Abstract
Purpose
We examined the effect of a single apple juice intake on the pharmacokinetics of fexofenadine enantiomers in healthy Japanese subjects.
Methods
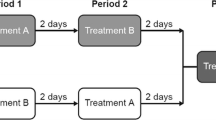
In a randomized two phase, open-label crossover study, 14 subjects received 60 mg of racemic fexofenadine simultaneously with water or apple juice. For the uptake studies, oocytes expressing organic anion-transporting polypeptide 2B1 (OATP2B1) were incubated with 100 μM (R)- and (S)-fexofenadine in the presence or absence of 10 % apple juice.
Results
One-time ingestion of apple juice significantly decreased the area under the plasma concentration–time curve (AUC0–24) for (R)- and (S)-fexofenadine by 49 and 59 %, respectively, and prolonged the time to reach the maximum plasma concentration (t max) of both enantiomers (P < 0.001). Although apple juice greatly reduced the amount of (R)- and (S)-fexofenadine excretion into urine (Ae0–24) by 54 and 58 %, respectively, the renal clearances of both enantiomers were unchanged between the control and apple juice phases. For in vitro uptake studies, the uptake of both fexofenadine enantiomers into OATP2B1 complementary RNA (cRNA)-injected oocytes was significantly higher than that into water-injected oocytes, and this effect was greater for (R)-fexofenadine. In addition, apple juice significantly decreased the uptake of both enantiomers into OATP2B1 cRNA-injected oocytes.
Conclusions
These results suggest that OATP2B1 plays an important role in the stereoselective pharmacokinetics of fexofenadine and that one-time apple juice ingestion probably inhibits intestinal OATP2B1-mediated transport of both enantiomers. In addition, this study demonstrates that the OATP2B1 inhibition effect does not require repeated ingestion or a large volume of apple juice.



Similar content being viewed by others
References
Giacomini KM, Huang SM, Tweedie DJ et al (2010) Membrane transporters in drug development. Nat Rev Drug Discov 9:215–236
Kalliokoski A, Niemi M (2009) Impact of OATP transporters on pharmacokinetics. Br J Pharmacol 158:693–705
Xiong H, Carr RA, Locke CS, Katz DA, Achari R, Doan TT, Wang P, Jankowski JR, Sleep DJ (2007) Dual effects of rifampin on the pharmacokinetics of atrasentan. J Clin Pharmacol 47:423–429
van Giersbergen PL, Treiber A, Schneiter R, Dietrich H, Dingemanse J (2007) Inhibitory and inductive effects of rifampin on the pharmacokinetics of bosentan in healthy subjects. Clin Pharmacol Ther 81:414–419
Zheng HX, Huang Y, Frassetto LA, Benet LZ (2009) Elucidating rifampin’s inducing and inhibiting effects on glyburide pharmacokinetics and blood glucose in healthy volunteers: unmasking the differential effects of enzyme induction and transporter inhibition for a drug and its primary metabolite. Clin Pharmacol Ther 85:78–85
Bidstrup TB, Stilling N, Damkier P, Scharling B, Thomsen MS, Brosen K (2004) Rifampicin seems to act as both an inducer and an inhibitor of the metabolism of repaglinide. Eur J Clin Pharmacol 60:109–114
Shitara Y (2011) Clinical importance of OATP1B1 and OATP1B3 in drug-drug interactions. Drug Metab Pharmacokinet 26:220–227
Kusuhara H, Miura M, Yasui-Furukori N, Yoshida K, Akamine Y, Yokochi M, Fukizawa S, Ikejiri K, Kanamitsu K, Uno T, Sugiyama Y (2013) Effect of coadministration of single and multiple doses of rifampicin on the pharmacokinetics of fexofenadine enantiomers in healthy subjects. Drug Metab Dispos 41:206–213
Bailey DG (2010) Fruit juice inhibition of uptake transport: a new type of food-drug interaction. Br J Clin Pharmacol 70:645–655
Tapaninen T, Neuvonen PJ, Niemi M (2011) Orange and apple juice greatly reduce the plasma concentrations of the OATP2B1 substrate aliskiren. Br J Clin Pharmacol 71:718–726
Neuhofel AL, Wilton JH, Victory JM, Hejmanowsk LG, Amsden GW (2002) Lack of bioequivalence of ciprofloxacin when administered with calcium-fortified orange juice: a new twist on an old interaction. J Clin Pharmacol 42:461–466
Lilja JJ, Backman JT, Laitila J, Luurila H, Neuvonen PJ (2003) Itraconazole increases but grapefruit juice greatly decreases plasma concentrations of celiprolol. Clin Pharmacol Ther 73:192–198
Lilja JJ, Juntti-Patinen L, Neuvonen PJ (2004) Orange juice substantially reduces the bioavailability of the beta-adrenergic-blocking agent celiprolol. Clin Pharmacol Ther 75:184–190
Lilja JJ, Raaska K, Neuvonen PJ (2005) Effects of orange juice on the pharmacokinetics of atenolol. Eur J Clin Pharmacol 61:337–340
Schwarz UI, Seemann D, Oertel R, Miehlke S, Kuhlisch E, Fromm MF, Kim RB, Bailey DG, Kirch W (2005) Grapefruit juice ingestion significantly reduces talinolol bioavailability. Clin Pharmacol Ther 77:291–301
Dresser GK, Bailey DG, Leake BF, Schwarz UI, Dawson PA, Freeman DJ, Kim RB (2002) Fruit juices inhibit organic anion transporting polypeptide-mediated drug uptake to decrease the oral availability of fexofenadine. Clin Pharmacol Ther 71:11–20
Satoh H, Yamashita F, Tsujimoto M, Murakami H, Koyabu N, Ohtani H, Sawada Y (2005) Citrus juices inhibit the function of human organic anion-transporting polypeptide OATP-B. Drug Metab Dispos 33:518–523
Rodriguez-Fragoso L, Martinez-Arismendi JL, Orozco-Bustos D, Reyes-Esparza J, Torres E, Burchiel SW (2011) Potential risks resulting from fruit/vegetable-drug interactions: effects on drug-metabolizing enzymes and drug transporters. J Food Sci 76:R112–R124
Bailey DG, Dresser GK, Leake BF, Kim RB (2007) Naringin is a major and selective clinicalinhibitor of organic anion-transporting polypeptide 1A2 (OATP1A2) in grapefruitjuice. Clin Pharmacol Ther 81:495–502
Imanaga J, Kotegawa T, Imai H, Tsutsumi K, Yoshizato T, Ohyama T, Shirasaka Y, Tamai I, Tateishi T, Ohashi K (2011) The effects of the SLCO2B1 c.1457C > T polymorphism and apple juice on the pharmacokinetics of fexofenadine and midazolam in humans. Pharmacogenet Genomics 21:84–93
Shirasaka Y, Shichiri M, Mori T, Nakanishi T, Tamai I (2013) Major active components in grapefruit, orange, and apple juices responsible for OATP2B1-mediated drug interactions. J Pharm Sci 102:280–288
Shirasaka Y, Shichiri M, Murata Y, Mori T, Nakanishi T, Tamai I (2013) Long-lasting inhibitory effect of apple and orange juices, but not grapefruit juice, on OATP2B1-mediated drug absorption. Drug Metab Dispos 41:615–621
Robbins DK, Castles MA, Pack DJ, Bhargava VO, Weir SJ (1998) Dose proportionality and comparison of single and multiple dose pharmacokinetics of fexofenadine (MDL 16455) and its enantiomers in healthy male volunteers. Biopharm Drug Dispos 19:455–463
Tateishi T, Miura M, Suzuki T, Uno T (2008) The different effects of itraconazole on the pharmacokinetics of fexofenadine enantiomers. Br J Clin Pharmacol 65:693–700
Sakugawa T, Miura M, Hokama N, Suzuki T, Tateishi T, Uno T (2009) Enantioselective disposition of fexofenadine with the P-glycoprotein inhibitor verapamil. Br J Clin Pharmacol 67:535–540
Akamine Y, Miura M, Yasui-Furukori N, Kojima M, Uno T (2012) Carbamazepine differentially affects the pharmacokinetics of fexofenadine enantiomers. Br J Clin Pharmacol 73:478–481
Cvetkovic M, Leake B, Fromm MF, Wilkinson GR, Kim RB (1999) OATP and P-glycoprotein transporters mediate the cellular uptake and excretion of fexofenadine. Drug Metab Dispos 27:866–871
Matsushima S, Maeda K, Inoue K, Ohta KY, Yuasa H, Kondo T, Nakayama H, Horita S, Kusuhara H, Sugiyama Y (2009) The inhibition of human multidrug and toxin extrusion 1 is involved in the drug-drug interaction caused by cimetidine. Drug Metab Dispos 37:555–559
Akamine Y, Miura M, Sunagawa S, Kagaya H, Yasui-Furukori N, Uno T (2010) Influence of drug-transporter polymorphisms on the pharmacokinetics of fexofenadine enantiomers. Xenobiotica 40:782–789
Miura M, Uno T, Tateishi T, Suzuki T (2007) Determination of fexofenadine enantiomers in human plasma with high-performance liquid chromatography. J Pharm Biomed Anal 43:741–745
Shirasaka Y, Li Y, Shibue Y, Kuraoka E, Spahn-Langguth H, Kato Y, Langguth P, Tamai I (2009) Concentration-dependent effect of naringin on intestinal absorption of beta(1)-adrenoceptor antagonist talinolol mediated by p-glycoprotein and organic anion transporting polypeptide (OATP). Pharm Res 26:560–567
Center for Drug Evaluation and Research (2003) Guidance for industry: bioavailability and bioequivalence studies for orally administered drug products: general considerations. U.S. Food and Drug Administration, Center for Drug Evaluation and Research, Rockville, MD
Product information (2003) Allegra®; Sanofi-Aventis K.K., Tokyo, Japan
Glaeser H, Bailey DG, Dresser GK, Gregor JC, Schwarz UI, McGrath JS, Jolicoeur E, Lee W, Leake BF, Tirona RG, Kim RB (2007) Intestinal drug transporter expression and the impact of grapefruit juice in humans. Clin Pharmacol Ther 81:362–370
Rebello S, Zhao S, Hariry S, Dahlke M, Alexander N, Vapurcuyan A, Hanna I, Jarugula V (2012) Intestinal OATP1A2 inhibition as a potential mechanism for the effect of grapefruit juice on aliskiren pharmacokinetics in healthy subjects. Eur J Clin Pharmacol 68:697–708
Pauli-Magnus C, von Richter O, Burk O, Ziegler A, Mettang T, Eichelbaum M, Fromm MF (2000) Characterization of the major metabolites of verapamil as substrates and inhibitors of P-glycoprotein. J Pharmacol Exp Ther 293:376–382
Conflict of interest
The authors have no conflicts of interest in relation to this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Tsukasa Uno is a former member of the University of the Ryukyus.
Rights and permissions
About this article
Cite this article
Akamine, Y., Miura, M., Komori, H. et al. Effects of one-time apple juice ingestion on the pharmacokinetics of fexofenadine enantiomers. Eur J Clin Pharmacol 70, 1087–1095 (2014). https://doi.org/10.1007/s00228-014-1705-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-014-1705-y