtman M, Wagner M (2008) Microbial diversity and the genetic nature of microbial species. Nat Rev Microbiol 6:431–440
Horner-Devine MC, Carney KM, Bohannan BJM (2004) An ecological perspective on bacterial biodiversity. In: Proceedings of the Royal Society of London Series B-Biological Sciences 271:113–122
Cardenas E, Tiedje JM (2008) New tools for discovering and characterizing microbial diversity. Curr Opin Biotechnol 19:544–549
Mardis E (2008) The impact of next-generation sequencing technology on genetics. Trends Genet 24:133–141
Rothberg JM, Leamon JH (2008) The development and impact of 454 sequencing. Nat Biotechnol 26:1117–1124
Keller M, Hettich R (2009) Environmental Proteomics: a paradigm shift in characterizing microbial activities at the molecular level. Microbiol Mol Biol Rev 73:62–70
Cho JC, Tiedje JM (2000) Biogeography and degree of endemicity of fluorescent Pseudomonas strains in soil. Appl Environ Microbiol 66:5448–5456
Papke RT, Ramsing NB, Bateson MM, Ward DM (2003) Geographical isolation in hot spring cyanobacteria. Environ Microbiol 5:650–659
Hughes Martiny JB, Bohannan BJM, Brown JH, Colwell RK, Fuhrman JA, Green JL, Horner-Devine MC, Kane M, Krumins JA, Kuske CR, Morin PJ, Naeem S, Øvreås L, Reysenbach A, Smith VH, Staley JT (2006) Microbial biogeography: putting microorganisms on the map. Nat Rev Microbiol 4:102–112
Whitaker RJ, Grogan DW, Taylor JW (2003) Geographic barriers isolate endemic populations of hyperthermophilic archaea. Science 301:976–978
Telford RJ, Vandvik V, Birks HJB (2006) Dispersal limitations matter for microbial morphospecies. Science 312:1015
Baas-Becking LGM (1934) In: van Stockum WP, Zoon NV (eds) Geobiologie of Inleiding Tot de Milieukunde. The Hague, The Netherlands
Lomolino MV, Riddle BR, Brown JH (2005) Biogeography. Sinauer, Sunderland
Biek R, Drummond AJ, Poss M (2006) A virus reveals population structure and recent demographic history of its carnivore host. Science 311:538–541
Wirth T, Myer A, Achtman M (2005) Describing host migrations and origins by means of their microbes. Mol Ecol 14:3289–3306
Zhou J, Xia B, Treves DS, Wu LY, Marsh TL, O’Neill RV, Palumbo AV, Tiedje JM (2002) Spatial and resource factors influencing high microbial diversity in soil. Appl Environ Microbiol 68:326–334
Treves DS, Xia B, Zhou J, Tiedje JM (2003) A two-species test of the hypothesis that spatial isolation influences microbial diversity in soil. Microb Ecol 45:20–28
Buckley HL, Miller TE, Ellison AM, Gotelli NJ (2003) Reverse latitudinal trends in species richness of pitcher-plant food webs. Ecol Lett 6:825–829
Peterson CN, Day S, Wolfe BE, Ellison AM, Kolter R, Pringle A (2008) A keystone predator controls bacterial diversity in the pitcher-plant (Sarracenia purpurea) microecosystem. Environ Microbiol 10:2257–2266
Harvey E, Miller TE (1996) Variance in composition of inquiline communities in leaves of Sarracenia purpurea L on multiple spatial scales. Oecologia 108:562–566
Koopman MM, Fuselier D, Hird S, Carstens B (2010) Bacterial characterization of the carnivorous Pale Pitcher Plant reveals diverse, distinct and time-dependent communities. Appl Environ Microbiol 76:1851–1860
Plummer GL, Jackson TH (1963) Bacterial activities within the sarcophagus of the insectivorous plan, Sarracenia flava. Am Midl Nat 69:462–469
Prankevicius AB, Cameron DM (1991) Bacterial dinitrogen fixation in the leaf of the northern piticher plant (Sarracenia purpurea). Can J Bot Rev Canadienne De Botanique 69:2296–2298
Sheridan PM (1991) What is the identity of the West Gulf Coastal pitcher plant, Sarracenia alata? Carnivorous Plant Newsletter 20:102–110
Koopman MM, Carstens BC (2010) Conservation genetic inferences in the carnivorous pitcher plant Sarracenia alata (Sarraceniaceae). Conservation Genetics (in press)
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Huelsenbeck JP, Huelsenbeck ET, Andolfatto P (2007) Structurama: Bayesian inference of population structure. Bioinformatics (in press)
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software structure: a simulation study. Mol Ecol 14:2611–2620
Excoffier L, Smouse P, Quattro J (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 136:343–359
Parnell LD, Rompato G, Latta L, Pfrender M, van Nostrand J, He Z, Zhou J, Anderson G, Champine P, Ganesan B, Weimer B, Aziz R (2010) Functional biogeography as evidence of gene transfer in hypersaline microbial communities. PLoS ONE 5:e12919
Hamady M, Walker J, Harris J, Gold N, Knight R (2008) Error-correcting barcoded primers allow hundreds of samples to be pyrosequenced in multiplex. Nat Meth 5:235–237
Price MN, Dehal PS, Arkin AP (2010) FastTree 2—approximately maximum-likelihood trees for large alignments. PLoS ONE 5:e9490. doi:9410.1371/journal.pone.0009490
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267
Clarke KR, Gorley RN (2006) PRIMER v6: user manual/tutorial. PRIMER-E Ltd., Plymouth Marine Laboratory, Ivybridge, UK
Lozupone C, Knight R (2005) UniFrac: a new phylogenetic method for comparing microbial communities. Appl Environ Microbiol 71:8228–8235
Mantel NA (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Dethlefsen L, McFall-Ngai M, Relman DA (2007) An ecological and evolutionary perspective on human-microbe mutualism and disease. Nature 449:811–818
Felsenstein J (2004) Inferring phylogenies. Sinauer Sunderland, Mass
Foissner W (2006) Biologeography and dispersal of microorganisms: a review emphasizing protists. Acta Protozoologica 45:111–136
Fierer N (2008) Microbial biogeography: patterns in microbial diversity across space and time. ASM Press, Washington
Buckley HL, Burns JH, Kneitel JM, Walters EL, Munguia P, Miller E (2004) Small-scale patterns in community structure of Sarracenia purpurea inquilines. Community Ecol 5:1588–2755
Addicott JF (1974) Predation and prey community structure- experimental study of effect of mosquito larvae on protozoan communities of pitcher plants. Ecology 55:475–492
Kneitel JM, Miller TE (2002) Resource and top-predator regulation in the pitcher plant (Sarracenia purpurea) inquiline community. Ecology 83:680–688
Webb CO, Ackerly DD, McPeek MA, Donoghue MJ (2002) Phylogenies and community ecology. Annu Rev Ecol Syst 33:475–505
Wimp GM, Whitham TG (2001) Biodiversity consequences of predation and host plant hybridization on an aphid-ant mutualism. Ecology 82:440–452
Schweitzer JA, Bailey JK, Fischer DG, LeRoy CJ, Lonsdorf EV, Whitham TG, Hart SC (2008) Heritable relationship between plant genotype and associated soil microorganisms. Ecology 89:773–781
Shuster SM, Lonsdorf E, Wimp GM, Bailey JK, Whitham TG (2007) Community heritability measures the evolutionary consequences of indirect genetic effects on community structure. Evolution 60:991–1003
Srivastava DS, Kolasa J, Bengtsson J, Gonzalez A, Lawler SP, Miller TE, Munguia P, Romanuk T, Schneider DC, Trzcinski MK (2004) Are natural microcosms useful model systems for ecology? Trends Ecol Evol 19:379–384
Ellison AM, Gotelli NJ (2009) Energetics and the evolution of carnivorous plants—Darwin’s ‘most wonderful plants in the world’. J Exp Biol 60:19–42
Acknowledgments
We thank members of the Carstens laboratory, especially Sarah Hird and Daniel Ence for assistance with bioinformatics processing. We thank Brent Christner, Gary King, and Kyle Harms for valuable comments on this project and manuscript. This work has been supported by grants from the LSU Board of Regents Research Competitiveness Grant, the LSU Faculty Research Program, the LSU Pfund program, and the National Science Foundation (DEB 0956069). Sequences are deposited in the NCBI Genbank (accession numbers XXX).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Materials
Below is the link to the electronic supplementary material.
SI Table 1
Distribution of microbial taxonomic and sequence diversity from S. alata across four taxonomic ranks. Eleven phyla, 16 classes, 30 orders, and 76 families are classified using all sequences from all pitchers (sequences assigned to a particular rank that are found in every pitcher are bolded), the number of total sequences at each taxonomic level are listed as well as the abundance distribution between east and western populations. (DOC 139 kb)
SI Fig. 1
(Column A) Distribution of microbial taxonomic diversity from S. alata across four taxonomic ranks. Eleven phyla, 16 classes, 30 orders, and 76 Families. Families are classified using all sequences from all pitchers (names available for supplementary table if deemed necessary). (Column B) Proportion of sequences assigned to a particular rank that are found in every pitcher (black, ubiquitous) vs. not (white, rare). (Column C) Distribution of ubiquitous taxa in S. alata pitchers. Four phyla, four classes, four orders, and three families are represented in every pitcher (DOC 468 kb)
Rights and permissions
About this article
Cite this article
Koopman, M.M., Carstens, B.C. The Microbial Phyllogeography of the Carnivorous Plant Sarracenia alata . Microb Ecol 61, 750–758 (2011). https://doi.org/10.1007/s00248-011-9832-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-011-9832-9