Abstract
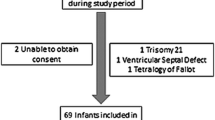
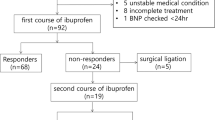
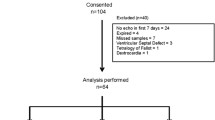
This study investigated whether N-terminal-pro-B-type natriuretic peptide (NT-proBNP) levels could serve as prognostic indicators of the therapeutic responsiveness of the patent ductus arteriosus to pharmacologic treatment. The levels of NT-proBNP in premature neonates with hemodynamically significant patency of the ductus arteriosus (hsPDA) were assessed before and after treatment using ibuprofen, indomethacin, or both. The baseline NT-proBNP levels were similar in both the infants who responded and those who did not respond to medical treatment. The combined data for all the subjects showed that NT-ProBNP decreased after treatment, but the decrease did not correlate significantly with treatment success or failure. Of the 38 infants, 11 did not respond to treatment with ductal closure. Although the pretreatment NT-proBNP levels were similar, the posttreatment levels in the nonresponders remained significantly higher than in the responders. Moreover, in 3 (27%) of the 11 nonresponders, NT-proBNP actually increased rather than decreased with treatment. The NT-proBNP levels of seven infants increased over the course of the study. Within this group, however, the pretreatment NT-proBNP levels were significantly lower than in the overall population, with no differences in the posttreatment levels. Overall, the decrease in NT-proBNP with treatment, presented as the ratio of pretreatment–post-treatment/pretreatment was not well correlated with the ductal therapeutic outcome. In summary, in the study population, NT-proBNP was not sufficiently sensitive for accurate prediction of ductal therapeutic responsiveness.

Similar content being viewed by others
References
Choi M, Lee KH, Eun BL, Yoo KH, Hong YS, Son CS, Lee JW (2005) Utility of rapid B-type natriuretic peptide assay for diagnosis of symptomatic patent ductus arteriosus in preterm infants. Pediatrics 115:e255–e261
Czernik C, Lemmer J, Metze B, Koehne PS, Mueller C, Obladen M (2008) B-type natriuretic peptide to predict ductus intervention in infants < 28 weeks. Pediatr Res 64:286–290
Farombi-Oghuvbu IO, Matthews T, Mayne PD, Guerin H, Corcoran D (2008) N-terminal pro-B-type natriuretic peptide: a measure of significant patent ductus arteriosus. Arch Dis Child Fetal Neonatal Ed 93:F257–F260
Flynn PA, DaGraca RL, Auld P, Nesin M, Kleinman C (2005) The use of a bedside assay for plasma B-type natriuretic peptide as a biomarker in the management of patent ductus arteriosus in premature neonates. J Pediatr 147:38–42
Hammerman C, Shchors I, Jacobson S, Schimmel MS, Bromiker R, Kaplan M, Nir A (2008) Ibuprofen vs. continuous indomethacin in premature neonates with patent ductus arteriosus: is the difference in the mode of administration? Pediatr Res 64:291–297
Holmstrom H, Hall C, Thaulow E (2001) Plasma levels of natriuretic peptides and hemodynamic assessment of patent ductus arteriosus in preterm infants. Acta Paediatr 90:184–191
Puddy VF, Amirmansour C, Williams AF, Singer DRJ (2002) Plasma brain natriuretic peptide as a predictor of haemodynamically significant patent ductus arteriosus in preterm infants. Clin Sci 103:75–77
Sanjeev S, Pettersen M, Lua J, Thomas R, Shankaran S, L’Ecuyer T (2005) Role of plasma B-type natriuretic peptide in screening for hemodynamically significant patent ductus arteriosus in preterm neonates. J Perinatol 25:709–713
Acknowledgment
We thank the NICU nurses for their cooperation. We also express our gratitude to the Hammerman-Fisch Foundation for its generous support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hammerman, C., Shchors, I., Schimmel, M.S. et al. N-terminal-Pro-B-Type Natriuretic Peptide in Premature Patent Ductus Arteriosus: A Physiologic Biomarker, But Is It a Clinical Tool?. Pediatr Cardiol 31, 62–65 (2010). https://doi.org/10.1007/s00246-009-9568-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-009-9568-1