Abstract
We report on application of conductive silver paste smeared glass slides as Raman spectroscopy sample substrates for label-free detection of HIV-1 p24 antigen in blood plasma. We also show that the same substrates can be applied in Raman spectroscopic screening of blood plasma for presence of HIV. The characteristic Raman spectrum of HIV-1 p24 antigen displayed prominent bands that were assigned to ribonucleic acids (RNA) and proteins that constitute the antigen. This spectrum can be used as reference during Raman spectroscopic screening for HIV in plasma within the first few days after exposure (<7 days). The Raman spectra obtained from HIV+ plasma displayed unique peaks centered at wavenumbers 928, 990, 1270, 1397, and 1446 cm−1 attributed to the Raman active vibrations in the virion carbohydrates, lipids, and proteins. Other bands similar to those reported in literature were also seen and assignments made. The attachment of the HIV virions to silver nanoparticles via gp120 glycoprotein knobs was thought to be responsible for the enhanced Raman signals of proteins associated with the virus. The principal component analysis (PCA) applied on the combined spectral data showed that HIV− and HIV+ spectra had differing spectral patterns. This indicated the great power of Raman spectroscopy in HIV detection when plasma samples are deposited onto silver paste smeared glass substrates. The Raman peaks responsible for the segregation of the spectral data in PCA were mainly those assigned to the viral proteins (645, 725, 813, 1270, and 1658 cm−1). Excellent results were obtained from Artificial Neural Network (ANN) applied on the HIV+ Raman spectral data around the prominent peak centered at 1270 cm−1 with R (coefficient of correlation) and R 2 (coefficient of determination) values of 0.9958 and 0.9895, respectively. The method has the potential of being used as quick blood screening for HIV before blood transfusion with the Raman peaks assigned to the virion proteins acting as reference.

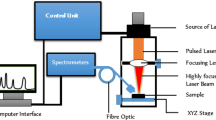
The HIV type 1 virus particle gets attached to the silver nanoparticle contained in the conductive silver paste smear onto a glass slide. This results in strong Raman signals associated with the components of the virion. The signals are collected, dispersed in a spectrometer and displayed on a computer screen. Method can be used as a label-free and rapid HIV screening in blood plasma





Similar content being viewed by others
References
(UNAIDS) JUNP on H, (UNAIDS) JUNP on H, others. Global AIDS Update 2016. Geneva Switz Httpwww Unaids Orgsitesdefaultfilesmediaassetglobal-AIDS-Update-2016en Pdf Accessed. 2016;28:16.
Olivia Block LN, Anirban M, Dykes C. A rapid label-free method for quantitation of human immunodeficiency virus type-1 particles by nanospectroscopy. J Virol Methods. 2012;182:70–5.
Shafiee H, Lidstone EA, Jahangir M, Inci F, Hanhauser E, Henrich TJ, et al. Nanostructured optical photonic crystal biosensor for HIV viral load measurement. Sci Rep. 2014;4:4116.
Tooley L. Detecting HIV earlier: advances in HIV testing. Prevention. 2010. http://www.catie.ca/en/pif/fall-2010/detecting-hiv-earlier-advances-hivtesting.
Shafiee H, Wang S, Inci F, Toy M, Henrich TJ, Kuritzkes DR, et al. Emerging technologies for point-of-care management of HIV infection. Annu Rev Med. 2015;66:387–405.
Lee J-H, Oh B-K, Choi J-W. Development of a HIV-1 virus detection system based on nanotechnology. Sensors. 2015;15(5):9915–27.
Sakudo A, Tsenkova R, Onozuka T, Morita K, Li S, Warachit J, et al. A novel diagnostic method for human immunodeficiency virus type-1 in plasma by near-infrared spectroscopy. Microbiol Immunol. 2005;49(7):695–701.
Barletta JM, Edelman DC, Constantine NT. Lowering the detection limits of HIV-1 viral load using real-time immuno-PCR for HIV-1 p24 antigen. Am J Clin Pathol. 2004;122(1):20–7.
Brust S, Duttmann H, Feldner J, Gürtler L, Thorstensson R, Simon F. Shortening of the diagnostic window with a new combined HIV p24 antigen and anti-HIV-1/2/O screening test. J Virol Methods. 2000;90(2):153–65.
Gan N, Du X, Cao Y, Hu F, Li T, Jiang Q. An ultrasensitive electrochemical immunosensor for HIV p24 based on Fe3O4@ SiO2 nanomagnetic probes and nanogold colloid-labeled enzyme–antibody copolymer as signal tag. Materials. 2013;6(4):1255–69.
Lee J-H, Kim B-C, Byeung-Keun O, Choi J-W. Rapid and sensitive determination of HIV-1 virus based on surface enhanced Raman spectroscopy. J Biomed Nanotechnol. 2015;11(12):2223–30.
Zhou L, Huang J, Yu B, Liu Y, You T. A novel electrochemiluminescence immunosensor for the analysis of HIV-1 p24 antigen based on P-RGO@ Au@ Ru-SiO2 composite. ACS Appl Mater Interfaces. 2015;7(44):24438–45.
Zinin PV, Hu N, Kamemoto LE, Yu Q, Misra AK, Sharma SK. Raman spectroscopy of HIV-1 antigen and antibody. In: SPIE Defense, Security, and Sensing. International Society for Optics and Photonics. 2011. p. 80250D.
Xu Y, Lu C. Raman spectroscopic study on structure of human immunodeficiency virus (HIV) and hypericin-induced photosensitive damage of HIV. Sci China Ser C Life Sci. 2005;48(2):117–32.
Isola NR, Stokes DL, Vo-Dinh T. Surface-enhanced Raman gene probe for HIV detection. Anal Chem. 1998;70(7):1352–6.
Hu J, Zheng P-C, Jiang J-H, Shen G-L, Yu R-Q, Liu G-K. Sub-attomolar HIV-1 DNA detection using surface-enhanced Raman spectroscopy. Analyst. 2010;135(5):1084–9.
Benuel N, Michael KK, Raphael WL, Samoel AK, Ernest PM, Joyceline GK, et al. HIV type 1 gag genetic diversity among antenatal clinic attendees in North Rift Valley, Kenya. AIDS Res Hum Retroviruses. 2012;28(5):523–6.
Sheila K, Raphael WL, Vincent O, Matilu M, Fredrick AO, Elijah MS, et al. HIV type 1 subtype surveillance in Central Kenya. AIDS Res Hum Retroviruses. 2012;28(2):228–31.
Zhao J, Lui H, McLean DI, Zeng H. Automated autofluorescence background subtraction algorithm for biomedical Raman spectroscopy. Appl Spectrosc. 2007;61(11):1225–32.
Freed EO. HIV-1 gag proteins: diverse functions in the virus life cycle. Virology. 1998;251(VY989398):1–15.
Mascarenhas AP, Musier-Forsyth K. The capsid protein of human immunodeficiency virus: interactions of HIV-1 capsid with host protein factors. Febs J. 2009;276(21):6118–27.
Virkler K, Lednev IK. Raman spectroscopic signature of semen and its potential application to forensic body fluid identification. Forensic Sci Int. 2009;193(1):56–62.
Li J, Du Y, Qi J, Sneha R, Chang A, Mohan C, et al. Raman spectroscopy as a diagnostic tool for monitoring acute nephritis. J Biophotonics. 2016;9(3):260–269.
Gonchukov S, Sukhinina A, Bakhmutov D, Minaeva S. Raman spectroscopy of saliva as a perspective method for periodontitis diagnostics. Laser Phys Lett. 2012;9:73–7.
Depciuch J, Kaznowska E, Zawlik I, Wojnarowska R, Cholewa M, Heraud P, et al. Application of Raman spectroscopy and infrared spectroscopy in the identification of breast cancer. Appl Spectrosc. 2016;70(2):251–63.
Tuma R. Raman spectroscopy of proteins: from peptides to large assemblies. J Raman Spectrosc. 2005;36:307–19.
Elechiguerra JL, Burt JL, Morones JR, Camacho-Bragado A, Gao X, Lara HH, et al. Interaction of silver nanoparticles with HIV-1. J Nanobiotechnol. 2005;3(1):1.
Abdi H, Williams LJ. Principal component analysis. WIREs Comput Stat. 2010;2:433–59.
Ilin A, Raiko T. Practical approaches to principal component analysis in the presence of missing values. J Mach Learn Res. 2010;11:1957–2000.
Acknowledgements
The authors would like to thank Dr. Julius O. Oyugi of Department of Medical Microbiology, University of Nairobi, for providing the samples used in the study. The first author thanks NACOSTI, Kenya, for funding his studies. We also thank ISP Uppsala University for grant KEN004.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was approved by the Ethics and Research Committee of the University of Nairobi (KNH-UoN:P637=10=2015).
Conflict of interest
The authors declare that they have no competing interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 251 kb)
Rights and permissions
About this article
Cite this article
Otange, B.O., Birech, Z., Okonda, J. et al. Conductive silver paste smeared glass substrates for label-free Raman spectroscopic detection of HIV-1 and HIV-1 p24 antigen in blood plasma. Anal Bioanal Chem 409, 3253–3259 (2017). https://doi.org/10.1007/s00216-017-0267-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-017-0267-0