Abstract
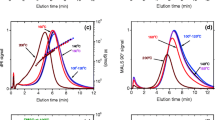
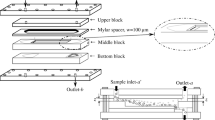
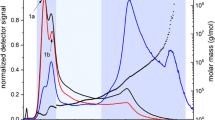
In this paper, we investigate whether dissolution in water under autoclaving conditions (140 °C, 20 min) or in dimethyl sulfoxide, DMSO (100 °C, 1 h), is preferable for characterization of amylose. Two types of amylose, potato and maize, were dissolved either in water using an autoclave or in DMSO. On the aqueous solutions obtained, the extent of molecular dissolution of the sample (referred to as the dissolution yield) was determined by enzymatic analysis as well as the molecular properties, such as molar mass and root-mean-square radius, obtained with asymmetrical flow field-flow fractionation coupled to multi-angle light scattering and differential refractive index detection (AF4-MALS-dRI). The results showed that both dissolution methods are efficient at dissolving amylose. However, AF4-MALS-dRI analysis revealed substantial differences. Amylose aqueous solutions obtained by dissolution in DMSO were relatively stable over time, but the dissolution method in autoclave caused some degradation of the molecules, and their solutions display a high tendency to retrograde.







Similar content being viewed by others
References
Nilsson GS, Gorton L, Bergquist K-E, Nilsson U (1996) Determination of the degree of branching in normal and amylopectin type potato starch with 1H-NMR spectroscopy improved resolution and two-dimensional spectroscopy. Starch-Stärke 48(10):352–357
Fernandez C, Rojas C, Nilsson L (2011) Size, structure and scaling relationships in glycogen from various sources investigated with asymmetrical flow field-flow fractionation and 1H NMR. Int J Biol Macromol 49(4):458–465
Perez-Rea D, Bergenståhl B, Nilsson L (2015) Development and evaluation of methods for starch dissolution using asymmetrical flow field-flow fractionation. Part I. Dissolution of amylopectin. Accepted for publication in Anal Bioanal Chem. doi:10.1007/s00216-015-8611-8
Rojas C, Wahlund K-G, Br B, Nilsson L (2008) Macromolecular geometries determined with field-flow fractionation and their impact on the overlap concentration. Biomacromolecules 9(6):1684–1690
Rolland-Sabaté A, Guilois S, Jaillais B, Colonna P (2011) Molecular size and mass distributions of native starches using complementary separation methods: asymmetrical flow field flow fractionation (A4F) and hydrodynamic and size exclusion chromatography (HDC-SEC). Anal Bioanal Chem 399(4):1493–1505
Aberle T, Burchard W, Vorwerg W, Radosta S (1994) Conformational contributions of amylose and amylopectin to the structural properties of starches from various sources. Starch-Stärke 46(9):329–335
Wahlund K-G, Leeman M, Santacruz S (2011) Size separations of starch of different botanical origin studied by asymmetrical-flow field-flow fractionation and multiangle light scattering. Anal Bioanal Chem 399(4):1455–1465
Hizukuri S, J-i A, Hanashiro I (2006) In: Eliasson A-C (ed) Carbohydrate in food, 2nd edn. Taylor & Francis Group, Boca Ratón
Gidley MJ, Hanashiro I, Hani NM, Hill SE, Huber A, Jane J-L, Liu Q, Morris GA, Rolland-Sabaté A, Striegel AM, Gilbert RG (2010) Reliable measurements of the size distributions of starch molecules in solution: current dilemmas and recommendations. Carbohydr Polym 79(2):255–261
Eliasson A-C, Gudmundsson M (2006) In: Eliasson A-C (ed) Carbohydrate in food, 2nd edn. Taylor & Francis Group, Boca Ratón
Colonna P, Mercier C (1985) Gelatinization and melting of maize and pea starches with normal and high-amylose genotypes. Phytochemistry 24(8):1667–1674
Vorwerg W, Radosta S, Leibnitz E (2002) Study of a preparative-scale process for the production of amylose. Carbohydr Polym 47(2):181–189
Han J-A, Lim S-T (2004) Structural changes of corn starches by heating and stirring in DMSO measured by SEC-MALLS-RI system. Carbohydr Polym 55(3):265–272
Roger P, Baud B, Colonna P (2001) Characterization of starch polysaccharides by flow field-flow fractionation–multi-angle laser light scattering–differential refractometer index. J Chromatogr A 917(1–2):179–185
van Bruijnsvoort M, Wahlund KG, Nilsson G, Kok WT (2001) Retention behaviour of amylopectins in asymmetrical flow field-flow fractionation studied by multi-angle light scattering detection. J Chromatogr A 925(1–2):171–182
You S, Stevenson SG, Izydorczyk MS, Preston KR (2002) Separation and characterization of barley starch polymers by a flow field-flow fractionation technique in combination with multiangle light scattering and differential refractive index detection. Cereal Chem 79(5):624–630
Rolland-Sabaté A, Colonna P, Mendez-Montealvo MG, Planchot V (2007) Branching features of amylopectins and glycogen determined by asymmetrical flow field flow fractionation coupled with multiangle laser light scattering. Biomacromolecules 8(8):2520–2532
Juna S, Williams PA, Davies S (2011) Determination of molecular mass distribution of amylopectin using asymmetrical flow field-flow fractionation. Carbohydr Polym 83(3):1384–1396
Fishman ML, Rodriguez L, Chau HK (1996) Molar masses and sizes of starches by high-performance size-exclusion chromatography with on-line multi-angle laser light scattering detection. J Agric Food Chem 44(10):3182–3188
Bello-Pérez LA, Roger P, Baud B, Colonna P (1998) Macromolecular features of starches determined by aqueous high-performance size exclusion chromatography. J Cereal Sci 27(3):267–278
Praznik W, Mundigler N, Kogler A, Pelzl B, Huber A, Wollendorfer M (1999) Molecular background of technological properties of selected starches. Starch-Stärke 51(6):197–211
Radosta S, Haberer M, Vorwerg W (2001) Molecular characteristics of amylose and starch in dimethyl sulfoxide. Biomacromolecules 2(3):970–978
Zhong F, Yokoyama W, Wang Q, Shoemaker CF (2006) Rice starch, amylopectin, and amylose: molecular weight and solubility in dimethyl sulfoxide-based solvents. J Agric Food Chem 54(6):2320–2326
Roger P, Colonna P (1993) Evidence of the presence of large aggregates contaminating amylose solutions. Carbohydr Polym 21(2–3):83–89
Roger P, Colonna P (1996) Molecular weight distribution of amylose fractions obtained by aqueous leaching of corn starch. Int J Biol Macromol 19(1):51–61
Cave RA, Seabrook SA, Gidley MJ, Gilbert RG (2009) Characterization of starch by size-exclusion chromatography: the limitations imposed by shear scission. Biomacromolecules 10(8):2245–2253
Kim W, Eum CH, Lim S, Han J, You S, Lee S (2007) Separation of amylose and amylopectin in corn starch using dual-programmed flow field-flow fractionation. Bull Korean Chem Soc 28(12):2489
Messaud FA, Sanderson RD, Runyon JR, Otte T, Pasch H, Williams SKR (2009) An overview on field-flow fractionation techniques and their applications in the separation and characterization of polymers. Prog Polym Sci 34(4):351–368
Nilsson L (2012) In: Williams K, Caldwell K (eds) Field-flow fractionation in biopolymer analysis. Springer-Verlag/Wien, NewYork
Lee S, Nilsson P-O, Nilsson GS, Wahlund K-G (2003) Development of asymmetrical flow field-flow fractionation–multi angle laser light scattering analysis for molecular mass characterization of cationic potato amylopectin. J Chromatogr A 1011(1–2):111–123
Nilsson L (2013) Separation and characterization of food macromolecules using field-flow fractionation: A review. Food Hydrocoll 30(1):1–11
Håkansson A, Magnusson E, Bergenståhl B, Nilsson L (2012) Hydrodynamic radius determination with asymmetrical flow field-flow fractionation using decaying cross-flows. Part I. A theoretical approach. J Chromatogr A 1253:120–126
Leeman M, Wahlund K-G, Wittgren B (2006) Programmed cross flow asymmetrical flow field-flow fractionation for the size separation of pullulans and hydroxypropyl cellulose. J Chromatogr A 1134(1–2):236–245
Berry GC (1966) Thermodynamic and conformational properties of polystyrene. I. Light-scattering studies on dilute solutions of linear polystyrenes. J Chem Phys 44(12):4550–4564
Andersson M, Wittgren B, Wahlund K-G (2003) Accuracy in multiangle light scattering measurements for molar mass and radius estimations. Model calculations and experiments. Anal Chem 75(16):4279–4291
Brandrup J, Immergut EH, Grulke EA (eds) (1999) Polymer handbook, 4th edn. Wiley, New York
Nilsson L, Leeman M, Wahlund K-G, Bergenståhl B (2006) Mechanical degradation and changes in conformation of hydrophobically modified starch. Biomacromolecules 7(9):2671–2679
Takeda Y, Hizukuri S, Juliano BO (1986) Purification and structure of amylose from rice starch. Carbohydr Res 148(2):299–308
Roger P, Colonna P (1992) The influence of chain length on the hydrodynamic behaviour of amylose. Carbohydr Res 227:73–83
Vilaplana F, Gilbert RG (2010) Two-dimensional size/branch length distributions of a branched polymer. Macromolecules 43(17):7321–7329
Gidley MJ, Bulpin PV (1989) Aggregation of amylose in aqueous systems: the effect of chain length on phase behavior and aggregation kinetics. Macromolecules 22(1):341–346
Gidley MJ, Bulpin PV (1987) Crystallisation of malto-oligosaccharides as models of the crystalline forms of starch: minimum chain-length requirement for the formation of double helices. Carbohydr Res 161(2):291–300
Hizukuri S, Takeda Y, Yasuda M, Suzuki A (1981) Multi-branched nature of amylose and the action of debranching enzymes. Carbohydr Res 94(2):205–213
Caldwell K (2000) In: Schimpf M, Caldwell K, Giddings JC (eds) Field-flow fractionation handbook. Wiley, New York
Sievert D, Wüsch P (1993) Amylose chain association based on differential scanning calorimetry. J Food Sci 58(6):1332–1335
Jackson DS (1991) Solubility behavior of granular corn starches in methyl sulfoxide (DMSO) as measured by high performance size exclusion chromatography. Starch-Stärke 43(11):422–427
Acknowledgments
Financial support from the Swedish International Development Agency (Sida/SAREC) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in the topical collection Field- and Flow-based Separations with guest editors Gaetane Lespes, Catia Contado, and Bruce Gale.
Rights and permissions
About this article
Cite this article
Perez-Rea, D., Bergenståhl, B. & Nilsson, L. Development and evaluation of methods for starch dissolution using asymmetrical flow field-flow fractionation. Part II: Dissolution of amylose. Anal Bioanal Chem 408, 1399–1412 (2016). https://doi.org/10.1007/s00216-015-8894-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-015-8894-9