Abstract
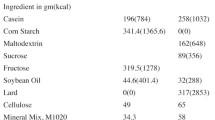
Diets high in fat or carbohydrates can lead to obesity and diabetes, two interrelated conditions that have been associated with osteoporosis. Here, we contrasted the effects of a high fat (HF) versus fructose-enriched carbohydrate (CH) versus regular chow (SC) diet on bone morphology, fat content and metabolic balance in BALB/cByJ mice over a 15-week period. For 13 weeks, there were no differences in body mass between groups with small differences in the last 2 weeks. Even without the potentially confounding factor of altered body mass and levels of load bearing, HF consumption was detrimental to bone in the distal femur with lower trabecular bone volume fraction and thinner cortices than controls. These differences in bone were accompanied by twofold greater abdominal fat content and fourfold greater plasma leptin concentrations. High-fat feeding caused a decrease in de-novo lipid synthesis in the liver, kidney, white adipose and brown adipose tissue. In contrast to HF, the fructose diet did not significantly impact bone quantity or architecture. Fructose consumption also did not significantly alter leptin levels or de-novo lipid synthesis but reduced epididymal adipose tissue and increased brown adipose tissue. Cortical stiffness was lower in the CH than in HF mice. There were no differences in glucose or insulin levels between groups. Together, a diet high in fat had a negative influence on bone structure, adipose tissue deposition and lipid synthesis, changes that were largely avoided with a fructose-enriched diet.


Similar content being viewed by others
References
Hill JO, Peters JC (1998) Environmental contributions to the obesity epidemic. Science (New York, N.Y.) 280:1371–1374
Asomaning K, Bertone-Johnson ER, Nasca PC, Hooven F, Pekow PS (2006) The association between body mass index and osteoporosis in patients referred for a bone mineral density examination. J Women’s Health 15:1028–1034
Viapiana O, Gatti D, Dalle Grave R, Todesco T, Rossini M, Braga V, Idolazzi L, Fracassi E, Adami S (2007) Marked increases in bone mineral density and biochemical markers of bone turnover in patients with anorexia nervosa gaining weight. Bone 40:1073–1077
Yancy WS Jr, Olsen MK, Guyton JR, Bakst RP, Westman EC (2004) A low-carbohydrate, ketogenic diet versus a low-fat diet to treat obesity and hyperlipidemia: a randomized, controlled trial. Ann Intern Med 140:769–777
McLaughlin T, Carter S, Lamendola C, Abbasi F, Yee G, Schaaf P, Basina M, Reaven G (2006) Effects of moderate variations in macronutrient composition on weight loss and reduction in cardiovascular disease risk in obese, insulin-resistant adults. Am J Clin Nutr 84:813–821
Sacks FM, Bray GA, Carey VJ, Smith SR, Ryan DH, Anton SD, McManus K, Champagne CM, Bishop LM, Laranjo N, Leboff MS, Rood JC, de Jonge L, Greenway FL, Loria CM, Obarzanek E, Williamson DA (2009) Comparison of weight-loss diets with different compositions of fat, protein, and carbohydrates. N Engl J Med 360:859–873
Softic S, Cohen DE, Kahn CR (2016) Role of dietary fructose and hepatic de novo lipogenesis in fatty liver disease. Dig Dis Sci 61:1282–1293
Chen Q, Shou P, Zheng C, Jiang M, Cao G, Yang Q, Cao J, Xie N, Velletri T, Zhang X (2016) Fate decision of mesenchymal stem cells: adipocytes or osteoblasts? Cell Death Differ 23:1128–1139
Styner M, Thompson WR, Galior K, Uzer G, Wu X, Kadari S, Case N, Xie Z, Sen B, Romaine A, Pagnotti GM, Rubin CT, Styner MA, Horowitz MC, Rubin J (2014) Bone marrow fat accumulation accelerated by high fat diet is suppressed by exercise. Bone 64:39–46
Halade GV, El Jamali A, Williams PJ, Fajardo RJ, Fernandes G (2011) Obesity-mediated inflammatory microenvironment stimulates osteoclastogenesis and bone loss in mice. Exp Gerontol 46:43–52
Chan ME, Adler BJ, Green DE, Rubin CT (2012) Bone structure and B-cell populations, crippled by obesity, are partially rescued by brief daily exposure to low-magnitude mechanical signals. FASEB J 26:4855–4863
Shu L, Beier E, Sheu T, Zhang H, Zuscik MJ, Puzas EJ, Boyce BF, Mooney RA, Xing L (2015) High-fat diet causes bone loss in young mice by promoting osteoclastogenesis through alteration of the bone marrow environment. Calcif Tissue Int 96:313–323
Cao JJ, Gregoire BR, Gao H (2009) High-fat diet decreases cancellous bone mass but has no effect on cortical bone mass in the tibia in mice. Bone 44:1097–1104
Patsch JM, Kiefer FW, Varga P, Pail P, Rauner M, Stupphann D, Resch H, Moser D, Zysset PK, Stulnig TM, Pietschmann P (2011) Increased bone resorption and impaired bone microarchitecture in short-term and extended high-fat diet-induced obesity. Metab Clin Exp 60:243–249
Lecka-Czernik B, Stechschulte LA, Czernik PJ, Dowling AR (2015) High bone mass in adult mice with diet-induced obesity results from a combination of initial increase in bone mass followed by attenuation in bone formation; implications for high bone mass and decreased bone quality in obesity. Mol Cell Endocrinol 410:35–41
Upadhyay J, Farr OM, Mantzoros CS (2015) The role of leptin in regulating bone metabolism. Metab Clin Exp 64:105–113
Lozano I, Van der Werf R, Bietiger W, Seyfritz E, Peronet C, Pinget M, Jeandidier N, Maillard E, Marchioni E, Sigrist S, Dal S (2016) High-fructose and high-fat diet-induced disorders in rats: impact on diabetes risk, hepatic and vascular complications. Nutr Metab 13:1–13
Whiting SJ, Healey A, Psiuk S, Mirwald R, Kowalski K, Bailey DA (2001) Relationship between carbonated and other low nutrient dense beverages and bone mineral content of adolescents. Nutr Res 21:1107–1115
Tjäderhane L, Larmas M (1998) A high sucrose diet decreases the mechanical strength of bones in growing rats. J Nutr 128:1807–1810
Lorincz C, Reimer RA, Boyd SK, Zernicke RF (2010) High-fat, sucrose diet impairs geometrical and mechanical properties of cortical bone in mice. Br J Nutr 103:1302–1308
Bass EF, Baile CA, Lewis RD, Giraudo SQ (2013) Bone quality and strength are greater in growing male rats fed fructose compared with glucose. Nutr Res 33:1063–1071
Yarrow JF, Toklu HZ, Balaez A, Phillips EG, Otzel DM, Chen C, Wronski TJ, Aguirre JI, Sakarya Y, Tümer N, Scarpace PJ (2016) Fructose consumption does not worsen bone deficits resulting from high-fat feeding in young male rats. Bone 85:99–106
Vos MB, Kimmons JE, Gillespie C, Welsh J, Blanck HM (2008) Dietary fructose consumption among US children and adults: the Third National Health and Nutrition Examination Survey. Medscape J Med 10:160
Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509
Leavens KF, Easton RM, Shulman GI, Previs SF, Birnbaum MJ (2009) Akt2 is required for hepatic lipid accumulation in models of insulin resistance. Cell Metab 10:405–418
Haas JT, Miao J, Chanda D, Wang Y, Zhao E, Haas ME, Hirschey M, Vaitheesvaran B, Farese RV Jr, Kurland IJ, Graham M, Crooke R, Foufelle F, Biddinger SB (2012) Hepatic insulin signaling is required for obesity-dependent expression of SREBP-1c mRNA but not for feeding-dependent expression. Cell Metab 15:873–884
Luu YK, Lublinsky S, Ozcivici E, Capilla E, Pessin JE, Rubin CT, Judex S (2009) In vivo quantification of subcutaneous and visceral adiposity by micro-computed tomography in a small animal model. Med Eng Phys 31:34–41
Judex S, Luu YK, Ozcivici E, Adler B, Lublinsky S, Rubin CT (2010) Quantification of adiposity in small rodents using micro-CT. Methods 50:14–19
Lublinsky S, Luu YK, Rubin CT, Judex S (2009) Automated separation of visceral and subcutaneous adiposity in in vivo microcomputed tomographies of mice. J Digit Imaging 22:222–231
Lublinsky S, Ozcivici E, Judex S (2007) An automated algorithm to detect the trabecular-cortical bone interface in micro-computed tomographic images. Calcif Tissue Int 81:285–293
Busa B, Miller LM, Rubin CT, Qin YX, Judex S (2005) Rapid establishment of chemical and mechanical properties during lamellar bone formation. Calcif Tissue Int 77:386–394
Scheller EL, Khoury B, Moller KL, Wee NK, Khandaker S, Kozloff KM, Abrishami SH, Zamarron BF, Singer K (2016) Changes in skeletal integrity and marrow adiposity during high-fat diet and after weight loss. Front Endocrinol 7. doi:10.3389/fendo.2016.00102
Buie HR, Moore CP, Boyd SK (2008) Postpubertal architectural developmental patterns differ between the L3 vertebra and proximal tibia in three inbred strains of mice. J Bone Miner Res 23:2048–2059
Zernicke RF, Salem GJ, Barnard RJ, Schramm E (1995) Long-term, high-fat-sucrose diet alters rat femoral neck and vertebral morphology, bone mineral content, and mechanical properties. Bone 16:25–31
Lidell M, Enerbäck S (2015) Brown adipose tissue and bone. Int J Obes Suppl 5:S23–S27
Tsanzi E, Light HR, Tou JC (2008) The effect of feeding different sugar-sweetened beverages to growing female Sprague–Dawley rats on bone mass and strength. Bone 42:960–968
Bergstra AE, Lemmens AG, Beynen AC (1993) Dietary fructose vs. glucose stimulates nephrocalcinogenesis in female rats. J Nutr 123:1320–1327
Milne DB, Nielsen FH (2000) The interaction between dietary fructose and magnesium adversely affects macromineral homeostasis in men. J Am Coll Nutr 19:31–37
Miller LM, Little W, Schirmer A, Sheik F, Busa B, Judex S (2007) Accretion of bone quantity and quality in the developing mouse skeleton. J Bone Miner Res 22:1037–1045
Tommasini SM, Trinward A, Acerbo AS, De Carlo F, Miller LM, Judex S (2012) Changes in intracortical microporosities induced by pharmaceutical treatment of osteoporosis as detected by high resolution micro-CT. Bone 50:596–604
Acerbo AS, Carr GL, Judex S, Miller LM (2012) Imaging the material properties of bone specimens using reflection-based infrared microspectroscopy. Anal Chem 84:3607–3613
Van Heek M, Compton DS, France CF, Tedesco RP, Fawzi AB, Graziano MP, Sybertz EJ, Strader CD, Davis HR Jr (1997) Diet-induced obese mice develop peripheral, but not central, resistance to leptin. J Clin Investig 99:385–390
Montgomery MK, Hallahan NL, Brown SH, Liu M, Mitchell TW, Cooney GJ, Turner N (2013) Mouse strain-dependent variation in obesity and glucose homeostasis in response to high-fat feeding. Diabetologia 56:1129–1139
Choi MS, Kim YJ, Kwon EY, Ryoo JY, Kim SR, Jung UJ (2015) High-fat diet decreases energy expenditure and expression of genes controlling lipid metabolism, mitochondrial function and skeletal system development in the adipose tissue, along with increased expression of extracellular matrix remodelling- and inflammation-related genes. Br J Nutr 113:867–877
Hwa JJ, Fawzi AB, Graziano MP, Ghibaudi L, Williams P, Van Heek M, Davis H, Rudinski M, Sybertz E, Strader CD (1997) Leptin increases energy expenditure and selectively promotes fat metabolism in ob/ob mice. Am J Physiol 272:R1204–R1209
Lin S, Thomas T, Storlien L, Huang X (2000) Development of high fat diet-induced obesity and leptin resistance in C 57 Bl/6 J mice. Int J Obes 24:639–646
Delgado TC, Pinheiro D, Caldeira M, Castro MM, Geraldes CF, Lopez-Larrubia P, Cerdan S, Jones JG (2009) Sources of hepatic triglyceride accumulation during high-fat feeding in the healthy rat. NMR Biomed 22:310–317
Oosterveer MH, van Dijk TH, Tietge UJF, Boer T, Havinga R, Stellaard F, Groen AK, Kuipers F, Reijngoud D-J (2009) High fat feeding induces hepatic fatty acid elongation in mice. PLoS ONE 4:e6066
Acknowledgements
We are grateful to Andrea Trinward, Charles Trujillo, Priya Vaitheesvaran, and Steven Tommasini for their technical contributions. Financial support from Stony Brook University School of Medicine TRO FUSION Award (SJ), NIH/NIAMS R01AR052778 (SJ), NASA NNX-12AL25G (SJ), and Einstein-Mount Sinai Diabetes Research Center Grant P60DK020541 (IJK) was greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Aditi Jatkar, Irwin J. Kurland and Stefan Judex declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
All procedures involving animals were approved by the IACUC at Stony Brook University.
Rights and permissions
About this article
Cite this article
Jatkar, A., Kurland, I.J. & Judex, S. Diets High in Fat or Fructose Differentially Modulate Bone Health and Lipid Metabolism. Calcif Tissue Int 100, 20–28 (2017). https://doi.org/10.1007/s00223-016-0205-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-016-0205-8