Abstract
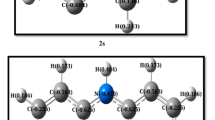
A theoretical study of singlet- and triplet-state cyclic vinylidenes was performed using the B3LYP, B3PW91, CCSD, CCSD(T), QCISD, and QCISD(T) methods at the 6-311G(d), 6-311++G(d,p), 6-311++G(df,pd), 6-311++G(2df,2pd), and 6-311++G(3df,3pd) basis sets. Fifty-eight vinylidenes, featuring the N-heterocyclic carbenic (NHC) unit, were chosen as the model molecules for this study. The computations demonstrate that fifty-one cyclic vinylidenes are predicted to have a triplet ground state with a singlet and triplet splitting energy of approximately −0.11 to −20 and −1.8 to −21 kcal/mol, for the B3LYP/6-311++G(d,p) and B3PW91/6-311++G(df,pd) levels of theory. Our theoretical findings suggest that it is the π-electron-rich ability of the NHC ring that makes cyclic vinylidene preferably adopt the triplet ground state. A valence electron model was used to explain the computational results. The theoretical observations strongly suggest that besides the traditional fact that the carbenic center is attached by two sterically bulky substituents, the use of an NHC skeleton can significantly increase the probability of producing a vinylidene that has a triplet ground state.






Similar content being viewed by others
Notes
All computations were done using the Gaussian 03 package. For details, see the Supporting Information.
We thank one reviewer for this suggestion.
For instance, in the case of NHCHCHNH)C=C:, using the NBO analysis, one can obtain two π C=C → σ NC* interactions, each having 33.5 kcal/mol stabilizing energy for the triplet state. Also, there are two σ NC → π C=C* interactions, each stabilizing the triplet state by 14.2 kcal/mol. For details, see http://www.chem.wisc.edu/~nbo5.
References
Moss RA (1980) Acc Chem Res 13:58
Moss RA (1989) Acc Chem Res 22:15
Moss RA (2002) In: Bertrand G (ed) Carbene chemistry. Marcel Dekker, New York, pp 57–101
Hoffmann R, Zeiss GD, Van Dine GW (1968) J Am Chem Soc 90:1485
Baird NC, Taylor KF (1978) J Am Chem Soc 100:1333
Arduengo AJ III, Harlow RL, Kline M (1991) J Am Chem Soc 113:361
Arduengo AJ III (1999) Acc Chem Res 32:913
(2007) Coord Chem Rev 251:595–895
(2009) Chem Rev 109(8)
(2009) Eur J Inorg Chem (13)
(2009) Dalton Trans (35)
Melaimi M, Soleihavoup M, Bertrand G (2010) Angew Chem Int Ed 49:8810
Tomioka H (1997) Acc Chem Res 30:315
Tomioka H (1998) In: Brinker U (ed) Advances in carbene chemistry, vol 2. JAI Press, Greenwich, CT, pp 175–214
Tomioka H (2002) In: Bertrand G (ed) Carbene chemistry. Fontis Media S. A, Lausanne, pp 103–152
Kirmse W (2003) Angew Chem Int Ed 42:2117
Hahn FE, Jahnke MC (2008) Angew Chem Int Ed 47:3122
Hirai K, Itoh T, Tomioka H (2009) Chem Rev 109:3275
Tomioka H, Hattori M, Hirai K, Murata S (1996) J Am Chem Soc 118:8723
Hirai K, Tomioka H (1999) J Am Chem Soc 121:10213
Hu Y, Hirai K, Tomioka H (1999) J Phys Chem A 103:9280
Itakura H, Mizuno H, Hirai K, Tomioka H (2000) J Org Chem 65:8797
Tomioka H, Iwamoto E, Itakura H, Hirai K (2001) Nature 412:626
Kawano M, Hirai K, Tomioka H, Ohashi Y (2001) J Am Chem Soc 123:6904
Iiba E, Hirai K, Tomioka H, Yoshioka Y (2002) J Am Chem Soc 124:14308
Iwamoto E, Hirai K, Tomioka H (2003) J Am Chem Soc 125:14664
Iwamoto E, Hirai K, Tomioka H (2003) J Am Chem Soc 125:14664
Itoh T, Takada A, Hirai K, Tomioka H (2005) Org Lett 7:811
Su M-D, Chu S-Y (1999) Chem Phys Lett 308:283
Su M-D, Chu S-Y (2000) Chem Phys Lett 320:475
Woodcock HL, Moran D, Schleyer PvR, Schaefer HF III (2001) J Am Chem Soc 123:4331
Trindle C (2003) J Org Chem 68:9669
Trindle C (2005) J Phys Chem A 109:898
Woodcock HL, Moran D, Brooks BR, Schleyer PvR, Schaefer HF III (2007) J Am Chem Soc 129:3763
Gu S-Y, Su M-D (2007) Chem Phys Lett 443:211
Díez-González S, Marion N, Nolan SP (2009) Chem Rev 109:3612
Enders D, Niemeier O, Henseler A (2007) Chem Rev 107:5606
Poyatos M, Mata JA, Peris E (2009) Chem Rev 109:3677
de Frémont P, Marion N, Nolan SP (2009) Coord Chem Rev 253:862 (and related references therein)
Frisch MJ et al (2003) Gaussian 03, revision C 02. Gaussian, Inc., Pittsburgh, PA
Albright TA, Burdett JK, Whangbo MH (1985) In: Orbital interaction in chemistry, chap 2. Wiley, New York
Davis JH, Goddard WA III, Harding LB (1977) J Am Chem Soc 99:2919
Dykstra CE, Schaefer HF III (1978) J Am Chem Soc 100:1378
Kenney JW, Simons J, Purvis GD, Bartlett RJ (1978) J Am Chem Soc 100:6930
Ervin KM, Ho J, Lineberger WC (1989) J Chem Phys 91:5974
Callo MM, Hamilton TP, Schaefer HF III (1990) J Am Chem Soc 112:8714
Gilles MK, Lineberger WC, Ervin KM (1031) J Am Chem Soc 1993:115
DeLeeuw BJ, Fermann JT, Xie Y, Schaefer HF III (1039) J Am Chem Soc 1993:115
Gunion RF, Lineberger WC (1996) J Phys Chem 100:4395
Robinson MS, Polak ML, Bierbaum VM, DePuy CH, Lineberger WC (1995) J Am Chem Soc 117:6766
Gunion RF, Koppel H, Leach GW, Lineberger WC (1995) J Chem Phys 103:1250
Worthington SE, Cramer CJ (1997) J Phys Org Chem 10:755
Reed AE, Curtiss LA, Weinhold F (1998) Chem Rev 88:899
Acknowledgments
The authors are grateful to the National Center for High-Performance Computing of Taiwan for generous amounts of computing time. They also thank the National Science Council of Taiwan for the financial support. We express our gratitude to the referees for their valuable comments.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Su, MD., Chuang, CC. Theory predicts triplet ground-state carbene containing the N-heterocyclic carbenic unit. Theor Chem Acc 132, 1360 (2013). https://doi.org/10.1007/s00214-013-1360-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-013-1360-x