Abstract
A simple and highly reproducible toxicity assay method was studied by employing 2,6-dichlorophenolindophenol (DCIP) as a redox color indicator, baker’s yeast Saccharomyces cerevisiae, and a thermostable three-consecutive-stir unit. The absorbance of DCIP was decreased by increasing the metabolism activity of S. cerevisiae to intake glucose as an organic substance. By optimizing the measurement conditions, we obtained highly sensitive responses to glucose between 0.75 and 30 mg/L (eight points, n = 3) with an incubation time of the reaction mixture of 10 min at 30 °C. An excellent value of 1.15% was obtained as the average of the repeatability from eight points. Next, for the characterization of this method, we investigated the influence on the colorimetric response of dissolved substances, such as inorganic ions and surfactants, in natural water. Furthermore, the colorimetric responses to several toxicants were examined using Cu2+, Mn2+, Zn2+, Cr3+, and Fe3+ as heavy-metal ions and simazine as an agricultural chemical. As a result, notable colorimetric responses were obtained for Cu2+ and Mn2+ at several concentrations, and the results were compared with those obtained using river water as a real sample. In the stability test, responses to 30 mg/L glucose were obtained for 28 days when the yeast cell suspension was stored at 4 °C (response reduction, 43.9%; average of the relative standard deviation for nine testing days, 22.7%; average of repeatability, 1.01%).

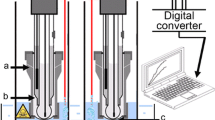
Graphical image of the colorimetric toxicity assay





Similar content being viewed by others
References
Nakamura H, Karube I (2003) Anal Bioanal Chem 377:446–468
Nakamura H, Karube I (2005) In: Grimes CA et al (eds) Encyclopedia of sensors, vol 6. American Scientific, New York, pp 87–126
Lelie D, Corbisiera P, Baeyensa W, Wuertza S, Dielsa L, Mergeaya M (1994) Res Microbiol 145:67–74
Lazarova V, Manem J (1995) Water Res 29:2227–2245
Bousse L (1996) Sens Actuators B 34:270–275
Blaise C (1998) Ecotoxicol Environ Saf 40:115–119
D’Souza SF (2001) Biosens Bioelectron 16:337–353
Belkin S (2003) Curr Opin Microbiol 6:206–212
Baronian KHR (2004) Biosens Bioelectron 19:953–962
Karube I, Mitsuda S, Matsunaga T, Suzuki S (1977) J Ferment Technol 55:243–248
Kong Z, Vanrolleghem PA, Verstraete W (1993) Biosens Bioelectron 8:49–58
Nakanishi K, Ikebukuro K, Karube I (1996) Appl Biochem Biotechnol 60:97–106
Tizzard A, Webber J, Gooneratne R, John R, Hay J, Pasco N (2004) Anal Chim Acta 522:197–205
Bentley A, Atkinson A, Jezek J, Rawson DM (2001) Toxicol In Vitro 15:469–475
Steinberg SM, Poziomek EJ, Engelmann WH, Rogers KR (1995) Chemosphere 30:2155–2197
Bulich AA (1979) In: Markings LL, Kimerle RA (eds) Aquatic toxicology.. American Society for Testing and Materials, West Conshohocken, pp 98–106
Nakamura H, Suzuki M (2007) Anal Bioanal Chem (in press)
Nakamura H, Suzuki M (2007) Anal Bioanal Chem (in press)
Yoshida N, McNiven SJ, Morita T, Nakamura H, Karube I (2002) Anal Lett 35:1541–1549
Nakamura H, Kobayashi S, Hirata Y, Suzuki K, Mogi Y, Karube I (2007) Anal Biochem (in press)
Nakamura H, Suzuki K, Ishikuro H, Kinoshita S, Koizumi R, Okuma S, Gotoh M, Karube I (2007) Talanta 72:210–216
Collins YE, Stotzky G (1989) In: Beveridge TJ, Doyle RJ (eds) Metal ions and bacteria. Wiley, New York, pp 31–90
Acknowledgements
The authors acknowledge Y. Abe, T. Ohmomo, M. Yataka, S. Okuma, R. Koizumi, A. Yoshimura, and K. Sudo for assistance with the experiments.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Nakamura, H., Hirata, Y., Mogi, Y. et al. A simple and highly repeatable colorimetric toxicity assay method using 2,6-dichlorophenolindophenol as the redox color indicator and whole eukaryote cells. Anal Bioanal Chem 389, 835–840 (2007). https://doi.org/10.1007/s00216-007-1527-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-007-1527-1