Abstract
Introduction
Magnetic resonance (MR) images from children with optic pathway glioma (OPG) are complex. We initiated this study to evaluate the accuracy of MR imaging (MRI) interpretation and to propose a simple and reproducible imaging classification for MRI.
Methods
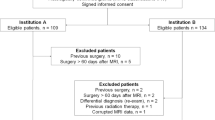
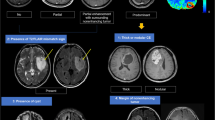
We randomly selected 140 MRIs from among 510 MRIs performed on 104 children diagnosed with OPG in France from 1990 to 2004. These images were reviewed independently by three radiologists (F.T., 15 years of experience in neuroradiology; D.L., 25 years of experience in pediatric radiology; and J.L., 3 years of experience in radiology) using a classification derived from the Dodge and modified Dodge classifications. Intra- and interobserver reliabilities were assessed using the Bland–Altman method and the kappa coefficient. These reviews allowed the definition of reliable criteria for MRI interpretation.
Results
The reviews showed intraobserver variability and large discrepancies among the three radiologists (kappa coefficient varying from 0.11 to 1). These variabilities were too large for the interpretation to be considered reproducible over time or among observers. A consensual analysis, taking into account all observed variabilities, allowed the development of a definitive interpretation protocol. Using this revised protocol, we observed consistent intra- and interobserver results (kappa coefficient varying from 0.56 to 1). The mean interobserver difference for the solid portion of the tumor with contrast enhancement was 0.8 cm3 (limits of agreement = −16 to 17).
Conclusion
We propose simple and precise rules for improving the accuracy and reliability of MRI interpretation for children with OPG. Further studies will be necessary to investigate the possible prognostic value of this approach.






Similar content being viewed by others
Abbreviations
- MRI:
-
Magnetic resonance imaging
- OPG:
-
Optic pathway glioma
- BB-SFOP:
-
Baby Brain–Société Française d’Oncologie Pédiatrique
- DC:
-
Dodge classification
- MDC:
-
Modified Dodge classification
- C+:
-
Solid portion of the tumor with contrast enhancement
- C-:
-
Solid portion of the tumor without contrast enhancement
- RANO:
-
Response Assessment in Neuro-Oncology
References
Binning MJ, Liu JK, Kestle JR, Brockmeyer DL, Walker ML (2007) Optic pathway gliomas: a review. Neurosurg Focus 23(5):E2
Dutton JJ (1994) Gliomas of the anterior visual pathway. Surv Ophtalmol 38(5):427–452
Czyzyk E, Jóźwiak S, Roszkowski M, Schwartz RA (2003) Optic pathway gliomas in children with and without neurofibromatosis 1. J Child Neurol 18(7):471–478
Cappelli C, Grill J, Raquin M, Pierre-Kahn A, Lellouch-Tubiana A, Terrier-Lacombe MJ, Habrand JL, Couanet D, Brauner R, Rodriguez D, Hartmann O, Kalifa C (1998) Long term follow up of 69 patients treated for optic pathway tumours before the chemotherapy era. Arch Dis Child 79(4):334–338
Sievert AJ, Fisher MJ (2009) Pediatric low-grade gliomas. J Child Neurol 24(11):1397–1408
Shaw EG, Wisoff JH (2003) Prospective clinical trials of intracranial low-grade glioma in adults and children. Neuro Oncol 5(3):153–160
Mishra MV, Andrews DW, Glass J, Evans JJ, Dicker AP, Shen X, Lawrence YR (2012) Characterization and outcomes of optic nerve gliomas: a population-based analysis. J Neurooncol 107:591–597
Nicolin G, Parkin P, Mabbott D, Hargrave D, Bartels U, Tabori U, Rutka J, Buncic JR, Bouffet E (2009) Natural history and outcome of optic pathway gliomas in children. Pediatr Blood Cancer 53:1231–1237
Stokland T, Liu JF, Ironside JW, Ellison DW, Taylor R, Robinson KJ, Picton SV, Walker DA (2010) A multivariate analysis of factors determining tumor progression in childhood low-grade glioma: a population-based cohort study (CCLG CNS9702). Neuro-Oncology 12(12):1257–1268
Chateil JF, Soussotte C, Pédespan JM, Brun M, Le Manh C, Diard F (2001) MRI and clinical differences between optic pathway tumours in children with and without neurofibromatosis. Br J Radiol 74(877):24–31
Shuper A, Horev G, Kornreich L, Michowiz S, Weitz R, Zaizov R, Cohen IJ (1997) Visual pathway glioma: an erratic tumour with therapeutic dilemmas. Arch Dis Child 76(3):259–263
Henson JW, Ulmer S, Harris GJ (2008) Brain tumor imaging in clinical trials. AJNR Am J Neuroradiol 29(3):419–424
Dodge HW Jr, Love JG, Craig WM, Dockerty MB, Kearns TP, Holman CB, Hayles AB (1958) Gliomas of the optic nerves. AMA Arch Neurol Psychiatr 79(6):607–621
Taylor T, Jaspan T, Milano G, Gregson R, Parker T, Ritzmann T, Benson C, Walker D, PLAN Study Group (2008) Radiological classification of optic pathway gliomas: experience of a modified functional classification system. Br J Radiol 81(970):761–766
Wen PY, Macdonald DR, Reardon DA, Cloughesy TF, Sorensen AG, Galanis E, Degroot J, Wick W, Gilbert MR, Lassman AB, Tsien C, Mikkelsen T, Wong ET, Chamberlain MC, Stupp R, Lamborn KR, Vogelbaum MA, van den Bent MJ, Chang SM (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28(11):1963–1972
Weizman L, Ben Sira L, Joskowicz L, Constantini S, Precel R, Shofty B, Ben Bashat D (2012) Automatic segmentation, internal classification, and follow-up of optic pathway gliomas in MRI. Med Image Anal 16(1):177–188
Shofty B, Weizman L, Joskowicz L, Constantini S, Kesler A, Ben-Bashat D, Yalon M, Dvir R, Freedman S, Roth J, Ben-Sira L (2011) MRI internal segmentation of optic pathway gliomas: clinical implementation of a novel algorithm. Childs Nerv Syst 27(8):1265–1272
Sorensen AG, Patel S, Harmath C, Bridges S, Synnott J, Sievers A, Yoon YH, Lee EJ, Yang MC, Lewis RF, Harris GJ, Lev M, Schaefer PW, Buchbinder BR, Barest G, Yamada K, Ponzo J, Kwon HY, Gemmete J, Farkas J, Tievsky AL, Ziegler RB, Salhus MR, Weisskoff R (2001) Comparison of diameter and perimeter methods for tumor volume calculation. J Clin Oncol 19(2):551–557
Rakotonjanahary J, De Carli E, Delion M, Kalifa C, Grill J, Doz F, Leblond P, Bertozzi AI, Rialland X, for the Brain Tumor Committee of SFCE (2015) Mortality in children with optic pathway glioma treated with up front BBSFOP chemotherapy. Analysis of a French pediatric historical cohort. PLoS One 10(6):e0127676
Bland JM, Altman DG (1986) Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 8476:307–310
British Standards Institution (1975) Precision of test methods 1: guide for the determination and reproducibility for a standard test method (BS 597, part 1). BSI, London
Walter SD, Eliasziw M, Donner A (1998) Sample size and optimal designs for reliability studies. Stat Med 17:101–110
Sim J, Wright CC (2005) The kappa statistic in reliability studies: use, interpretation, and sample size requirements. Phys Ther 85:257–268
Vos MJ, Uitdehaag BM, Barkhof F, Heimans JJ, Baayen HC, Boogerd W, Castelijns JA, Elkhuizen PH, Postma TJ (2003) Interobserver variability in the radiological assessment of response to chemotherapy in glioma. Neurology 60(5):826–830
Hsu TR, Wong TT, Chang FC, Ho DM, Tang RB, Thien PF, Chang KP (2008) Responsiveness of progressive optic pathway tumors to cisplatin-based chemotherapy in children. Childs Nerv Syst 24(12):1457–1461
Tao ML, Barnes PD, Billett AL, Leong T, Shrieve DC, Scott RM, Tarbell NJ (1997) Childhood optic chiasm gliomas: radiographic response following radiotherapy and long-term clinical outcome. Int J Radiat Oncol Biol Phys 39(3):579–587
Laithier V, Grill J, Le Deley MC, Ruchoux MM, Couanet D, Doz F, Pichon F, Rubie H, Frappaz D, Vannier JP, Babin-Boilletot A, Sariban E, Chastagner P, Zerah M, Raquin MA, Hartmann O, Kalifa C, French Society of Pediatric Oncology (2003) Progression-free survival in children with optic pathway tumors: dependence on age and the quality of the response to chemotherapy. Results of the first French prospective study for the French Society of Pediatric Oncology. J Clin Oncol 21(24):4572–4578
Liu J, Udupa JK, Odhner D, Hackney D, Moonis G (2005) A system for brain tumor volume estimation via MR imaging and fuzzy connectedness. Comput Med Imaging Graph 29(1):21–34
Prastawa M, Bullitt E, Moon N, Van Leemput K, Gerig G (2003) Automatic brain tumor segmentation by subject specific modification of atlas priors. Acad Radiol 10(12):1341–1348
Mehta AI, Kanaly CW, Friedman AH, Bigner DD, Sampson JH (2011) Monitoring radiographic brain tumor progression. Toxins 3(3):191–200
Clarke LP, Velthuizen RP, Clark M, Gaviria J, Hall L, Goldgof D, Murtagh R, Phuphanich S, Brem S (1998) MRI measurement of brain tumor response: comparison of visual metric and automatic segmentation. Magn Reson Imaging 16(3):271–279
Acknowledgments
We are grateful to the following investigators of Société Française des Cancers de l’Enfant (SFCE) for providing the patients’ MRIs: J Grill, Gustave-Roussy Institute Villejuif; C Dufour, Gustave-Roussy Institute Villejuif; F Doz, Curie Institute and University Paris Descartes Paris; P Leblond, Oscar Lambret Center Lille; A Bertozzi, University Hospital Toulouse; D Frappaz, Pediatric Hematology-Oncology Institute Lyon; V Laithier, University Hospital Besançon; D Plantaz, University Hospital Grenoble; Y Perel, University Hospital Bordeaux; P Chastagner, University Hospital Nancy; C Chappé, University Hospital Rennes; N Sirvent, University Hospital Montpellier; JC Gentet, University Hospital La Timone Marseille; P Schneider, University Hospital Rouen; C Berger, University Hospital St. Etienne; P Lutz, University Hospital Strasbourg; P Blouin, University Hospital Tours; C Piguet, University Hospital Limoges; P Lemoine, University Hospital Brest; F Millot, University Hospital Poitiers; O Minckes, University Hospital Caen; and F Demeocq, University Hospital Clermont-Ferrand. We are grateful to E Berardi and S Chalal, University Hospital Angers, for collecting the data. We are also grateful to C Alberti, INSERM CIE5, Robert Debré Hospital, Paris, for reviewing the study’s design.
This work was supported by Ligue Nationale Contre le Cancer and the Pfizer Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
We declare that all human studies have been approved by the Comité de Protection des Personnes - Ouest 3, Poitiers, and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. We declare that all patients gave informed consent prior to inclusion in this study.
Conflict of interest
We declare that we have no conflict of interest.
Additional information
JL and JR contributed equally to this work.
Rights and permissions
About this article
Cite this article
Lambron, J., Rakotonjanahary, J., Loisel, D. et al. Can we improve accuracy and reliability of MRI interpretation in children with optic pathway glioma? Proposal for a reproducible imaging classification. Neuroradiology 58, 197–208 (2016). https://doi.org/10.1007/s00234-015-1612-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-015-1612-7