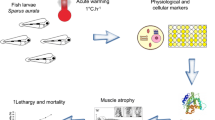
Abstract
Temperate species undergo wide variations of abiotic factors according to season, with temperature and photoperiod being two of the main factors that change. Given that several laboratory studies have reported the capacity of intertidal species to trigger the cellular stress response when exposed to challenging environmental conditions, the aim of this study was to address this capacity in situ (38°42′N, 9°29′W) in a key intertidal species, the shrimp Palaemon elegans. This study assessed changes in (1) thermal stress (heat-shock protein 70 kDa—Hsp70, total ubiquitin—Tub) and oxidative stress biomarkers (antioxidant enzymes: catalase—CAT, superoxide dismutase—SOD, glutathione-S-transferase—GST, and oxidation products: lipid peroxidation—LPO) in the muscle and visceral mass, and assessed changes in (2) ion concentrations (Ca2+, Na+, K+) in the muscle of shrimps under spring and summer conditions in the wild. Tissue-specific biomarker responses were detected, with greater induction in the muscle (increased Tub, CAT, SOD and LPO). Visceral mass showed decreased (Tub, CAT, GST) or constant (Hsp70, SOD) biomarker levels except for LPO, which increased in summer-acclimatized animals. Potassium ion increased in the muscle of summer-acclimatized shrimps suggesting ionic regulation. This study showed that P. elegans employs different biochemical strategies depending on the season, potentially as a mechanism of seasonal acclimatization involving the protein quality control system, oxidative stress pathways and ionic regulation. These effects should be considered in environmental monitoring programs to avoid the interference of confounding variables, which change seasonally.
Graphical Abstract






Similar content being viewed by others
References
Abele D, Puntarulo S (2004) Formation of reactive species and induction of antioxidant defence systems in polar and temperate marine invertebrates and fish. Comp Biochem Physiol A 138:405–415
Abele D, Heise K, Pörtner HO, Puntarulo S (2002) Temperature-dependence of mitochondrial function and production of reactive oxygen species in the intertidal mud clam Mya arenaria. J Exp Biol 205:1831–1841
Ahearn GA, Mandal PK, Mandal A (2004) Calcium regulation in crustaceans during the molt cycle: a review and update. Comp Biochem Physiol A 137:247–257
Bradford MM (1976) Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Botton MI, Pogorzelska M, Smoral L, Shehata A, Hamilton MG (2006) Thermal biology of horseshoe crab embryos and larvae: a role for heat shock proteins. J Exp Mar Biol Ecol 336:65–73
Cayman (2013) Catalase Assay Kit, item 707002, 13 pg. https://www.caymanchem.com/pdfs/707002.pdf. Acessed 15 May 2015
Charmantier G, Charmatier-Doures M, Towle D (2009) Osmotic and ionic regulation in aquatic arthropods. In: Evans DH (ed) Osmotic and ionic regulation: cells and animals. CRC Press, Taylor and Francis, Boca Raton, pp 165–230
Cripe GM (1994) Induction of maturation and spawning of pink shrimp, Panaeus duorarum, by changing water temperature, and survival and growth of young. Aquaculture 128(3–4):255–260
Cossins AR, Schwarzbaum PJ, Wieser W (1995) Effects of temperature on cellular ion regulation and membrane transport systems. In: Hochachka PW, Mommsen TP (eds) Biochemistry and molecular biology of fishes. Environmental and Ecological Biochemistry, vol 5. Elsevier, Amsterdam, pp 101–126
Cuculescu M, Hyde D, Bowler K (1998) Thermal tolerance of two species of marine crab, Cancer pagurus and Carcinus maenas. J Therm Biol 23(2):107–110
Dietz TJ, Somero GN (1992) The threshold induction temperature of the 90-kDa heat shock protein is subject to acclimatization in eurythermal goby fishes (genus Gillichthys). Proc Natl Acad Sci USA 89:3389–3393
Dietz TJ, Somero GN (1993) Species- and tissue-specific synthesis patterns for heat shock proteins HSP70 and HSP90 in several marine teleost fishes. Physiol Zool 66:863–880
Dorgelo J (1981) Blood osmoregulation and temperature in crustaceans. Hydrobiologia 81:113–130
Dowd WW (2012) Challenges for biological interpretation of environmental proteomics data in non-model organisms. Integr Comp Biol 52(5):705–720
Fiszbein A, Cánepa M, Vázquez GR, Maggese C, Pandolfi M (2010) Photoperiodic modulation of reproductive physiology and behavior in the cichlid fish Cichlassoma dimerus. Physiol Behav 99:425–432
Freire CA, Welker AF, Storey JM, Storey KB, Hermes-Lima M (2011) Oxidative stress in estuarine and intertidal environments (temperate and tropical). In: Abele D, Vazquez-Medina JP, Zenteno-Savin T (eds) Oxidative stress in aquatic ecosystems. Wiley Blackwell, USA, pp 41–57
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione-S-transferases: the first enzymatic step in mercapturicacid formation. J Biol Chem 246:7130–7139
Halliwell B, Gutteridge JMC (1999) Free radicals in biology and medicine. Oxford University Press, New York
Hamdoun AM, Cheney DP, Cherr GN (2003) Phenotypic plasticity of HSP70 and HSP70 gene expression in the Pacific oyster (Crassostrea gigas): implications for thermal limits and induction of thermal tolerance. Biol Bull 205:160–169
Helmuth B, Mieszkowska N, Moore P, Hawkins SJ (2006) Living on the edge of two changing worlds: forecasting the responses of rocky intertidal ecosystems to climate change. Annu Rev Ecol Evol Syst 37:373–404
Hershko A, Ciechanover A (1992) The ubiquitin system for protein degradation. Annu Rev Biochem 61:761–807
Hochachka PW, Somero GN (2002) Biochemical adaptation: Mechanism and process in physiological evolution. Oxford University Press, New York
Hoffman GL (1996) Methods of analysis by the U.S. Geological Survey National Water Quality Laboratory—Preparation procedure for aquatic biological material determined for trace metals: U.S. Geological Survey Open-File Report 96-362
Hofmann GE (1999) Ecologically relevant variation in induction and function of heat shock proteins in marine organisms. Am Zool 39:889–900
Hofmann GE, Somero GN (1995) Evidence for protein damage at environmental temperatures: seasonal changes in levels of ubiquitin conjugates and Hsp70 in the intertidal mussel Mytilus trossulus. J Exp Biol 198:1509–1518
Hopkin RS, Qari S, Bowler K, Hyde D, Cuculescu M (2006) Seasonal thermal tolerance in marine Crustacea. J Exp Mar Biol Ecol 331:74–81
Janas U, Spicer J (2010) Seasonal and temperature effects on osmoregulation by the invasive prawn Palaemon elegans Rathke, 1837 in the Baltic Sea. Mar Biol Res 6:333–337
Johansson LH, Borg LAH (1988) A spectrophotometric method for determination of catalase activity in small tissue samples. Anal Biochem 174:331–336
Kagawa N, Mugyia Y (2002) Brain HSP70 mRNA expression is linked with plasma cortisol levels in goldfish (Carassius auratus) exposed to a potential predator. Zool Sci 19:735–740
Lahdes EO, Vainio LA (2003) Effect of season on the acute thermal tolerance and thermal inactivation of membrane (Mg2+ + Na+ + K+)- and Mg2+-adenosine triphosphatase activity of the Baltic Sea amphipods, Monoporeia affinis and Gammarus spp. Boreal Environ Res 8:147–157
Leal I (2014) Thermal tolerance and acclimation capacity in tropical and temperate coastal organisms. Dissertation, University of Lisbon, Portugal
Lesser MP, Kruse VA (2004) Seasonal temperature compensation in the horse mussel, Modiolus modiolus: metabolic enzymes, oxidative stress and heat shock proteins. Comp Biochem Physiol A 137:495–504
Logan CA, Somero GN (2011) Effects of thermal acclimation on transcriptional responses to acute heat stress in the eurythermal fish Gillichthys mirabilis (Cooper). Am J Physiol Regul Integr Comp Physiol 300:R1373–R1383
Madeira D, Narciso L, Cabral HN, Vinagre C, Diniz MS (2012) Hsp70 production patterns in coastal and estuarine organisms facing increasing temperatures. J Sea Res 73:137–147
Madeira D, Narciso L, Diniz MS, Vinagre C (2014) Synergy of environmental variables alters the thermal window and heat shock response: an experimental test with the crab Pachygrapsus marmoratus. Mar Environ Res 98:21–28
Madeira D, Mendonça V, Dias M, Roma J, Costa PM, Larguinho M, Vinagre C, Diniz MS (2015) Physiological, cellular and biochemical thermal stress response of intertidal shrimps with different vertical distributions: Palaemon elegans and Palaemon serratus. Comp Biochem Physiol A 183:107–115
Malanga G, Estevez MS, Calvo J, Abele D, Puntarulo S (2007) The effect of seasonality on oxidative metabolism in Nacella (Patinigera) magellanica. Comp Biochem Physiol A 146:551–558
Morris JP, Thatje S, Hauton C (2013) Opinion: the use of stress-70 proteins in physiology: a re-appraisal. Mol Ecol 22:1494–1502
Pörtner HO (2012) Integrating climate-related stressor effects on marine organisms: unifying principles linking molecule to ecosystem-level changes. Mar Ecol Prog Ser 470:273–290
Pörtner HO, Knust R (2007) Climate change affects marine fishes through the oxygen limitation of thermal tolerance. Science 315:95–97
Rabergh CM, Airaksinen S, Soitamo A, Bjorklund HV, Johansson T, Nikinmaa M, Sistonen L (2000) Tissue-specific expression of zebrafish (Danio rerio) heat shock factor 1 mRNAs in response to heat stress. J Exp Biol 203:1817–1824
Roberts DA, Hofmann GE, Somero GN (1997) Heat-shock protein expression in Mytilus californianus: acclimatization (seasonal and tidal-height comparisons) and acclimation effects. Biol Bull 192(2):309–320
Rosa R, Lopes AR, Pimentel M, Faleiro F, Baptista M, Trübenbach K, Narciso L, Dionísio G, Pegado MR, Repolho T, Calado R, Diniz M (2014) Ocean cleaning stations under a changing climate: biological responses of tropical and temperate fish-cleaner shrimp to global warming. Glob Chang Biol 20(10):3068–3079
Schröer M, Saphörster J, Bock C, Pörtner H-O (2011) Oxygen and capacity limited thermal tolerance of the lugworm Arenicola marina: a seasonal comparison. J Exp Mar Biol Ecol 409:300–309
Somero GN (2002) Thermal physiology and vertical zonation of intertidal animals: optima, limits, and costs of living. Integr Comp Biol 42(4):780–789
Somero GN (2010) The physiology of climate change: how potentials for acclimatization and genetic adaptation will determine ‘winners’ and ‘losers’. J Exp Biol 213:912–920
Sørensen JG, Kristensen TN, Loeschcke V (2003) The evolutionary and ecological role of heat shock proteins. Ecol Lett 6:1025–1037
Spicer JI (2014) What can an ecophysiological approach tell us about the physiological responses of marine invertebrates to hypoxia? J Exp Biol 217:46–56
Stephens PJ (1990) The effects of temperature on the physiology of crustacean nerves and muscles. J Therm Biol 15:15–24
Stillman JH, Tagmount A (2009) Seasonal and latitudinal acclimatization of cardiac transcriptome responses to thermal stress in porcelain crabs, Petrolisthes cinctipes. Mol Ecol 18:4206–4226
Sun Y, Oberley LW, Li Y (1988) A simple method for clinical assay of superoxide dismutase. Clin Chem 34:497–500
Uchiyama M, Mihara M (1978) Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem 86:271–278
Vernberg FJ, Silverthon SU (1979) Temperature and osmoregulation in aquatic species. In: Gilles R (ed) Mechanisms of osmoregulation in animals. Wiley, New York, pp 537–562
Viarengo A, Canesi L, Livingstone DR (1991) Seasonal variations in the antioxidant defence enzymes and lipid peroxidation of the digestive gland of mussels. Comp Biochem Physiol C 100:187–190
Vinagre C, Ferreira T, Matos L, Costa MJ, Cabral HN (2009) Latitudinal gradients in growth and spawning of sea bass, Dicentrarchus labrax, and their relationship with temperature and photoperiod. Estuar Coast Shelf Sci 81:375–380
Vinagre C, Madeira D, Mendonça V, Dias M, Roma J, Diniz MS (2014a) Effect of temperature in multiple biomarkers of oxidative stress in coastal shrimp. J Therm Biol 41:38–42
Vinagre C, Madeira D, Mendonça V, Dias M, Roma J, Diniz MS (2014b) Effect of increasing temperature in the differential activity of oxidative stress biomarkers in various tissues of the rock goby, Gobius paganellus. Mar Env Res 97:10–14
Woodin SA, Hilbish TJ, Helmuth B, Jones SJ, Wethey DS (2013) Climate change, species distribution models, and physiological performance metrics: predicting when biogeographic models are likely to fail. Ecol Evol 3(10):3334–3346
Yamane L, Gilman SE (2009) Opposite responses by an intertidal predator to increasing aquatic and aerial temperatures. Mar Ecol Prog Ser 393:27–36
Acknowledgments
The authors would like to thank Marta Dias and Carolina Madeira for the help provided during field sampling. In addition, authors would like to thank Carolina Madeira for the photograph used in the graphical abstract. This study had the support of the Portuguese Fundação Calouste Gulbenkian through the award given to D. Madeira ‘Research Stimulus Program 2012, Reference Number 126739’. This study also had the support of the Portuguese Fundação para a Ciência e a Tecnologia (FCT) [individual Grants: senior researcher position to C.V., SFRH/BD/80613/2011 to D.M.; Project Grants PTDC/MAR/119068/2010 and PTDC/MAR-EST/2141/2012; strategic Project Grants UID/Multi/04378/2013 and UID/MAR/04292/2013].
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Responsible Editor: H.-O. Pörtner.
Reviewed by undisclosed experts.
Rights and permissions
About this article
Cite this article
Madeira, D., Mendonça, V., Vinagre, C. et al. Is the stress response affected by season? Clues from an in situ study with a key intertidal shrimp. Mar Biol 163, 41 (2016). https://doi.org/10.1007/s00227-015-2803-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-015-2803-9