Abstract
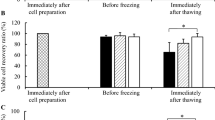
New cryopreservation approaches for medically applicable cells are of great importance in clinical medicine. Current protocols employ the use of dimethyl sulfoxide (DMSO), which is toxic to cells and causes undesirable side effects in patients, such as cardiac arrhythmias, neurological events, and others. Trehalose, a nontoxic disaccharide, has been already studied as a cryoprotectant. However, an efficient approach for loading this impermeable sugar into mammalian cells is missing. In our study, we assessed the efficiency of combining reversible electroporation and trehalose for cryopreservation of human adipose-derived stem cells. First, we determined reversible electroporation threshold by loading of propidium iodide into cells. The highest permeabilization while maintaining high cell viability was reached at 1.5 kV/cm, at 8 pulses, 100 µs, and 1 Hz. Second, cells were incubated in 250 or 400 mM trehalose and electroporated before cryopreservation. After thawing, 83.8 ± 1.8 % (mean ± SE) cell recovery was obtained at 250 mM trehalose. By using a standard freezing protocol (10 % DMSO in 90 % fetal bovine serum), cell survival after thawing was about 91.5 ± 1.6 %. We also evaluated possible effects of electroporation on cells’ functionality before and after thawing. Successful cell growth and efficient adipogenic and osteogenic differentiation were achieved. In conclusion, electroporation seems to be an efficient method for loading nonpermeable trehalose into human adipose-derived stem cells, allowing long-term cryopreservation in DMSO-free and xeno-free conditions.





Similar content being viewed by others
References
Beattie GM, Crowe JH, Lopez AD, Crulli V, Ricordi C, Hayek A (1997) Trehalose: a cryoprotectant that enhances recovery and preserves function of human pancreatic islets after long-term storage. Diabetes 46:519–523
Brockbank KGM, Campbell LH, Greene ED, Brockbank MCG, Duman JG (2010) Lessons from nature for preservation of mammalian cells, tissues, and organs. Vitro Cell Dev Biol Anim 47(3):210–217
Campbell LH, Brockbank KG (2012) Culturing with trehalose produces viable endothelial cells after cryopreservation. Cryobiology 64(3):240–244
Cox MA, Kastrup J, Hrubiško M (2012) Historical perspectives and the future of adverse reactions associated with haemopoietic stem cells cryopreserved with dimethyl sulfoxide. Cell Tissue Bank 13(2):203–215
Crowe JH, Crowe LM (2000) Preservation of mammalian cells-learning nature’s tricks. Nat Biotechnol 18(2):145–146
Crowe JH, Crowe LM, Carpenter JF, Rudolph AS, Wistrom CA, Spargo BJ, Anchordoguy TJ (1988) Interactions of sugars with membranes. Biochim Biophys Acta 947(2):367–384
Crowe JH, Crowe LM, Hoekstra FA (1989) Phase transitions and permeability changes in dry membranes during rehydration. J Bioenerg Biomembr 1:77–91
Delteil C, Teissié J, Rols MP (2000) Effect of serum on in vitro electrically mediated gene delivery and expression in mammalian cells. Biochim Biophys Acta 1467(2):362–368
Dovgan B, Dermol J, Barlič A, Knežević M, Miklavčič D (2015) Cryopreservation of Human Umbilical Stem Cells in Combination with Trehalose and Reversible Electroporation. 1st World Congress on Electroporation and Pulsed Electric Fields in Biology, Medicine and Food & Environmental Technologies. IFMBE Proc 53:307–310
Dymek K, Dejmek P, Galindo FG (2014) Influence of Pulsed Electric Field Protocols on the Reversible Permeabilization of Rucola Leaves. Food Bioprocess Tech 7:761–773
Eroglu A, Russo MJ, Bieganski R, Fowler A, Cheley S, Bayley H, Toner M (2000) Intracellular trehalose improves the survival of cryopreserved mammalian cells. Nat Biotechnol 18(2):163–167
Fang B, Song Y, Lin Q, Zhang Y, Cao Y, Zhao RC, Ma Y (2007) Human adipose tissue-derived mesenchymal stromal cells as salvage therapy for treatment of severe refractory acute graft-vs.-host disease in two children. Pediatr Transpl 11(7):814–817
Golberg A, Sack M, Teissie J, Pataro G, Pliquett U, Saulis G, Töpfl S, Miklavčič D, Vorobiev E, Frey W (2016) Energy-efficient biomass processing with pulsed electric fields for bioeconomy and sustainable development. Biotechnol Biofuels 9:94
Grigg A, Hoyt R, Szer J (2000) Neurological events associated with the infusion of cryopreserved bone marrow and/or periph-eral blood progenitor cells. Bone Marrow Transpl 25:1285–1287
Guo N, Puhlev I, Brown DR, Mansbridge J, Levine F (2000) Trehalose expression confers desiccation tolerance on human cells. Nat Biotechnol 18(2):168–171
Gurtovenko AA, Anwar J (2007) Modulating the Structure and Properties of Cell Membranes: the Molecular Mechanism of Action of Dimethyl Sulfoxide. J Phys Chem B 111:10453–10460
Haberl S, Miklavčič D, Serša G, Frey W, Rubinsky B (2013) Cell Membrane Electroporation—Part 2: the Applications. IEEE Electr Insul M 29(1):29–37
Higman MA, Port JD, Beauchamp NJ, Chen AR Jr (2000) Re-versible leukoencephalopathy associated with re-infusion of DMSO preserved stem cells. Bone Marrow Transpl 26:797–800
Jakštys B, Ruzgys P, Tamošiūnas M, Šatkauskas S (2015) Different Cell Viability Assays Reveal Inconsistent Results After Bleomycin Electrotransfer In Vitro. J Membr Biol 248(5):857–863
Kanias T, Acker JP (2006) Mammalian cell desiccation: facing the challenges. Cell Presev Technol 4(4):253–277
Karlsson OMJ, Toner M (1996) Long-term storage of tissues by cryopreservation: critical issues. Biomaterials 17(3):243–256
Katkov II, Kim MS, Bajpai R, Altman YS, Mercola M, Loring JF, Terskikh AV, Snyder EY, Levine F (2006) Cryopreservation by slow cooling with DMSO diminished production of Oct-4 pluripotency marker in human embryonic stem cells. Cryobiology 53(2):194–205
Konov KB, Leonov DV, Isaev NP, Fedotov KY, Voronkova VK, Dzuba SA (2015) Membrane-Sugar Interactions Probed by Pulsed Electron Paramagnetic Resonance of Spin Labels. J Phys Chem B 119(32):10261–10266
Kotnik T, Kramar P, Pucihar G, Miklavčič D, Tarek M (2012) Cell Membrane Electroporation—Part 1: the Phenomenon. IEEE Electr Insul M 28(5):14–23
Larson DJ, Middle L, Vu H, Zhang W, Serianni AS, Duman J, Barnes BM (2014) Wood frog adaptations to overwintering in Alaska: new limits to freezing tolerance. J Exp Biol 217(Pt 12):2193–2200
Lendeckel S, Jödicke A, Christophis P, Heidinger K, Wolff J, Fraser JK, Hedrick MH, Berthold L, Howaldt HP (2004) Autologous stem cells (adipose) and fibrin glue used to treat widespread traumatic calvarial defects: case report. J Craniomaxillofac Surg 32(6):370–373
Mahnič-Kalamiza S, Vorobiev E, Miklavčič D (2014) Electroporation in food processing and biorefinery. J Membr Biol 247(12):1279–1304
Mazur P (1984) Freezing of living cells: mechanisms and implications. Am J Physiol 247(3 Pt 1):C125–C142
Miklavčič D, Semrov D, Mekid H, Mir LM (2000) A validated model of in vivo electric field distribution in tissues for electrochemotherapy and for DNA electrotransfer for gene therapy. Biochim Biophys Acta 1523(1):73–83
Miklavčič D, Mali B, Kos B, Heller R, Serša G (2014) Electrochemotherapy: from the drawing board into medical practice. Biomed Eng Online 13(1):29
Mlakar V, Todorovic V, Cemazar M, Glavac D, Sersa G (2009) Electric pulses used in electrochemotherapy and electrogene therapy do not significantly change the expression profile of genes involved in the development of cancer in malignant melanoma cells. BMC Cancer 9:299
Mohr JC, de Pablo JJ (2006) Palecek SP (2006) Electroporation of Human Embryonic Stem Cells: small and macromolecule Loading and DNA Transfection. Biotechnol Prog 22:825–834
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65(1–2):55–63
Neumann E, Rosenheck K (1972) Permeability changes induced by electric impulses in vesicular membranes. J Membr Biol 10(3):279–290
Oliver AE, Jamil K, Crowe JH, Tablin F (2004) Loading Human Mesenchymal Stem Cells with Trehalose by Fluid-Phase Endocytosis. Cell Preserv Technol 2(1):35–49
Petrenko YA, Rogulska OY, Mutsenko VV, Petrenko AY (2014) A sugar pretreatment as a new approach to the Me2SO- and xeno-free cryopreservation of human mesenchymal stromal cells. Cryo Letters 35(3):239–246
Phoon PY, Galindo FG, Vicente A, Dejmek P (2008) Pulsed electric field in combination with vacuum impregnation with trehalose improves the freezing tolerance of spinach leaves. J Food Eng 88(1):144–148
Polge C, Smith AU, Parkes AS (1949) Revival of spermatozoa after vitrification and dehydration at low temperatures. Nature 164(4172):666
Puissant B, Barreau C, Bourin P, Clavel C, Corre J, Bousquet C, Taureau C, Cousin B, Abbal M, Laharrague P, Penicaud L, Casteilla L, Blancher A (2005) Immunomodulatory effect of human adipose tissue-derived adult stem cells: comparison with bone marrow mesenchymal stem cells. Br J Haematol 129(1):118–129
Rada T, Reis RL, Gomes ME (2009) Adipose tissue-derived stem cells and their application in bone and cartilage tissue engineering. Tissue Eng 15:113–125
Rems L, Miklavčič D (2016) Tutorial: electroporation of cells in complex materials and tissue. J Appl Phys 119:201101
Ryden M, Dicker A, Gotherstrom C, Aström G, Tammik C, Arner P, Le Blanc K (2003) Functional characterization of human mesenchymal stem cell–derived adipocytes. Biochem Biophys Res Commun 311:391–397
Sharp DMC, Picken A, Morris TJ, Hewitt CJ, Coopman K, Slater NKH (2013) Amphipathic polymer-mediated uptake of trehalose for dimethyl sulfoxide-free human cell cryopreservation. Cryobiology 67:305–311
Shayanfan S, Chauhan O, Toepfl S, Heinz V (2013) The interaction of pulsed electric fields and texturizing - antifreezing agents in quality retention of defrosted potato strips. Int J Food Sci Tech 48:1289–1295
Shirakashi R, Köstner CM, Müller KJ, Kürschner M, Zimmernann U, Sukhorukov VL (2002) Intracellular delivery of trehalose into mammalian cells by electropermeabilization. J Membrane Biol 189:45–54
Spiller DGl, Giles RV, Grzybowski J, Tidd DM, Clark RE (1998) Improving the intracellular delivery and molecular efficacy of antisense oligonucleotides in chronic myeloid leukemia cells: a comparison of streptolysin-O permeabilization, electroporation, and lipophilic conjugation. Blood 91(12):4738–4746
Syed BA, Evans JB (2013) Stem cell therapy market. Nat Rev Drug Discov 12(3):185–186
Syme R, Bewick M, Stewart D, Porter K, Chadderton T, Glück S (2004) The role of depletion of dimethyl sulfoxide before autografting: on hematologic recovery, side effects, and toxicity. Biol Blood Marrow Transplant 10(2):135–141
Todorovic V, Sersa G, Mlakar V, Glavac D, Flisar K, Cemazar M (2011) Metastatic potential of melanoma cells is not affected by electrochemotherapy. Melanoma Res 21(3):196–205
Tsujimoto M, Imura S, Kanda H (2016) Recovery and reproduction of an Antarctic tardigrade retrieved from a moss sample frozen for over 30 years. Cryobiology 72(1):78–81
Wagner W, Wein F, Seckinger A, Frankhauser M, Wirkner U, Krause U, Blake J, Schwager C, Eckstein V, Ansorge W, Ho AD (2005) Comparative characteristics of mesenchymal stem cells from human bone marrow, adipose tissue, and umbilical cord blood. Exp Hematol 33:1402–1416
Wang HY, Lun ZR, Lu SS (2011) Cryopreservation of umbilical cord blood-derived mesenchymal stem cells without dimethyl sulfoxide. Cryo Letters 32(1):81–88
Wolkers WF, Walker NJ, Tablin F, Crowe JH (2001) Human platelets loaded with trehalose survive freeze-drying. Cryobiology 42(2):79–87
Yarmush ML, Golberg A, Serša G, Kotnik T, Miklavčič D (2014) Electroporation-based technologies for medicine: principles, applications, and challenges. Annu Rev Biomed Eng 16:295–320
Zenhausern R, Tobler A, Leoncini L, Hess OM, Ferrari P (2000) Fatal cardiac arrhythmia after infusion of dimethyl sulfoxide-cryopreserved hematopoietic stem cells in a patient with severe primary cardiac amyloidosis and end-stage renal failure. Ann Hematol 79:523–526
Zhang M, Oldenhof H, Sieme H (2016) Wolkers WF (2016) Freezing-induced uptake of trehalose into mammalian cells facilitates cryopreservation. Biochim Biophys Acta 1858(6):1400–1409
Zhou X, Yuan J, Liu J, Liu B (2010) Loading trehalose into red blood cells by electroporation and its application in freeze-drying. Cryo Letters 31(2):147–156
Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH (2001) Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 7:211–228
Acknowledgments
The authors would like to thank Prof. Dr. Marija Bešter Rogač and Anton Kelbl from the Faculty of Chemistry and Chemical Technology, the University of Ljubljana and Janja Dermol from Faculty of Electrical Engineering, and the University of Ljubljana for their help with the osmolality measurements. Research was supported with Slovenian Research Agency through Research Programme, Electroporation-Based Technologies and Treatments (P2-0249), and Infrastructure Programme, Network of research infrastructure centres at University of Ljubljana (IP-0510). The work was performed in the scope of LEA EBAM.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Dovgan, B., Barlič, A., Knežević, M. et al. Cryopreservation of Human Adipose-Derived Stem Cells in Combination with Trehalose and Reversible Electroporation. J Membrane Biol 250, 1–9 (2017). https://doi.org/10.1007/s00232-016-9916-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00232-016-9916-z