Abstract
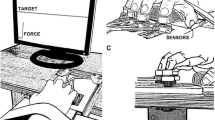
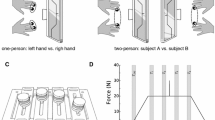
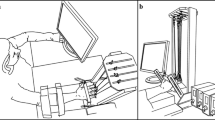
We explored stability of multi-finger cyclical accurate force production action by analysis of responses to small perturbations applied to one of the fingers and inter-cycle analysis of variance. Healthy subjects performed two versions of the cyclical task, with and without an explicit target. The “inverse piano” apparatus was used to lift/lower a finger by 1 cm over 0.5 s; the subjects were always instructed to perform the task as accurate as they could at all times. Deviations in the spaces of finger forces and modes (hypothetical commands to individual fingers) were quantified in directions that did not change total force (motor equivalent) and in directions that changed the total force (non-motor equivalent). Motor equivalent deviations started immediately with the perturbation and increased progressively with time. After a sequence of lifting–lowering perturbations leading to the initial conditions, motor equivalent deviations were dominating. These phenomena were less pronounced for analysis performed with respect to the total moment of force with respect to an axis parallel to the forearm/hand. Analysis of inter-cycle variance showed consistently higher variance in a subspace that did not change the total force as compared to the variance that affected total force. We interpret the results as reflections of task-specific stability of the redundant multi-finger system. Large motor equivalent deviations suggest that reactions of the neuromotor system to a perturbation involve large changes in neural commands that do not affect salient performance variables, even during actions with the purpose to correct those salient variables. Consistency of the analyses of motor equivalence and variance analysis provides additional support for the idea of task-specific stability ensured at a neural level.






Similar content being viewed by others
References
Bernstein NA (1967) The co-ordination and regulation of movements. Pergamon Press, Oxford
Danion F, Schöner G, Latash ML, Li S, Scholz JP, Zatsiorsky VM (2003) A mode hypothesis for finger interaction during multi-finger force-production tasks. Biol Cybern 88:91–98
Diedrichsen J, Shadmehr R, Ivry RB (2010) The coordination of movement: optimal feedback control and beyond. Trends Cogn Sci 14:31–39
Feldman AG (1966) Functional tuning of the nervous system with control of movement or maintenance of a steady posture. II. Controllable parameters of the muscle. Biophysics 11:565–578
Feldman AG (1986) Once more on the equilibrium-point hypothesis (λ model) for motor control. J Mot Behav 18:17–54
Feldman AG (2009) Origin and advances of the equilibrium-point hypothesis. Adv Exp Med Biol 629:637–643
Friedman J, SKM V, Zatsiorsky VM, Latash ML (2009) The sources of two components of variance: an example of multifinger cyclic force production tasks at different frequencies. Exp Brain Res 196:263–277
Hogan N, Sternad D (2007) On rhythmic and discrete movements: reflections, definitions and implications for motor control. Exp Brain Res 181:13–30
Hughes OM, Abbs JH (1976) Labial-mandibular coordination in the production of speech: implications for the operation of motor equivalence. Phonetica 33:199–221
Kelso JA, Tuller B, Vatikiotis-Bateson E, Fowler CA (1984) Functionally specific articulatory cooperation following jaw perturbations during speech: evidence for coordinative structures. J Exp Psychol Hum Percept Perform 10:812–832
Krishnamoorthy V, Latash ML, Scholz JP, Zatsiorsky VM (2003) Muscle synergies during shifts of the center of pressure by standing persons. Exp Brain Res 152:281–292
Latash ML (2010) Motor synergies and the equilibrium-point hypothesis. Mot Control 14:294–322
Latash ML (2012) The bliss (not the problem) of motor abundance (not redundancy). Exp Brain Res 217:1–5
Latash ML, Scholz JF, Danion F, Schöner G (2001) Structure of motor variability in marginally redundant multifinger force production tasks. Exp Brain Res 141:153–165
Latash ML, Shim JK, Smilga AV, Zatsiorsky VM (2005) A central back-coupling hypothesis on the organization of motor synergies: a physical metaphor and a neural model. Biol Cybern 92:186–191
Latash ML, Scholz JP, Schöner G (2007) Toward a new theory of motor synergies. Motor Control 11:276–308
Levin O, Wenderoth N, Steyvers M, Swinnen SP (2003) Directional invariance during loading-related modulations of muscle activity: evidence for motor equivalence. Exp Brain Res 148:62–76
Li ZM, Latash ML, Zatsiorsky VM (1998) Force sharing among fingers as a model of the redundancy problem. Exp Brain Res 119:276–286
Loeb GE (1999) What might the brain know about muscles, limbs and spinal circuits? Prog Brain Res 123:405–409
Martin V, Scholz JP, Schöner G (2009) Redundancy, self-motion, and motor control. Neural Comput 21:1371–1414
Martin JR, Budgeon MK, Zatsiorsky VM, Latash ML (2011) Stabilization of the total force in multi-finger pressing tasks studied with the ‘inverse piano’ technique. Hum Mov Sci 30:446–458
Mattos DJ, Latash ML, Park E, Kuhl J, Scholz JP (2011) Unpredictable elbow joint perturbation during reaching results in multijoint motor equivalence. J Neurophysiol 106:1424–1436
Mattos D, Kuhl J, Scholz JP, Latash ML (2013) Motor equivalence (ME) during reaching: is ME observable at the muscle level? Mot Control 17:145–175
Prochazka A, Clarac F, Loeb GE, Rothwell JC, Wolpaw JR (2000) What do reflex and voluntary mean? Modern views on an ancient debate. Exp Brain Res 130:417–432
Scholz JP, Schöner G (1999) The uncontrolled manifold concept: identifying control variables for a functional task. Exp Brain Res 126:289–306
Scholz JP, Schöner G, Latash ML (2000) Identifying the control structure of multijoint coordination during pistol shooting. Exp Brain Res 135:382–404
Scholz JP, Danion F, Latash ML, Schöner G (2002) Understanding finger coordination through analysis of the structure of force variability. Biol Cybern 86:29–39
Scholz JP, Schöner G, Hsu WL, Jeka JJ, Horak F, Martin V (2007) Motor equivalent control of the center of mass in response to support surface perturbations. Exp Brain Res 180:163–179
Scholz JP, Dwight-Higgin T, Lynch JE, Tseng YW, Martin V, Schöner G (2011) Motor equivalence and self-motion induced by different movement speeds. Exp Brain Res 209:319–332
Schöner G (1995) Recent developments and problem in human movement science and their conceptual implications. Ecol Psychol 7:23
Schöner G, Martin V, Reimann H, Scholz J (2008) Motor equivalence and the uncontrolled manifold. In: Proceedings 8th international seminar on speech production, Strasbourg, France, pp 23–28
Todorov E, Jordan MI (2002) Optimal feedback control as a theory of motor coordination. Nat Neurosci 5:1226–1235
Valero-Cuevas FJ, Smaby N, Venkadesan M, Peterson M, Wright T (2003) The strength-dexterity test as a measure of dynamic pinch performance. J Biomech 36:265–270
Wilhelm L, Zatsiorsky VM, Latash ML (2013) Equifinality and its violations in a redundant system: multifinger accurate force production. J Neurophysiol 110:1965–1973
Wing AM (2000) Motor control: mechanisms of motor equivalence in handwriting. Curr Biol 10:R245–R248
Zatsiorsky VM, Li ZM, Latash ML (1998) Coordinated force production in multi-finger tasks: finger interaction and neural network modeling. Biol Cybern 79:139–150
Zatsiorsky VM, Li ZM, Latash ML (2000) Enslaving effects in multi-finger force production. Exp Brain Res 131:187–195
Zhang W, Scholz JP, Zatsiorsky VM, Latash ML (2008) What do synergies do? Effects of secondary constraints on multidigit synergies in accurate force-production tasks. J Neurophysiol 99:500–513
Zhou T, Solnik S, Wu Y-H, Latash ML (2014) Equifinality and its violations in a redundant system: control with referent configurations in a multi-joint positional task. Motor Control 18:405–424
Acknowledgments
The study was supported by NIH Grants NS-035032 and AR-048563. We are grateful to Dr. Yen-Hsun Wu for his help at early stages of this project.
Author information
Authors and Affiliations
Corresponding author
Appendix: Variance and motor equivalence analyses
Appendix: Variance and motor equivalence analyses
The force data f were converted into a mode vector m by using the enslaving matrix E, where f = [f I, f M, f R, f L]T (T represents matrix transpose).
The Jacobian matrix J defining the linear map between changes in finger forces (df) modes (dm) and changes in total force dF TOT was defined:
The J matrix defining the changes between the finger force and modes (ƒ/m) and changes in total moment about the longitudinal axis of the forearm/hand with respect to the midpoint of the hand is:
where the d i entries representing the lever arm of fingers, d I = 4.5 cm, d M = 1.5 cm, d R = 1.5 cm, and d L = −4.5 cm. The UCM is the null-space of the Jacobian matrix J, spanned by the basis vectors ε i , solving:
For the variance analysis, the mean-free ƒ/m (∆x jk ) for a given j trial and k phase (pre-, during- and post-perturbation) was computed:
where x was either force or mode. The ∆x was projected into the null-space and orthogonal space of J as follows:
where \(\varvec{f}_{\parallel }\) is the ƒ parallel component and \(\varvec{f}_{ \bot }\) is the orthogonal component, n is the number of elemental variables (ƒ/m), and p is the number of constraints defined by the performance variable. There are n–p basis vectors, so that the null-space has n–p dimensions.
The variance across trials per degree of freedom along V ucm and orthogonal V ort to the UCM was computed.
For the motor equivalence analysis, the force/mode deviation vectors ∆x j were computed for each j trial by subtracting the mean pre-perturbed force/mode x 0,AV.
The alignment between x 0,AV and the x j involved temporal normalization of x 0,AV for each cycle of j trial. The ∆x j was projected along and orthogonal to the UCM according to Eqs. 11 and 12. The motor equivalence (ME) and non-motor equivalence components (nME) were computed as the length of the projection vector in each subspace, respectively, and normalized by the square root of the degrees of freedom of each space:
Rights and permissions
About this article
Cite this article
Mattos, D., Schöner, G., Zatsiorsky, V.M. et al. Motor equivalence during multi-finger accurate force production. Exp Brain Res 233, 487–502 (2015). https://doi.org/10.1007/s00221-014-4128-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-014-4128-1