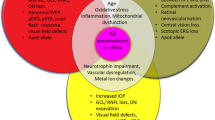
Abstract
Many age-related degenerative diseases of the central nervous system (CNS) increasingly appear to have similarities in their underlying causes. By applying knowledge between disorders, and in particular between degenerative diseases of different components of the CNS (e.g. the eye and the brain), we can begin to elucidate general mechanisms of neural degeneration. Age-related macular degeneration and glaucoma, two diseases of retinal neurons, which have recently been discussed in view of their common mechanisms with Alzheimer’s disease, highlight this perspective. This review discusses the common roles of the complement system (an immunological system) and glial cells (providing, amongst other functions, trophic support to neurons) in these three disorders. A number of facets of these systems would seem to be involved in the mechanisms of degeneration in at least two of the three diseases considered here. Regulatory proteins of the complement system (such as factor H), neurotrophin levels, and the interaction of microglia with the complement system in particular may be general to all three presentations of neural degeneration. Investigating the functioning of these fundamental systems across different diseases exemplifies the importance of considering advances in knowledge across a wider base than specific disease pathology. This may give insights both for understanding the function of these supporting systems and providing an avenue for developing future therapeutic targets general to neural degenerative diseases.

Similar content being viewed by others
References
Abbas A, Lichtman A, Pillai S (2007) Cellular and molecular immunology. Elsevier, Philadelphia
Ambati J, Fowler B (2012) Mechanisms of age-related macular degeneration. Neuron 75:26–39. doi:10.1016/j.neuron.2012.06.018
Anderson D, Talaga K, Rivest A, Barron E, Hageman G, Johnson L (2004) Characterisation of beta amyloid assemblies in drusen: the deposits associated with ageing and age-related macular degeneration. Exp Eye Res 78:243–256. doi:10.1016/j.exer.2003.10.011
Anderson D, Radeke M, Gallo N, Chapin E, Johnson P, Curletti C, Hancox L, Hu J, Ebright J, Malek G, Hauser M, Rickman C, Bok D, Hageman G, Johnson L (2010) The pivotal role of the complement system in ageing and age-related macular degeneration: hypothesis re-visited. Prog Retin Eye Res 29(2):95–112. doi:10.1016/j.preteyeres.2009.11.003
Atwood C, Martins R, Smith M, Perry G (2002) Senile plaque composition and posttranslational modification of amyloid-beta peptide and associated proteins. Peptides 23:1343–1350. doi:10.1016/S0196-9781(02)00070-0
Ballatore C, Lee V, Trojanowski J (2007) Tau-mediated neurodegeneration in Alzheimer’s disease and related disorders. Nat Rev Neurosci 8:663–672. doi:10.1038/nrn2194
Barghorn S, Nimmrich V, Striebinger A, Krantz C, Keller P, Janson B, Bahr M, Schmidt M, Bitner R, Harlan J, Varlow E, Ebert U, Hillen H (2005) Globular amyloid beta-peptide 1-42 oligomer—a homogenous and stable neuropathological protein in Alzheimer’s disease. J Neurochem 10:1–14. doi:10.1111/j.1471-4159.2005.03407.x
Bayer A, Ferrari F, Erb C (2002) High occurrence rate of glaucoma among patients with Alzheimer’s disease. Eur Neurol 47:165–168. doi:10.1159/000047976
Behl C, Davis J, Lesley R, Schubert D (1994) Hydrogen peroxide mediates amyloid-β protein toxicity. Cell 77:817–827. doi:10.1016/0092-8674(94)90131-7
Bhutto I, McLeod S, Hasegawa T, Kim S, Merges C, Tong P, Lutty G (2006) Pigment epithelium-derived factor and vascular endothelial growth factor in aged human choroid and eyes with age-related macular degeneration. Exp Eye Res 82(1):99–110. doi:10.1016/j.exer.2005.05.007
Binder C, Weismann D (2013) Patent No 20130011857. US Patent and Trademark Office, Washington, DC
Bora N, Kaliappan S, Jha P, Xu Q, Sivasankar B, Harris C, Morgan B, Bora P (2007) CD59: a complement regulatory protein, controls choroidal neovascularisation in a mouse model of wet-type age-related macular degeneration. J Immunol 178:1783–1790
Bora N, Jha P, Lyzogubov V, Kaliappan S, Liu J, Tytarenko R, Fraser D, Morgan B, Bora P (2010) Recombinant membrane-targeted form of CD59 inhibits the growth of choroidal neovascular complex in mice. J Biol Chem 285(44):33826–33833. doi:10.1074/jbc.M110.153130
Bosco A, Inman DM, Steele MR, Wu G, Soto I, Marsh-Armstrong N, Hubbard WC, Calkins DJ, Horner PJ, Vetter ML (2008) Reduced retina microglial activation and improved optic nerve integrity with minocycline treatment in the DBA/2J mouse model of glaucoma. Invest Ophthalmol Vis Sci 49(4):1437–1446. doi:10.1167/iovs.07-1337
Brantley M, Edelstein S, King J, Plotzke M, Apte R, Kymes S, Shiels A (2009) Association of complement factor H and LOC387715 genotypes with response of exudative age-related macular degeneration to photodynamic therapy. Eye 23:626–631. doi:10.1038/eye.2008.28
Bredt D, Snyder S (1994) Nitric oxide, a physiological messenger molecule. Ann Rev Biochem 63:175–195. doi:10.1146/annurev.bi.63.070194.001135
Buschini E, Piras A, Nuzzi R, Vercelli A (2011) Age related macular degeneration and drusen: neuroinflammation in the retina. Prog Neurobiol 95(1):14–25. doi:10.1016/j.pneurobio.2011.05.011
Caraci F, Battaglia G, Sortino M, Spampinato S, Molinaro G, Copani A, Nicoletti F, Bruno V (2012) Metabotropic glutamate receptors in neurodegeneration/neuroprotection: still a hot topic? Neurochem Int 61(4):559–565. doi:10.1016/j.neuint.2012.01.017
Cashman S, Ramo K, Kumar-Singh R (2011) A non membrane-targeted human soluble CD59 attenuates choroidal neovascularisation in a model of age related macular degeneration. PLoS One 6(4):e19078. doi:10.1371/journal.pone.0019078
Chandra J, Samali A, Orrenius S (2000) Triggering and modulation of apoptosis by oxidative stress. Free Radic Biol Med 29:323–333. doi:10.1016/S0891-5849(00)00302-6
Chiu K, Chan T, Wu A, Leung I, So K, Chang R (2012) Neurodegeneration of the retina in mouse models of Alzheimer’s disease: what can we learn from the retina? Age (Dordr) 34(3):633–649. doi:10.1007/s11357-011-9260-2
Chrysostomou V, Reznaia F, Trounce I, Crowston J (2012) Oxidative stress and mitochondrial dysfunction in glaucoma. Curr Opin Pharmacol 13:1–4. doi:10.1016/j.coph.2012.09.008
Cingle K, Kalski R, Bruner W, O’Brien C, Erhard P, Wyszynski R (1996) Age-related changes of glycosidases in human retinal pigment epithelium. Curr Eye Res 15(4):433–438. doi:10.3109/02713689608995834
Collard C, Väkevä A, Morrissey M, Agah A, Rollins S, Reenstra W, Buras J, Meri S, Stahl G (2000) Complement activation after oxidative stress: role of the lectin complement pathway. Am J Pathol 156(5):1549–1556. doi:10.1016/S0002-9440(10)65026-2
Connor B, Young D, Yan Q, Faull R, Synek B, Dragunow M (1997) Brain-derived neurotrophic factor is reduced in Alzheimer’s disease. Mol Brain Res 499(1–2):71–81. doi:10.1016/S0169-328X(97)00125-3
Cordeiro M, Guo L, Luong V, Harding G, Wang W, Jones H, Moss S, Sillito A, Fitzke F (2004) Real-time imaging of single nerve cell apoptosis in retinal neurodegeneration. Proc Natl Acad Sci USA 101:13352–13356. doi:10.1079/pnas.0405479101
Crabb J, Miyagi M, Gu X, Shadrach K, West K, Sakaguchi H, Kamei M, Hasan A, Yan L, Rayborn M, Salomon R, Hollyfield J (2002) Drusen proteome analysis: an approach to the etiology of age-related macular degeneration. Proc Natl Acad Sci 99(23):14682–14687. doi:10.1073/pnas.222551899
Curcio C, Allen K (1990) Topography of ganglion cells in human retina. J Comp Neurol 300(1):5–25. doi:10.1002/cne.903000103
Dawson V, Dawson T (1996) Nitric oxide actions in neurochemistry. Neurochem Int 29(2):97–110. doi:10.1016/0197-0186(95)00149-2
Day A, Clark S, Bishop P (2011) Understanding the molecular basis of age-related macular degeneration and how the identification of new mechanisms may aid the development of novel therapies. Expert Rev Ophthalmol 6(2):123–128. doi:10.1586/EOP.11.10
de Castro J, Mullins R, Manea A, Hernandez J, Wallen T, Kuehn M (2013) Lipofuscin in human glaucomatous optic nerves. Exp Eye Res 111:61–66. doi:10.1016/j.exer.2013.03.019
Ding X, Patel M, Chan C (2009) Molecular pathology of age-related macular degeneration. Prog Retin Eye Res 28(1):1–18. doi:10.1016/j.preteyeres.2008.10.001
Dowson J (1982) Neuronal lipofuscin accumulation in ageing and Alzheimer dementia: a pathogenic mechanism? Br J Psychiatry 140:142–148. doi:10.1192/byp.140.2.142
Dunaief J (2006) Iron induced oxidative damage as a potential factor in age-related macular degeneration: the Cogan lecture. Investig Ophthalmol Vis Sci 47:4660–4664. doi:10.1167/iovs.06-0568
Emerich D, Thanos C (2008) NT-501: an ophthalmic implant of polymer-encapsulated ciliary neurotrophic factor-producing cells. Curr Opin Mol Ther 10(5):506–515
Esparza T, Zhao H, Cirrito J, Cairns N, Bateman R, Holtzman D, Brody D (2013) Amyloid-beta oligomerization in Alzheimer dementia vs high pathology controls. Ann Neurol 73(1):104–119. doi:10.1002/ana.23748
Farina C, Aloisi F, Meinl E (2007) Astrocytes are active players in cerebral innate immunity. Trends Immunol 28(3):138–145. doi:10.1016/j.it.2007.01.005
Farkas R, Chowers I, Hackam A, Kageyama M, Nickells R, Otteson D, Duh E, Wang C, Valenta D, Gunatilaka T, Pease M, Quigley H, Zack D (2004) Increased expression of iron-regulating genes in monkey and human glaucoma. Investig Ophthalmol Vis Sci 45:1410–1417. doi:10.1167/iovs.03-0872
Fillit H, Ding W, Buee L, Kalman J, Altstiel L, Lawlor B, Wolf-Klein G (1991) Elevated circulating tumor necrosis factor levels in Alzheimer’s disease. Neurosci Lett 129(2):318–320. doi:10.1016/0304-3940(91)90490-K
Fischer B, Schmoll H, Platt D, Popa-Wagner A, Riederer P, Bauer J (1995) Complement C1q and C3 mRNA expression in the frontal cortex of Alzheimer’s patients. J Mol Med 73(9):465–471. doi:10.1007/BF00202265
Fishelson Z, Attali G, Mevorach D (2001) Complement and apoptosis. Mol Immunol 38(2–3):207–219. doi:10.1016/S0161-5890(01)00055-4
Gaipl U, Voll R, Sheriff A, Franz S, Kalden J, Hermann M (2005) Impaired clearance of dying cells in systemic lupus erythematosus. Autoimmun Rev 4:189–194. doi:10.1016/j.autrev.2004.10.007
Garcia P, Youssef I, Utvij J, Florent-Béchard S, Barthélémy V, Malaplate-Armand C, Kriem B, Stenger C, Koziel V, Olivier J, Escanye M, Hanse M, Allouche A, Desbene C, Yen F, Bjerkvig R, Oster T, Niclou S, Pillot T (2010) Ciliary neurotrophic factor cell-based delivery prevents synaptic impairment and improves memory in mouse models of Alzheimer’s disease. J Neurosci 30(22):4182–4190. doi:10.1523/JNEUROSCI.4182-09.2010
Gasque P, Dean Y, McGreal E, VanBeck J, Morgan B (2000) Complement components of the innate immune system in health and disease in the CNS. Immunopharmacol 49:171–186. doi:10.1016/S0162-3109(00)80302-1
Gellrich M, Gellrich N (1996) Quantitative relations in the retinal ganglion cell layer of the rat: neurons, glia, and capillaries before and after optic nerve section. Graefe’s Arch Clin Exp Ophthalmol 234(5):315–323. doi:10.1007/BF00220707
Girouard H, Iadecola C (2006) Neurovascular coupling in the normal brain and in hypertension, stroke, and Alzheimer disease. J Appl Physiol 100:328–335. doi:10.1152/japplphysiol.00966.2005
Glabe C (2006) Common mechanisms of amyloid oligomer pathogenesis in degenerative disease. Neurobiol Ageing 27:570–575. doi:10.1016/j.neurobiolaging.2005.04.017
Glovinsky Y, Quigley H, Pease M (1993) Foveal ganglion cell loss is size dependent in experimental glaucoma. Investig Ophthalmol Vis Sci 34(2):395–400
Goldberg J (2012) NT-501 CNTF implant for glaucoma: safety, neuroprotection, and neuroenhancement In: ClinicalTrialsgov [Internet] Bethesda (MD): Natl Library of Med (US). http://clinicaltrials.gov/show/NCT01408472
Gonzalez C (2012) Age macular degeneration-Alzheimer disease: relevance and interest of ophthalmologic exam in detection and follow-up of Alzheimer disease. Acta Ophthalmol 90(s249). doi:10.1111/j.1755-3768.2012.S087.x
Griffith L, Mathes M, Schmitz B (1995) Beta-amyloid precursor protein is modified with O-linked N-acetylglucosamine. J Neurosci Res 41(2):270–278. doi:10.1002/jnr.490410214
Guo L, Salt T, Luong V, Wood N, Cheung W, Maass A, Ferrari G, Russo-Marie F, Sillito A, Cheetham M, Moss S, Fitzke F, Cordeiro M (2007) Targeting amyloid-β in glaucoma treatment. Proc Natl Acad Sci 104:13444–13449. doi:10.1073/pnas.0703707104
Gupta N, Brown K, Milam A (2003) Activated microglia in human retinitis pigmentosa, late-onset retinal degeneration, and age-related macular degeneration. Exp Eye Res 76(4):463–471. doi:10.1016/S0014-4835(02)00332-9
Hecker L, Edwards A, Ryu E, Tosakulwong N, Baratz K, Brown W, Issa P, Scholl H, Pollok-Kopp B, Schmid-Kubista K, Bailey K, Oppermann M (2010) Genetic control of the alternative pathway of complement in humans and age-related macular degeneration. Hum Mol Genet 19:209–215. doi:10.1093/hmg/ddp472
Holtkamp G, Kijlstra A, Peek R, de Vos A (2001) Retinal pigment epithelium-immune system interactions: cytokine production and cytokine-induced changes. Prog Retin Eye Res 20(1):29–48. doi:10.1016/S1350-9462(00)00017-3
Howell G, Zhu X, Van Eldik L, Watterson M, John S (2010) Assessing microglia/macrophages in DBA/2J glaucoma. Assoc Res Vis Ophthalmol 2098:A197
Jessen K (2004) Glial cells. Int J Biochem Cell Biol 36:1861–1867. doi:10.1016/j.biocell.2004.02.023
Jha P, Bora P, Bora N (2007) The complement system and ocular diseases. Mol Immunol 44(16):3901–3908. doi:10.1016/j.molimm.2007.06.145
Johnson L, Leitner W, Rivest A, Staples M, Radeke M, Anderson D (2002) The Alzheimer’s amyloid beta-peptide is deposited at sites of complement activation in pathologic deposits associated with ageing and age-related macular degeneration. Proc Natl Acad Sci USA 99:11830–11835. doi:10.1073/pnas.192203399
Kam J, Lenassi E, Jeffrey G (2010) Viewing ageing eyes: diverse sites of amyloid Beta accumulation in the ageing mouse retina and the up-regulation of macrophages. PLoS One 5:e13127. doi:10.1371/journal.pone.0013127
Khoury J, Luster A (2008) Mechanisms of microglia accumulation in Alzheimer’s disease: therapeutic implications. Trends Pharmacol Sci 29(12):626–632. doi:10.1016/j.tips.2008.08.004
Kim S, Kim J, Lee J, Cho S, Kang H, Kim K, Jin D (2013) Intravitreal human complement factor H in a rat model of laser-induced choroidal neovascularisation. Br J Ophthalmol 97(3):367–370. doi:10.1136/bjophthalmol-2012-302307
Kuehn M, Fingert J, Young K (2005) Retinal ganglion cell death in glaucoma: mechanisms and neuroprotective strategies. Ophthalmol Clin North Am 18:383–395. doi:10.1016/j.ohc.2005.04.002
Kumar S, Peña L, de Vellis J (1993) CNS glial cells express neurotrophin receptors whose levels are regulated by NGF. Mol Brain Res 17(1–2):163–168. doi:10.1016/0169-328X(93)90086-5
Kwon Y, Fingert J, Kuehn M, Alward W (2009) Primary open-angle glaucoma. N Engl J Med 360:1113–1124. doi:10.1056/NEJMra0804630
LaFerla F, Green K, Oddo S (2007) Intracellular amyloid-beta in Alzheimer’s disease. Nat Rev Neurosci 8:499–509. doi:10.1038/nrn2168
Lambert W, Agarwal R, Howe W, Clark A, Wordinger R (2001) Neurotrophin and neurotrophin receptor expression by cells of the human lamina cribrosa. Investig Ophthalmol Vis Sci 42(10):2315–2323
Lanzerein A, Jobst K, Thiel S, Jensenius J, Sim R, Perry H, Sim E (1998) Mannan-binding lectin in human serum, cerebrospinal fluid and brain tissue and its role in Alzheimer’s disease. Clin Neurosci 9(7):1491–1495. doi:10.1097/00001756-199805110-00045
Laquis S, Chaudhary P, Sharma S (1998) The patterns of retinal ganglion cell death in hypertensive eyes. Brain Res 784(1–2):100–104. doi:10.1016/S0006-8993(97)01189-X
Law A, Gauthier S, Quirion R (2001) Say NO to Alzheimer’s disease: the putative links between nitric oxide and dementia of the Alzheimer’s type. Brain Res Rev 35(1):73–96. doi:10.1016/S0165-0173(00)00051-5
Le Fur I, Laumet G, Richard F, Fievet N, Berr C, Rouaud O, Delcourt C, Amouyel P, Lambert J (2010) Association study of the CFH Y402H polymorphism with Alzheimer’s disease. Neurobiol Ageing 31(1):165–166. doi:10.1016/j.neurobiolaging.2008.03.003
LeVine S (1997) Iron deposits in multiple sclerosis and Alzheimer’s disease brains. Brain Res 760:298–303. doi:10.1016/S0006-8993(97)00470-8
Liu B, Neufeld AH (2001) Nitric oxide synthase-2 in human optic nerve head astrocytes induced by elevated pressure in vitro. Arch Ophthalmol 119(2):240–245. doi:10-1001/pubs.Ophthalmol.-ISSN-0003-9950-119-2-els00027
Liu B, Chen H, Johns T, Neufeld A (2006) Epidermal growth factor receptor activation: an upstream signal for transition of quiescent astrocytes into reactive astrocytes after neural injury. J Neurosci 26(28):7532–7540. doi:10.1523/JNEUROSCI.1004-06.2006
Lott I, Head E (2005) Alzheimer disease and Down syndrome: factors in pathogenesis. Neurobiol Ageing 26(3):383–389. doi:10.1016/j.neurobiolaging.2004.08.005
Lucin K, Wyss-Coray T (2009) Immune activation in brain ageing and neurodegeneration: too much or too little? Neuron 64(1):110–122. doi:10.1016/j.neuron.2009.08.039
Luibl V, Isas J, Kayed R, Glabe C, Langen R, Chen J (2006) Drusen deposits associated with ageing and age-related macular degeneration contain nonfibrillar amyloid oligomers. J Clin Investig 116(2):378–385. doi:10.1172/JCI25843
Lukiw W, Zhao Y, Cui J (2008) An NF-kB-sensitive micro RNA-146a-mediated inflammatory circuit in Alzheimer disease and in stressed human brain cells. J Biol Chem 283(46):31315–31322
Lukiw W, Surjyadipta B, Dua P, Alexandrov P (2012) Common microRNAs (miRNAs) target complement factor H (CFH) regulation in Alzheimer’s disease (AD) and in age-related macular degeneration (AMD). Int J Biochem Mol Biol 3:105–116
Ma W, Coon S, Zhao L, Fariss R, Wong W (2013) A2E accumulation influences retinal microglial activation and complement regulation. Neurobiol Aging 34:943–960. doi:10.1016/j.neurobiolaging.2012.06.010
Mattson M, Goodman Y (1995) Different amyloidogenic peptides share a similar mechanism of neurotoxicity involving reactive oxygen species and calcium. Brain Res 676:219–224. doi:10.1016/0006-8993(95)00148-J
McKinnon S (2003) Glaucoma: ocular Alzheimer’s disease? Front Biosci 8:1140–1156. doi:10.2741/1172
Mey J, Thanos S (1993) Intravitreal injections of neurotrophic factors support the survival of axotomized retinal ganglion cells in adult rats in vivo. Brain Res 602(2):304–317. doi:10.1016/0006-8993(93)90695-J
Mold C, Morris C (2001) Complement activation by apoptotic endothelial cells following hypoxia/reoxygenation. Immunology 102:359–364. doi:10.1046/j.1365-2567.2001.01192.x
Morgan J, Caprioli J, Koseki Y (1999) Nitric oxide mediates excitotoxic and anoxic damage in rat retinal ganglion cells co-cultured with astroglia. Arch Ophthalmol 117(11):1524–1529. doi:10.1001/archopht.117.11.1524
Mousa S, Lorelli W, Campochiaro P (1999) Role of hypoxia and extracellular matrix-integrin binding in the modulation of angiogenic growth factors secretion by retinal pigmented epithelial cells. J Cell Biochem 74:135–143. doi:10.1002/(SICI)1097-4644(19990701)74:1<135::AID-JCB15>3.0.CO;2-#
Mullins R, Hagemann G (1999) Human ocular drusen possess novel core domains with a distinct carbohydrate composition. J Histochem Cytochem 47(12):1533–1539. doi:10.1177/002215549904701205
Nathanson J, McKee M (1995) Alterations of ocular nitric oxide synthase in human glaucoma. Invest Ophthalmol Vis Sci 36(9):1774–1784
Neufeld A (1999) Nitric oxide: a potential mediator of retinal ganglion cell damage in glaucoma. Surv Ophthalmol 43(S1):S129–S135. doi:10.1016/S0039-6257(99)00010-7
Neufeld A, Sawada A, Becker B (1999) Inhibition of nitric-oxide synthase 2 by aminoguanidine provides neuroprotection of retinal ganglion cells in a rat model of chronic glaucoma. Proc Natl Acad Sci USA 96:9944–9948. doi:10.1073/pnas.96.17.9944
Newman E, Reichenbach A (1996) The Müller cell: a functional element of the retina. Trends Neurosci 19(8):307–312. doi:10.1016/0166-2236(96)10040-0
Nischler C, Oberkofer H, Ortner C, Paikl D, Riha W, Lang N, Patsch W, Egger S (2011) Complement factor H Y402H gene polymorphism and response to intravitreal bevacizumab in exudative age-related macular degeneration. Acta Ophthalmol 89(4):344–349. doi:10.1111/j.1755-3768.2010.02080.x
Nunomura A, Castellani R, Zhu X, Moreira P, Perry G, Smith M (2006) Involvement of oxidative stress in Alzheimer disease. J Neuropathol Exp Neurol 65(7):631–641. doi:10.1097/01.jnen.0000228136.58062.bf
Ohno-Mastui K (2011) Parallel findings in age-related macular degeneration and Alzheimer’s disease. Prog Retin Eye Res 30:217–238. doi:10.1016/j.preteyeres.2011.02.004
Pak K, Chan S, Mattson M (2003) Presenilin-1 mutation sensitizes oligodendrocytes to glutamate and amyloid toxicities and exacerbates which matter damage and memory impairment in mice. NeuroMol Med 3:53–64. doi:10.1385/NMM:3:1:53
Paulssen R, Moe B, Gonaas H, Orbo A (2008) Gene expression in endometrial cancer cells after short time high dose exposure to progesterone. Steroids 73(1):116–128. doi:10.1016/j.steroids.2007.09.010
Pease M, Zack D, Berlinicke C, Bloom K, Cone F, Wang Y, Klein R, Hauswirth W, Quiley H (2009) Effect of CNTF on retinal ganglion cell survival in experimental glaucoma. Investig Ophthalmol Vis Sci 50(5):2194–2200. doi:10.1167/iovs.08-3013
Posthumus R (1952) The use and the possibilities of progesterone in the treatment of glaucoma. Ophthalmol 124(1):17–25
Qu J, Wang D, Grosskreutz C (2010) Mechanisms of retinal ganglion cell injury and defense in glaucoma. Exp Eye Res 91:48–53. doi:10.1016/j.exer.2010.04.002
Quigley H, McKinnon S, Zack D, Pease M, Kerrigan-Baumrind L, Kerrigan D, Mitchell R (2000) Retrograde axonal transport of BDNF in retinal ganglion cells is blocked by acute IOP elevation in rats. Investig Ophthalmol Vis Sci 41(11):3460–3466
Roca-Santiago H, Lago-Bouza J, Millán-Calenti J, Gómez-Ulla-Irazazábal F (2006) Alzheimer’s disease and age-related macular degeneration. Arch Spanish Soc Ophthalmol 81:73–78. doi:10.4321/S0365-66912006000200006
Roh M, Zhang Y, Murakami Y, Thanos A, Lee S, Vavvas D, Benowitz L, Miller J (2012) Etancercept, a widely used inhibitor of tumor necrosis factor α prevents retinal ganglion cell loss in a rat model of glaucoma. PLoS One 7(7):e40065. doi:10.1371/journal.pone.0040065
Roth K, D’Sa C (2001) Apoptosis and brain development. Ment Retard Dev Disabil Res Rev 7(4):261–266. doi:10.1002/mrdd.1036
Rus H, Cudrici C, Niculescu F (2005) The role of the complement system in innate immunity. Immunol Res 33(2):103–112. doi:10.1385/IR:33:2:103
Saurenmann R, Levin A, Rose J, Parker S, Rabinovitch T, Tyrrell P, Feldman B, Laxer R, Schneider R, Silverman E (2006) Tumour necrosis factor α inhibitors in the treatment of childhood uveitis. Rheumatol 45(8):982–989. doi:10.1093/rheumatology/ke1030
Sávári M, Vágó I, Wéber C, Nagy J, Gál P, Mák M, Kósa J, Závodszky P, Pázmány T (2003) Inhibition of C1q-beta-amyloid binding protects hippocampal cells against complement mediated toxicity. J Neuroimmunol 137(1–2):12–18. doi:10.1016/S0165-5728(03)00040-7
Schindowki K, Belarbi K, Buée L (2008) Neurotrophic factors in Alzheimer’s disease: role of axonal transport. Genes Brain Behav 7(S1):43–56. doi:10.1111/j.1601-183X.2007.00378.x
Selkoe D (2002) Alzheimer’s disease is a synaptic failure. Science 298:789–791. doi:10.1126/science.1074069
Shepard AR, Clark A, Klimko P, Wax M (2009) WIPO Patent No 2009061721. World Intellectual Property Organization, Geneva, Switzerland
Sihota R, Lakshmaiah N, Walia K, Sharma S, Pailoor J, Agarwal H (2001) The trabecular meshwork in acute and chronic angle closure glaucoma. Indian J Ophthalmol 49(4):255–259
Sivak J (2013) The ageing eye: common degenerative mechanisms between the Alzheimer’s brain and retinal disease. Investig Ophthalmol Vis Sci 54(1):871–880. doi:10.1167/iovs.12-10827
Skurla E, Rogers J, Sunderland T (1988) Direct assessment of activities of daily living in Alzheimer’s disease: a controlled study. J Am Geriatr Soc 36(2):97–103
Sohn J, Bora P, Jha P, Tezel T, Kaplan H, Bora N (2007) Complement, innate immunity and ocular disease. In: Niederkorn J, Kaplan H (eds) Immune response and the eye, vol 92. Karger, Basel, pp 105–114
Stasi K, Nagel D, Yang X, Wang R, Ren L, Podos S, Mittag T, Danias J (2006) Complement component 1Q upregulation in retina of murine, primate, and human glaucomatous eyes. Investig Ophthalmol Vis Sci 47(3):1024–1029. doi:10.1167/iovs.05-0830
Stevens B, Allen N, Vazquez L, Howell G, Christopherson K, Nouri N, Micheva K, Mehalow A, Huberman A, Stafford B, Sher A, Litke A, Lambris J, Smith S, John S, Barres B (2007) The classical complement cascade mediates CNS synapse elimination. Cell 131:1164–1178. doi:10.1016/j.cell.2007.10.036
Strasser A, O’Connor L, Dixit V (2000) Apoptosis signalling. Ann Rev Biochem 69:217–245. doi:10.1146/annurev.biochem.69.1.217
Strauss O (2005) The retinal pigment epithelium in visual function. Physiol Rev 85(3):845–881. doi:10.1007/s00347-008-1869-x
Streit W (2005) Microglia and neuroprotection: implications for Alzheimer’s disease. Brain Res Rev 48(2):234–239. doi:10.1016/j.brainresrev.2004.12.013
Tan B, Choi R, Chin T, Kaur C, Ling E (2012) Manipulation of microglial activity as a therapy for Alzheimer’s disease. Front Biosci 4:1402–1412. doi:10.2741/342
Taylor C, Senchyna M, Flanagan J, Joyce E, Cliché D, Boone A, Culp-Stewart S, Thompson J (2004) Role of eIF5A in TNF-α-mediated apoptosis of lamina cribrosa cells. Investig Ophthalmol Vis Sci 45(10):3568–3576. doi:10.1167/iovs.03-1367
Taylor S, Calder C, Albon J, Erichsen J, Boulton M, Morgan J (2011) Involvement of the CD200 receptor complex in microglia activation in experimental glaucoma. Exp Eye Res 92(5):338–343. doi:10.1016/j.exer.2011.01.012
Tezel G, Wax M (2000) Increased production of tumor necrosis factor-alpha by glial cells exposed to simulated ischemia or elevated hydrostatic pressure induces apoptosis in co-cultured retinal ganglion cells. J Neurosci 20(23):8693–8700
Tezel G, Li L, Patil R, Wax M (2001) TNF-α and TNF-α receptor-1 in the retina of normal and glaucomatous eyes. Investig Ophthalmol Vis Sci 42(8):1787–1794
Tezel G, Yang X, Luo C, Kain A, Powell D, Kuehn M, Kaplan H (2010) Oxidative stress and the regulation of complement activation in human glaucoma. Investig Ophthalmol Vis Sci 51(10):5071–5082. doi:10.1167/iovs.10-5289
Theodossiadis P, Markomichelakis N, Sfikakis P (2007) Tumor necrosis factor antagonists: preliminary evidence for an emerging approach in the treatment of ocular inflammation. Retina 27(4):399–413. doi:10.1097/MAJ.0b013e3180318fbc
Tichaczek-Goska D (2012) Deficiencies and excessive human complement system activation in disorders of multifarious etiology. Adv Clin Exp Med 21(1):105–114
Tobinick E (2007) Perispinal etanercept for treatment of Alzheimer’s disease. Curr Alzheimer Res 4(5):550–552. doi:10.2174/156720507783018217
Tobinick E, Gross H (2008) Rapid cognitive improvement in Alzheimer’s disease following perispinal etanercept administration. J Neuroinflamm 5(2). doi:10.1186/1742-2094-5-2
Van der Schaft T, Mooy C, de Bruijn W, de Jong P (1993) Early stages of age-related macular degeneration: an immunofluorescence and electron microscopy study. Br J Ophthalmol 77:657–661. doi:10.1136/bjo.77.10.657
Walker D, Kim S, McGeer P (1995) Complement and cytokine gene expression in cultured microglia derived from post-mortem human brains. J Neurosci Res 40:478–493. doi:10.1002/jnr.490400407
Walton H, Dodd P (2007) Glutamate-glutamine cycling in Alzheimer’s disease. Neurochem Int 50(7–8):1052–1066. doi:10.1016/j.neuint.2006.10.007
Wang L, Chiang H, Wu W, Liang B, Xie Z, Yao X, Ma W, Du S, Zhong Y (2012) Epidermal growth factor receptor is a preferred target for treating Amyloid-beta-induced memory loss. Proc Natl Acad Sci USA 109(41):16743–16748. doi:10.1073/pnas.1208011109
Webster S, Lue L-F, Brachova L, Tenner A, McGeer P, Terai K, Walker D, Bradt B, Cooper N, Rogers J (1997) Molecular and cellular characterization of the membrane attack complex C5b-9 in Alzheimer’s disease. Neurobiol Aging 18(4):415–421. doi:10.1016/S0197-4580(97)00042-0
Wittchen H, Jacobi F, Rehm J, Gustavsson A, Svensson M, Jönsson B, Olesen J, Allgulander C, Alonso J, Faravelli C, Fratiglioni L, Jennum P, Lieb R, Maercker A, van Os J, Preisig M, Salvador-Carulla L, Simon R, Steinhausen H-C (2011) The size and burden of mental disorders and other disorders of the brain in Europe 2010. Eur Neuropsychopharmacol 21:655–679. doi:10.1016/j.euroneuro.2011.07.018
Wong W (2013) Microglial aging in the healthy CNS: phenotypes, drivers, and rejuvenation. Front Cell Neurosci 7(22):1–13. doi:10.3389/fncel.2013.00022
Wordinger R, Fleenor D, Hellberg P, Pang I, Tovar T, Zode G, Fuller J, Clark A (2007) Effects of TGF-β2: BMP-4: and Gremlin in the trabecular meshwork: implications for glaucoma. Investig Ophthalmol Vis Sci 48(3):1191–1200. doi:10.1167/iovs.06-0296
Yang L, Li R, Meri S, Rogers J, Shen Y (2000) Deficiency of complement defense protein CD59 may contribute to neurodegeneration in Alzheimer’s disease. J Neurosci 20(20):7505–7509
Yin H, Chen L, Chen X, Liu X (2008) Soluble amyloid beta oligomers may contribute to apoptosis of retinal ganglion cells in glaucoma. Med Hypotheses 71(1):77–80. doi:10.1016/j.mehy.2008.01.030
Yuan L, Neufeld A (2001) Activated microglia in the human glaucomatous optic nerve head. J Neurosci Res 64(5):523–532. doi:10.1002/jnr.1104
Yücel Y, Gupta N (2008) Glaucoma of the brain: a disease model for the study of transsynaptic neural degeneration. Prog Brain Res 173:465–478. doi:10.1016/S0079-6123(08)01132-1
Zetterberg M, Landgren S, Andersson M, Palmér M, Gustafson D, Skoog I, Minthon L, Thelle D, Wallin A, Bogdanovic N, Andreasen N, Blennow K, Zettenberg H (2008) Association of complement factor H Y402H gene polymorphism with Alzheimer’s disease. Am J Med Genet B Neuropsychiatr Genet 147(6):720–726. doi:10.1002/ajmg.b.30668
Zhang J, Dawson V, Dawson T, Snyder S (1994) Nitric oxide activation of poly (ADP-ribose) synthetase in neurotoxicity. Science 263:687–689. doi:10.1126/science.8080500
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Harvey, H., Durant, S. The role of glial cells and the complement system in retinal diseases and Alzheimer’s disease: common neural degeneration mechanisms. Exp Brain Res 232, 3363–3377 (2014). https://doi.org/10.1007/s00221-014-4078-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-014-4078-7