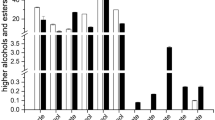
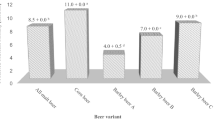
Abstract
Beer is a complex mixture of over 450 constituents and, in addition, it contains macromolecules such as proteins, nucleic acids, polysaccharides, and lipids. In beer, several different protein groups, originating from barley, barley malt, and yeast, are known to influence beer quality. Some of them play a role in foam formation and mouthfeel, and others are known to form haze and have to be precipitated to guarantee haze stability, since turbidity gives a first visual impression of the quality of beer to the consumer. These proteins are derived from the malt used and are influenced, modified, and aggregated throughout the whole malting and brewing process. During malting, barley storage proteins are partially degraded by proteinases into amino acids and peptides that are critical for obtaining high-quality malt and therefore high-quality wort and beer. During mashing, proteins are solubilized and transferred into the produced wort. Throughout wort boiling proteins are glycated and coagulated being possible to separate those coagulated proteins from the wort as hot trub. In fermentation and maturation process, proteins aggregate as well, because of low pH, and can be separated. The understanding of beer protein also requires knowledge about the barley cultivar characteristics on barley/malt proteins, hordeins, protein Z, and LTP1. This review summarizes the protein composition and functions and the changes of malt proteins in beer during the malting and brewing process. Also methods for protein identification are described.


Similar content being viewed by others
References
Kunze W (2007) Technologie Brauer und Mälzer, vol 9. VLB Berlin
Narziß L et al (1999) Die Technologie der Malzbereitung, Die Bierbrauerei. Ferdinand Enke Verlag, Stuttgart, Germany
Celus I et al (2006) The effects of malting and mashing on barley protein extractability. J Cereal Sci 44(2):203–211
Finnie C, Svensson B (2003) Feasibility study of a tissue-specific approach to barley proteome analysis: aleurone layer, endosperm, embryo and single seeds. J Cereal Sci 38(2):217–227
Finnie C, Svensson B (2009) Barley seed proteomics from spots to structures. J Proteomics 72(3):315–324
Jones BL et al (2000) Quantitative study of the formation of endoproteolytic activities during malting and their stabilities to kilning. J Agric Food Chem 48(9):3898–3905
Narziß L (2005) Abriss der Bierbrauerei, vol 7. Wiley VCH
Osman AM et al (2002) Characterisation and assessment of the role of barley malt endoproteases during malting and mashing. J Inst Brew 108(1):62–67
Sørensen S et al (1987) Primary structure of carboxypeptidase II from malted barley. Carlsberg Res Commun 52(4):285–295
Mikola L (1983) Germinating barley grains contain five acid carboxypeptidases with complementary substrate specificities. Biochim Biophys Acta (BBA) Protein Struct Mol Enzymol 747(3):241–252
Sørensen S et al (1986) Primary structure of carboxypeptidase I from malted barley. Carlsberg Res Commun 51(7):475–485
Sørensen S et al (1989) Primary structure of carboxypeptidase III from malted barley. Carlsberg Res Commun 54(5):193–202
Jones BL, Marinac L (2002) The effect of mashing on malt endoproteolytic activities. J Agric Food Chem 50(4):858–864
Jones BL (1997) Malt endoproteinases; their synthesis and inactivation during malting and mashing. Eur Symp Enzymes Grain Process Proc 1:54–64
Jones BL (2005) The endogenous endoprotease inhibitors of barley and malt and their roles in malting and brewing. J Cereal Sci 42(3):271–280
Jones BL (2008) The endoproteinases of barley and malt and their endogenous inhibitors. Tech Q MBAA Commun 45(3):279–282
Jones BL, Budde AD (1999) Endoproteinases and the hydrolysis of malt proteins during mashing. Proc Congr Eur Brew Conv 27:611–618
Osman AM (2003) Barley and malt proteins and proteinases: II. The purification and characterisation of five malt endoproteases, using the highly degradable barley protein fraction (HDBPF) substrate. J Inst Brew 109(2):142–149
Osman AM (2003) Barley and malt proteins and proteinases: I. Highly degradable barley protein fraction (HDBPF), a suitable substrate for Malt Endoprotease Assay. J Inst Brew 109(2):135–414
Osman AM (2003) Barley and malt proteins and proteinases: III. A simple method for estimating the combined actions of malt proteinases and the extent of protein degradation during malting. J Inst Brew 109(2):150–153
Jones BL (2005) Endoproteases of barley and malt. J Cereal Sci 42(2):139–156
Zhang NY, Jones BL (1995) Characterization of germinated barley endoproteolytic enzymes by 2-dimensional gel-electrophoresis. J Cereal Sci 21(2):145–153
Jones BL, Budde AD (2005) How various malt endoproteinase classes affect wort soluble protein levels. J Cereal Sci 41(1):95–106
Fontanini D, Jones Berne L (2002) SEP-1–a subtilisin-like serine endopeptidase from germinated seeds of Hordeum vulgare L. cv. Morex. Planta 215(6):885–893
Perrocheau L et al (2006) Stability of barley and malt lipid transfer protein 1 (LTP1) toward heating and reducing agents: relationships with the brewing process. J Agric Food Chem 54(8):3108–3113
Perrocheau L et al (2005) Probing heat-stable water-soluble proteins from barley to malt and beer. Proteomics 5(11):2849–2858
Vaag P, et al. (2000) Protein and cDNA sequences of e1 hordein from barley, wheat, and/or rye, and uses thereof to enhance the quality of foam in beer. Application: WOWO Patent 99-IB15972000014237
Vaag P et al (1999) Characterization of a beer foam protein originating from barley. Proc Congr Eur Brew Conv 27:157–166
Van Nierop SNE et al (2002) Studies on beer foam proteins in a commercial brewing process. Proce Conv Inst Brew (Asia Pacific Section) 27:35–40
Van Nierop SNE et al (2004) Impact of different wort boiling temperatures on the beer foam stabilizing properties of lipid transfer protein 1. J Agric Food Chem 52(10):3120–3129
Evans DE, et al. (1998) The influence of malt foam-positive proteins and non-starch polysaccharides on beer foam quality. Monogr Eur Brew Conv 27 (E.B.C.-Symp Beer Foam Q, 1998):114–128
Iimure T et al (2008) Novel prediction method of beer foam stability using protein Z, barley dimeric alpha -amylase inhibitor-1 (BDAI-1) and yeast thioredoxin. J Agric Food Chem 56(18):8664–8671
Osborne TB (1924) Vegetable proteins. 2nd ed. revised
Osborne TB (1924) Vegetable proteins. Am Food J 19:143
Osborne TB (1924) Vegetable proteins. Chem Age (London) 10:349
Osborne TB (1924) Vegetable proteins. Chem Ind 43:440
Osborne TB, Mendel LB (1924) Continuation and extension of work on vegetable proteins. Exp Stn Rec 53:364
Wieser H et al (1998) Quantitative determination of gluten protein types in wheat flour by reversed-phase high-performance liquid chromatography. Cereal Chem 75(5):644–650
Klose C et al (2008) Protein changes during barley malting. Brauwelt 148(36):1044–1045
Bak-Jensen KS et al (2004) Two-dimensional gel electrophoresis pattern (pH 6–11) and identification of water-soluble barley seed and malt proteins by mass spectrometry. Proteomics 4(3):728–742
Bobalova J et al (2008) Investigation of protein composition of barley by gel electrophoresis and MALDI mass spectrometry with regard to the malting and brewing process. J Inst Brew 114(1):22–26
Görg A et al (1992) Two-dimensional polyacrylamide gel electrophoresis, with immobilized pH gradients in the first dimension, of barley seed proteins: discrimination of cultivars with different malting grades. Electrophoresis 13(4):192–203
Görg A et al (1992) Detection of polypeptides and amylase isoenzyme modifications related to malting quality during malting process of barley by two-dimensional electrophoresis and isoelectric focusing with immobilized pH gradients. Electrophoresis 13(9–10):759–770
Kristoffersen HE, Flengsrud R (2000) Separation and characterization of basic barley seed proteins. Electrophoresis 21(17):3693–3700
Ostergaard O et al (2004) Proteome analysis of barley seeds: Identification of major proteins from two-dimensional gels (pI 4–7). Proteomics 4(8):2437–2447
Ostergaard O et al (2002) Initial proteome analysis of mature barley seeds and malt. Proteomics 2(6):733–739
Finnie C et al (2006) Differential appearance of isoforms and cultivar variation in protein temporal profiles revealed in the maturing barley grain proteome. Plant Sci (Amsterdam, Netherlands) 170(4):808–821
Finnie C et al (2004) Aspects of the barley seed proteome during development and germination. Biochem Soc Trans 32(3):517–519
Finnie C et al (2002) Proteome analysis of grain filling and seed maturation in barley. Plant Physiol 129(3):1308–1319
Finnie C et al (2003) Barley proteome analysis, starch degrading enzymes and proteinaceous inhibitors. J Appl Glycosci 50(2):277–282
Flengsrud R (1993) Separation of acidic barley endosperm proteins by two-dimensional electrophoresis. Electrophoresis 14(10):1060–1066
Flengsrud R, Kobro G (1989) A method for two-dimensional electrophoresis of proteins from green plant tissues. Anal Biochem 177(1):33–36
Metodiev MV et al (2002) Two-dimensional electrophoretic analysis of salicylic acid-induced changes in polypeptide pattern of barley leaves. Biol Plant 45(4):585–588
Shewry PR et al (1988) Two-dimensional electrophoresis of cereal prolamins: applications to biochemical and genetic analyses. Electrophoresis 9(11):727–737
Sorensen SB et al (1993) Barley lipid transfer protein 1 is involved in beer foam formation. Tech Q Master Brew Assoc Am 30(4):136–145
Stanislava G (2007) Barley grain non-specific lipid-transfer proteins (ns-LTPs) in beer production and quality. J Inst Brew 113(3):310–324
Weiss W et al (1991) Barley cultivar discrimination: I Sodium dodecyl sulfate-polyacrylamide gel electrophoresis and glycoprotein blotting. Electrophoresis 12(5):323–330
Weiss W et al (1991) Barley cultivar discrimination: II Sodium dodecyl sulfate-polyacrylamide gel electrophoresis and isoelectric focusing with immobilized pH gradients. Electrophoresis 12(5):330–337
Weiss W et al (1992) Qualitative and quantitative changes in barley seed protein patterns during the malting process analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis with respect to malting quality. Electrophoresis 13(9–10):787–797
Görg A et al (1988) Two-dimensional electrophoresis with immobilized pH gradients of leaf proteins from barley (Hordeum vulgare): method, reproducibility and genetic aspects. Electrophoresis 9(11):681–692
Williams KM, Marshall T (1995) Protein composition of beer as revealed by high-resolution two-dimensional electrophoresis. Anal Proc 32(1):25–28
Asano K, Hashimoto N (1980) Isolation and characterization of foaming proteins of beer. J Am Soc Brew Chem 38(4):129–137
Asano K et al (1982) Characterization of haze-forming proteins of beer and their roles in chill haze formation. J Am Soc Brew Chem 40(4):147–154
Iimure T et al (2009) Construction of a novel beer proteome map and its use in beer quality control. Food Chem 118(3):566–574
Iimure T et al (2009) Identification of novel haze-active beer proteins by proteome analysis. J Cereal Sci 49(1):141–147
Hejgaard J (1977) Origin of a dominant beer protein immunochemical identity with a beta-amylase-associated protein from barley. J Inst Brew 83(2):94–96
Hejgaard J (1982) Purification and properties of protein Z–a major albumin of barley endosperm. Physiol Plant 54:174–182
Hejgaard J (1984) Gene products of barley chromosomes 4 and 7 are precursors of the major antigenic beer protein. J Inst Brew 90(2):85–87
Hejgaard J, Bjoern SE (1985) Four major basic proteins of barley grain. Purification and partial characterization. Physiol Plant 64(3):301–307
Hejgaard J, Boeg-Hansen TC (1974) Quantitative immunoelectrophoresis of barley and malt proteins. J Inst Brew London 80(5):436–442
Hejgaard J, Carlsen S (1977) Immunoelectrophoretic identification of a heterodimer beta -amylase in extracts of barley grain. J Sci Food Agric 28(10):900–904
Hejgaard J, Gibbons GC (1979) Screening for a alpha -amylase in cereals. Improved gel-diffusion assay using a dye-labeled starch substrate. Carlsberg Res Commun 44(1):21–25
Hejgaard J, Soerensen SB (1975) Characterization of a protein-rich beer fraction by two-dimensional immunoelectrophoretic techniques. C R Trav Lab Carlsberg 40(16):187–203
Shewry PR (1993) Barley seed proteins. Barley, pp 131–97
Shewry PR et al (1978) Comparison of methods for extraction and separation of hordein fractions from 29 barley varieties. J Sci Food Agric 29(5):433–441
Shewry PR, Miflin BJ (1983) Characterization and synthesis of barley seed proteins. Seed Proteins, pp 143–205
Shewry PR, Miflin BJ (1985) Seed storage proteins of economically important cereals. Adv Cereal Sci Technol 7:1–83
Shewry PR et al (1978) Varietal identification of single seeds of barley by analysis of hordein polypeptides. J Sci Food Agric 29(7):587–596
Mills ENC et al (1998) Immunological study of hydrophobic polypeptides in beer. J Agric Food Chem 46(11):4475–4483
Ishibashi Y et al (1997) Application of ELISA to quantitative evaluation of foam-active protein in the malting and brewing processes. J Am Soc Brew Chem 55(1):20–23
Briggs D, Hough J (1981) Malting and brewing science: malt and sweet wort. Springer, New York
Michael G et al (1961) Die Eiweißqualität von Körnern verschiedener Getreidearten in Abhängigkeit von Stickstoffversorgung und Entwicklungszustand. Zeitschrift für Pflanzenernährung Düngung Bodenkunde 92(2):106–116
Wilson C et al (1981) The extraction and separation of barley glutelins and their relationship to other endosperm proteins. J Exp Bot 32(6):1287
Kirkman MA et al (1982) The effect of nitrogen nutrition on the lysine content and protein composition of barley seeds. J Sci Food Agric 33(2):115–127
Faulks AJ et al (1981) The polymorphism and structural homology of storage polypeptides (hordein) coded by the Hor-2 locus in barley (Hordeum vulgare). Biochem Genet 19(910):841–858
Miflin BJ, Shewry PR (1981) Seed storage proteins: genetics, synthesis, accumulation and protein quality. Dev Plant Soil Sci 3 (Nitrogen Carbon Metab): 195–248
Rahman S et al (1984) Hordein-gene expression during development of the barley (Hordeum vulgare) endosperm. Biochem J 223(2):315–322
Festenstein GN et al (1984) Immunochemical studies on barley seed storage proteins The specificity of an antibody to “C” hordein and its reaction with prolamins from other cereals. Planta 162(6):524–531
Faulks AJ et al (1981) The polymorphism and structural homology of storage polypeptides (hordein) coded by the Hor-2 locus in barley (Hordeum vulgare L). Biochem Genet 19(9–10):841–858
Melzer J, Kleinhofs A (1987) Molecular genetics of barley endosperm proteins. Barley Genet Newsl 17:13–24
Silva F et al (2008) Electrophoretic and HPLC methods for comparative study of the protein fractions of malts, worts and beers produced from Scarlett and Prestige barley (Hordeum vulgare L.) varieties. Food Chem 106:820–829
Baxter ED, Wainwright T (1979) Hordein and malting quality. J Am Soc Brew Chem 37(1):8–12
Forde BG et al (1985) Short tandem repeats shared by B- and C-hordein cDNAs suggest a common evolutionary origin for two groups of cereal storage protein genes. EMBO J 4(1):9–15
Shewry PR, Miflin BJ (1982) Genes for the storage proteins of barley. Qual Plant Plant Foods Hum Nutr 31(3):251–267
Baxter ED (1981) Hordein in barley and malt–a review. J Inst Brew 87(3):173–176
Wilson CM et al (1981) The extraction and separation of barley glutelins and their relationship to other endosperm proteins. J Exp Bot 32(131):1287–1293
Fox GP, Henry RJ (1995) Analysis of water-soluble proteins from barley by ion-exchange high performance liquid chromatography. J Inst Brew 101(3):181–185
Giese H, Hejgaard J (1984) Synthesis of salt-soluble proteins in barley. Pulse-labeling study of grain filling in liquid-cultured detached spikes. Planta 161(2):172–177
Hejgaard J (1976) Free and protein-bound beta -amylases of barley grain. Characterization by two-dimensional immunoelectrophoresis. Physiol Plant 38(4):293–299
Hejgaard J, Boisen S (1980) High-lysine proteins in Hiproly barley breeding: identification, nutritional significance and new screening methods. Hereditas (Lund, Swed.) 93(2):311–320
Rosenkrands I et al (1994) Serpins from wheat grain. FEBS Lett 343(1):75–80
Rasmussen SK et al (1991) cDNA cloning, characterization and expression of an endosperm-specific barley peroxidase. Plant Mol Biol 16(2):317–327
Giese H, Hopp H (1984) Influence of nitrogen nutrition on the amount of hordein, protein Z and -amylase messenger RNA in developing endosperms of barley. Carlsberg Res Commun 49(3):365–383
Brandt A et al (1990) A plant serpin gene. Structure, organization and expression of the gene encoding barley protein Z4. Eur J Biochem 194(2):499–505
Rasmussen SK et al (1984) A cDNA clone for protein Z, a major barley endosperm albumin. Carlsberg Res Commun 49(3):385–390
Doll H (1984) Nutritional aspects of cereal proteins and approaches to overcome their deficiencies. Philos Trans R Soc London Ser B 304(1120):373–380
Rasmussen SK (1993) A gene coding for a new plant serpin. Biochim Biophys Acta 1172(1–2):151–154
Gorjanovic S et al (2007) Antimicrobial activity of malting barley grain thaumatin-like protein isoforms, S and R. J Inst Brew 113(2):206–212
Bobalova J et al (2010) Monitoring of malting process by characterization of glycation of barley protein Z. Eur Food Res Technol 230(4):665–673
Heinemann B et al (1996) Structure in solution of a four-helix lipid binding protein. Protein Sci 5(1):13–23
Bakan B et al (2009) The crystal structure of oxylipin-conjugated barley LTP1 highlights the unique plasticity of the hydrophobic cavity of these plant lipid-binding proteins. Biochem Biophys Res Commun 390(3):780–785
Jegou S et al (2000) Purification and structural characterization of LTP1 polypeptides from beer. J Agric Food Chem 48(10):5023–5029
Douliez JP et al (2000) Structure, biological, and technological functions of lipid transfer proteins and indolines, the major lipid binding proteins from cereal kernels. J Cereal Sci 32(1):1–20
Gorjanovic S et al (2005) Malting barley grain non-specific lipid-transfer protein (ns-LTP): importance for grain protection. J Inst Brew 111(2):99–104
Jones BL, Marinac LA (1995) Barley LTP1 (PAPI) and LTP2: inhibitors of green malt cysteine endoproteinases. J Am Soc Brew Chem 53(4):194–195
Evans DE, Hejgaard J (1999) The impact of malt derived proteins on beer foam quality. Part I. The effect of germination and kilning on the level of protein Z4, protein Z7 and LTP1. J Inst Brew 105(3):159–169
Evans DE et al (1999) The impact of malt derived proteins on beer foam quality Part II: the influence of malt foam-positive proteins and non-starch polysaccharides on beer foam quality. J Inst Brew 105(3):171–177
Kaersgaard P, Hejgaard J (1979) Antigenic beer macromolecules: an experimental survey of purification methods. J Inst Brew 85(2):103–111
Lundgard R, Svensson B (1989) A 39 kD barley seed protein of the serpin superfamily inhibits alpha-chymotrypsin. Carlsberg Res Commun 54(5):173–180
Jones BL, Marinac LA (1997) Purification, identification, and partial characterization of a barley protein that inhibits green malt endoproteinases. J Am Soc Brew Chem 55(2):58–64
Steiner E, Back W (2009) A critical review of protein assays and further aspects of new methods in brewingscience. Brew Sci 62:90–94
Slack PT et al (1979) Inhibition by hordein of starch degradation. J Inst Brew 85(2):112–114
Osman AM et al (2003) The gel filtration chromatographic-profiles of proteins and peptides of wort and beer: effects of processing–malting, mashing, kettle boiling, fermentation and filtering. J Inst Brew 109(1):41–50
Bamforth CW (1999) Beer haze. J Am Soc Brew Chem 57(3):81–90
Curioni A et al (1995) Major proteins of beer and their precursors in barley: electrophoretic and immunological studies. J Agric Food Chem 43(10):2620–2626
Hejgaard J, Kaersgaard P (1983) Purification and properties of the major antigenic beer protein of barley origin. J Inst Brew 89(6):402–410
Nadzeyka A et al (1979) The significance of beer proteins in relationship to cold break and age-related haze formation. Brauwissenschaft 32(6):167–172
Siebert KJ et al (1996) Formation of protein-polyphenol haze in beverages. J Agric Food Chem 44(8):1997–2005
von Wettstein D (2007) From analysis of mutants to genetic engineering. Plant Biol 58(1):1
Leiper KA et al (2003) Beer polypeptides and silica gel. Part II. Polypeptides involved in foam formation. J Inst Brew 109(1):73–79
Leiper KA et al (2003) Beer polypeptides and silica gel Part I Polypeptides involved in haze formation. J Inst Brew 109(1):57–72
Loisa M et al (1971) Quantitative determination of some beer protein components by an immunological method. Brauwissenschaft 24(10):366–368
Bamforth C (2001) A brewer’s biochemistry. Brew Int 1(3):21–25
Siebert KJ (1999) Protein-polyphenol haze in beverages. Food Technol (Chicago) 53(1):54–57
Siebert KJ (1999) Effects of protein-polyphenol interactions on beverage haze, stabilization, and analysis. J Agric Food Chem 47(2):353–362
Djurtoft R (1965) Composition of the protein and polypeptide fraction of EBC beer haze preparations. J Inst Brew 71(4):305–315
Mussche R (1990) Physico-chemical stabilization of beer using new generation gallotannins. Proc Conv Inst Brew (Aust N Z Sect) 21:136–140
Outtrup H et al (1987) The interaction between proanthocyanidins and peptides. Proc Congr Eur Brew Conv 21:583–590
Belleau G, Dadic M (1981) Beer hazes. II. Further analyses of basic components by high-performance liquid chromatography. J Am Soc Brew Chem 39(4):142–146
Dadic M, Belleau G (1980) Beer hazes. I. Isolation and preliminary analysis of phenolic and carbohydrate components. J Am Soc Brew Chem 38(4):154–158
Portero-Otin M et al (2003) Protein modification by advanced Maillard adducts can be modulated by dietary polyunsaturated fatty acids. Biochem Soc Trans 31:1403–1405
Lapolla A et al (1993) The lysine glycation 1 A preliminary investigation on the products arising from the reaction of protected lysine and D-glucose. Amino Acids 5(3):389–401
Leiper KA et al (2005) Optimising beer stabilisation by the selective removal of tannoids and sensitive proteins. J Inst Brew 111(2):118–127
Evans DE et al (2003) Application of immunological methods to differentiate between foam-positive and haze-active proteins originating from malt. J Am Soc Brew Chem 61(2):55–62
Lázaro A et al (1985) Differential effects of high-lysine mutations on the accumulation of individual members of a group of proteins encoded by a disperse multigene family in the endosperm of barley (Hordeum vulgare L.). Eur J Biochem 149(3):617–623
Robinson LH et al (2007) The identification of a barley haze active protein that influences beer haze stability: the genetic basis of a barley malt haze active protein. J Cereal Sci 45(3):335–342
Robinson LH et al (2004) The interaction between malt protein quality and brewing conditions and their impact on beer colloidal stability. Tech Q MBAA Communicator 41(4):353–362
Evans DE, Sheehan MC (2002) Don’t be fobbed off: the substance of beer foam. J Am Soc Brew Chem 60(2):47–57
Kobayashi N et al (2002) A new method for evaluating foam-damaging effect by free fatty acids. J Am Soc Brew Chem 60(1):37–41
Bech LM et al (1995) Throughout the brewing process barley lipid transfer protein 1 (LTP1) is transformed into a more foam-promoting form. Proc Congr Eur Brew Conv 25:561–568
Hao J et al (2006) Identification of the major proteins in beer foam by mass spectrometry following sodium dodecyl sulfate-polyacrylamide gel electrophoresis. J Am Soc Brew Chem 64(3):166–174
Onishi A et al (1999) Monoclonal antibody probe for assessing beer foam stabilizing proteins. J Agric Food Chem 47(8):3044–3049
Kapp GR, Bamforth CW (2002) The foaming properties of proteins isolated from barley. J Sci Food Agric 82(11):1276–1281
Lusk L et al. (1998) Foam tower fractionation of beer proteins and bittering acids. Monograph European Brew Conv 27 (E.B.C.-Symp Beer Foam Q, 1998):166–187
Jin B et al (2009) Structural changes of malt proteins during boiling. Molecules 14(3):1081–1097
Jin B et al (2009) Proteomics study of silica eluent proteins in beer. J Am Soc Brew Chem 67(4):183–188
Kordialik-Bogacka E, Ambroziak W (2004) Investigation of foam-active polypeptides during beer fermentation. J Sci Food Agri 84(14):1960–1968
Kordialik-Bogacka E, Ambroziak W (2006) The relationship between polypeptides and foaming during fermentation. LWT Food Sci Technol 40(2):368–373
Okada Y et al (2008) The influence of barley malt protein modification on beer foam stability and their relationship to the barley dimeric alpha -amylase inhibitor-I (BDAI-I) as a possible foam-promoting protein. J Agric Food Chem 56(4):1458–1464
Marshall T, Williams KM (1987) High resolution two-dimensional electrophoresis of the proteins and macromolecular constituents of beer and wine. Electrophoresis 8(10):493–495
Hejgaard J (1978) ‘Free’ and ‘bound’ beta -amylases during malting of barley. Characterization by two-dimensional immunoelectrophoresis. J Inst Brew 84(1):43–46
Dahl SW et al (1996) Heterologous expression of three plant serpins with distinct inhibitory specificities. J Biol Chem 271(41):25083–25088
Dahl SW et al (1996) Inhibition of coagulation factors by recombinant barley serpin BSZx. FEBS Lett 394(2):165–168
Jegou S et al (2001) Evidence of the glycation and denaturation of LTP1 during the malting and brewing process. J Agric Food Chem 49(10):4942–4949
Dale CJ, Young TW (1988) Fractionation of high molecular weight polypeptides from beer using two dimensional gel electrophoresis. J Inst Brew 94(1):28–32
Esslinger HM (ed) (2009) Handbook of brewing: processes. Technology, Markets
Back W (2005) Ausgewählte Kapitel der Brauereitechnologie, vol 1. Fachverlag Hans Carl GmbH, Nürnberg
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Steiner, E., Gastl, M. & Becker, T. Protein changes during malting and brewing with focus on haze and foam formation: a review. Eur Food Res Technol 232, 191–204 (2011). https://doi.org/10.1007/s00217-010-1412-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-010-1412-6