Abstract
Purpose
Phenprocoumon, similar to other coumarin-derived anticoagulants, is associated with a large variation in the individual dose requirement to achieve stable anticoagulation. Polymorphisms in the vitamin K epoxide reductase complex subunit 1 (VKORC1) and the liver enzyme cytochrome P450 2C9 (CYP2C9) effectively account for the variability in warfarin and acenocoumarol response but are less well-defined pharmacogenetic predictors in phenprocoumon therapy.
Methods
A retrospective study was performed on 185 outpatients attending anticoagulation clinics in Austria and Germany. These patients were genotyped for the VKORC1 −1639G>A and 3730G>A polymorphisms as well as for the CYP2C9 *2 and *3 polymorphisms using a reverse hybridisation-based teststrip assay.
Results
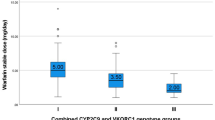
The VKORC1 −1639A allele, which was present at a frequency of 41.4% in the study cohort, significantly reduced the mean weekly phenprocoumon dose by 3 mg (19%) in the heterozygous and by 6.7 mg (43%) in the homozygous state compared to wild-type carriers (15.5 ± 6.8 mg, p < 0.0001). A stepwise multiple regression analysis revealed that VKORC1 −1639G>A, age and CYP2C9*3 were the major independent determinants of phenprocoumon dose, accounting for 14.2, 9.1 and 4.7% of its variability, respectively (p ≤ 0.0007). The CYP2C9*2 polymorphism had a marginal influence (1.4%) and failed to reach statistical significance (p = 0.062). The VKORC1 3730G>A genotype had no additional predictive power for individual dose variability.
Conclusion
Similar to warfarin and acenocoumarol, the VKORC1 −1639G>A polymorphism had the highest impact on the maintenance dose of phenprocoumon. The factor age was the second most important predictor and explained a greater percentage of the variability than CYP2C9 genotype.

Similar content being viewed by others
References
Li T, Chang CY, Jin DY, Lin PJ, Khvovora A, Stafford D (2004) Identification of the gene for vitamin K epoxide reductase. Nature 427:541–544
Rost S, Fregin A, Ivaskevicius V et al (2004) Mutations in VKORC1 cause warfarin resistance and multiple coagulation factor deficiency type 2. Nature 427:537–541
Harrington DJ, Underwood S, Morse C, Shearer MJ, Tuddenham EGD, Mumford AD (2005) Pharmacodynamic resistance to warfarin associated with a Val66Met substitution in vitamin K epoxide reductase complex subunit 1. Thromb Haemost 93:23–26
Bodin L, Horellou MH, Flaujac C, Loriot MA, Samama MM (2005) A vitamin K epoxide reductase complex subunit-1 (VKORC1) mutation in a patient with vitamin K antagonist resistance. J Thromb Haemost 3:1533–1535
Loebstein R, Dvoskin I, Halkin H et al (2007) A coding VKORC1 Asp36Tyr polymorphism predisposes to warfarin resistance. Blood 109:2477–2480
D’Andrea G, D’Ambrosio RL, Perna D et al (2005) A polymorphism in the VKORC1 gene is associated with an interindividual variability in the dose-anticoagulant effect of warfarin. Blood 105:645–649
Rieder MJ, Reiner AP, Gage BF et al (2005) Effect of VKORC1 Haplotypes on Transcriptional Regulation and Warfarin Doses. N Engl J Med 352:2285–2293
Wadelius M, Chen LY, Downes K et al (2005) Common VKORC1 and GGCX polymorphisms associated with warfarin dose. Pharmacogenomics J 5:262–270
Ufer M (2005) Comparative pharmacokinetics of vitamin K antagonists. Warfarin, phenprocoumon and acenocoumarol. Clin Pharmacokinet 12:1227–1246
Margaglione M, Colaizzo D, D’Andrea G, Brancaccio V, Ciampa A, Grandone E, Di Minno G (2000) Genetic modulation of oral anticoagulation with warfarin. Thromb Haemost 84:775–778
Higashi MK, Veenstra DL, Kondo LM, Wittkowsky AK, Srinouanprachanh SL, Farin FM, Rettie AE (2002) Association between CYP2C9 genetic variants and anticoagulation-related outcomes during warfarin therapy. JAMA 287:1690–1698
Voora D, Eby C, Linder MW et al (2005) Prospective dosing of warfarin based on cytochrome P-450 2C9 genotype. Thromb Haemost 93:700–705
Hillman MA, Wilke RA, Yale SH et al (2005) A prospective, randomized pilot trial of model-based warfarin dose initiation using CYP2C9 genotype and clinical data. Clin Med Res 3:137–145
Hermida J, Zarza J, Alberca I, Montes R, Lopez ML, Molina E, Rocha E (2002) Differential effects of 2C9*3 and 2C9*2 variants of cytochrome P-450 CYP2C9 on sensitivity to acenocoumarol. Blood 99:4237–4239
Tassies D, Freire C, Pijoan J, Maragall S, Monteagudo J, Ordinas A, Reverter JC (2002) Pharmacogenetics of acenocoumarol: cytochrome P450 CYP2C9 polymorphisms influence dose requirements and stability of anticoagulation. Haematologica 87:1185–1191
Bodin L, Verstuyft C, Tregouet DA et al (2005) Cytochrome P450 2C9 (CYP2C9) and vitamin K epoxide reductase (VKORC1) genotypes as determinants of acenocoumarol sensitivity. Blood 106:135–140
Kirchheiner J, Ufer M, Walter EC et al (2004) Effects of CYP2C9 polymorphisms on the pharmacokinetics of R- and S-phenprocoumon in healthy volunteers. Pharmacogenetics 14:19–26
Ufer M, Kammerer B, Kahlich R, Kirchheiner J, Yasar U, Brockmöller J, Rane A (2004) Genetic polymorphisms of cytochrome P450 2C9 causing reduced phenprocoumon (S)-7-hydroxylation in vitro and in vivo. Xenobiotica 34:847–859
Carlquist JF, Horne BD, Muhlestein JB et al (2006) Genotypes of the cytochrome p450 isoform, CYP2C9, and the vitamin K epoxide reductase complex subunit 1 conjointly determine stable warfarin dose: a prospective study. J Thromb Thrombolysis 22(3):191–197
Schwarz U, Ritchie MD, Bradford Y et al (2008) Genomic determinants of response to warfarin during initial anticoagulation. N Engl J Med 358:999–1008
Meckley LM, Wittkowsky AK, Rieder MJ, Rettie AE, Veenstra DL (2008) An analysis of the relative effects of VKORC1 and CYP2C9 variants on anticoagulation related outcomes in warfarin-treated patients. Thromb Haemost 100:229–239
Wadelius M, Chen LY, Lindh JD et al (2009) The largest prospective warfarin-treated cohort supports genetic forecasting. Blood 113:784–792
Schalekamp T, Brasse BP, Roijers JF et al (2006) VKORC1 and CYP2C9 genotypes and acenocoumarol anticoagulation status: interaction between both genotypes affects overanticoagulation. Clin Pharmacol Ther 80:13–22
Spreafico M, Lodigiani C, van Leeuwen Y et al (2008) Effects of CYP2C9 and VKORC1 on INR variations and dose requirements during initial phase of anticoagulation therapy. Pharmacogenomics 9:1237–1250
Schalekamp T, Oosterhof M, van Meegen E et al (2004) Effects of cytochrome P450 2C9 polymorphisms on phenprocoumon anticoagulation status. Clin Pharmacol Ther 76:409–417
Schalekamp T, Brasse BP, Roijers JF et al (2007) VKORC1 and CYP2C9 genotypes and phenprocoumon anticoagulation status: Interaction between both genotypes affects dose requirement. Clin Pharmacol Ther 81:185–193
Hummers-Pradier E, Hess S, Adham IM, Pieske B, Kochen MM (2003) Determination of bleeding risk using genetic markers in patients taking phenprocoumon. Eur J Clin Pharmacol 59:213–219
Visser LE, van Vliet M, van Schaik RH et al (2004) The risk of overanticoagulation in patients with cytochrome P450 CYP2C9*2 or CYP2C9*3 alleles on acenocoumarol or phenprocoumon. Pharmacogenetics 14:27–33
Sconce EA, Khan TI, Wynne HA et al (2005) The impact of CYP2C9 and VKORC1 genetic polymorphism and patient characteristics upon warfarin dose requirements: proposal for a new dosing regimen. Blood 106:2329–2333
Zhu Y, Shennan M, Reynolds KK, Johnson NA, Herrnberger MR, Valdes R Jr, Linder MW (2007) Estimation of warfarin maintenance dose based on VKORC1 (−1639G>A) and CYP2C9 genotypes. Clin Chem 53:1199–1205
Anderson JL, Horne BD, Stevens SM et al (2007) Randomized trial of genotyped-guided versus standard warfarin dosing in patients initiating oral anticoagulation. Circulation 116:2563–2570
Gage BF, Eby C, Johnson JA et al (2008) Use of pharmacogenetic and clinical factors to predict the therapeutic dose of warfarin. Clin Pharmacol Ther 84:326–331
The International Warfarin Pharmacogenetics Consortium (2009) Estimation of the warfarin dose with clinical and pharmacogenetic data. N Engl J Med 360:753–764
Oberkanins C, Moritz A, de Villiers JN, Kotze MJ, Kury F (2000) A reverse-hybridization assay for the rapid and simultaneous detection of nine HFE gene mutations. Genet Test 4:121–124
Montes R, Ruiz de Gaona E, Martinez-Gonzales MA, Alberca I, Hermida J (2006) The c.−1639G>A polymorphism if the VKORC1 gene is a major determinant of the response to acenocoumarol in anticoagulated patients. Br J Haematol 133:183–187
Yuan HY, Chen JJ, Lee MTM et al (2005) A novel functional VKORC1 promoter polymorphism is associated with inter-individual and inter-ethnic differences in warfarin sensitivity. Hum Mol Gen 14:1745–1751
Herman D, Peternel P, Stegnar M, Breskvar K, Dolzan V et al (2006) The influence of sequence variations in factor VII, γ-glutamyl carboxylase and vitamin K epoxide reductase complex genes on warfarin dose requirement. Throm Haemost 95:782–787
Borgiani P, Ciccacci C, Forte V, Romano S, Federici G, Novelli G (2007) Allelic variants in the CYP2C9 and VKORC1 loci and interindividual variability in the anticoagulant dose effect of warfarin in Italians. Pharmacogenomics 8:1545–1550
Cooper GM, Johnson JA, Langaee TY et al (2008) A genome-wide scan for common genetic variants with a large influence on warfarin maintenance dose. Blood 112:1022–1027
Takeuchi F, McGinnis R, Bourgeois S et al (2009) A genome-wide association study confirms VKORC1, CYP2C9, and CYP4F2 as principal Genetic Determinants of Warfarin Dose. PLoS Genet 5(3):e10000433
Caldwell MD, Awad T, Johnson JA et al (2008) CYP4F2 genetic variant alters required warfarin dose. Blood 111:4106–4112
Perez-Andreu V, Roldan V, Anton AI, Garcia-Barbera N, Corral J, Vicente V, Gonzalez-Conejero R (2009) Pharmacogenetic relevance of CYP4F2 V433M polymorphism on acenocoumarol therapy. Blood 113:4977–4979
Good AC, Henz S (2007) A clinical algorithm to predict the loading dose of phenprocoumon. Thromb Res 120:921–925
Werner D, Werner U, Wuerfel A, Grosch A, Lestin HG, Eschenhagen T, Rau T (2009) Pharmacogenetic characteristics of patients with complicated phenprocoumon dosing. Eur J Clin Pharmacol 65:783–788
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Puehringer, H., Loreth, R.M., Klose, G. et al. VKORC1 −1639G>A and CYP2C9*3 are the major genetic predictors of phenprocoumon dose requirement. Eur J Clin Pharmacol 66, 591–598 (2010). https://doi.org/10.1007/s00228-010-0809-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-010-0809-2