Abstract
Rationale
Cannabidiol (CBD) reduces craving in animal models of alcohol and cocaine use and is known to modulate nicotinic receptor function, suggesting that it may alleviate symptoms of nicotine withdrawal. However, preclinical evaluation of its efficacy is still lacking.
Objectives
The goal of this study was to test the preclinical efficacy of a chronic CBD treatment in reducing nicotine dependence using measures of withdrawal symptoms including somatic signs, hyperalgesia, and weight gain during acute and protracted abstinence.
Methods
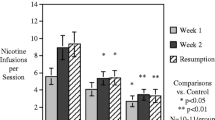
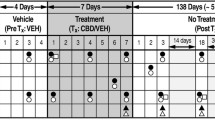
Male and female Wistar rats were made dependent on nicotine using osmotic minipumps (3.15 mg/kg/day) for 2 weeks, after which minipumps were removed to induce spontaneous withdrawal. Three groups received CBD injections at doses of 7.5, 15, and 30 mg/kg/day for 2 weeks, starting 1 week into chronic nicotine infusion. The control groups included rats with nicotine minipumps that received vehicle injections of sesame oil instead of CBD; rats implanted with saline minipumps received sesame oil injections (double vehicle) or the highest dose of CBD 30 mg/kg/day. Throughout the experiment, serum was collected for determination of CBD and nicotine concentrations, mechanical sensitivity threshold and withdrawal scores were measured, and body weight was recorded.
Results
CBD prevented rats from exhibiting somatic signs of withdrawal and hyperalgesia during acute and protracted abstinence. There was no dose–response observed for CBD, suggesting a ceiling effect at the doses used and the potential for lower effective doses of CBD. The saline minipump group did not show either somatic signs of withdrawal or hyperalgesia during acute and protracted abstinence, and the highest dose of CBD used (30 mg/kg/day) did not alter these results.
Conclusions
This preclinical study suggests that using CBD as a strategy to alleviate the withdrawal symptoms upon nicotine cessation may be beneficial.





Similar content being viewed by others
References
(2018) Cannabidiol (Epidiolex) for epilepsy. Med Lett Drugs Ther 60(1559): 182–184
Alvarez FJ, Lafuente H, Rey-Santano MC, Mielgo VE, Gastiasoro E, Rueda M, Pertwee RG, Castillo AI, Romero J, Martinez-Orgado J (2008) Neuroprotective effects of the nonpsychoactive cannabinoid cannabidiol in hypoxic-ischemic newborn piglets. Pediatr Res 64(6):653–658
Anderson GD, Chan LN (2016) Pharmacokinetic drug interactions with tobacco, cannabinoids and smoking cessation products. Clin Pharmacokinet 55(11):1353–1368
Bagdas D, Alkhlaif Y, Jackson A, Carroll FI, Ditre JW, Damaj MI (2018) New insights on the effects of varenicline on nicotine reward, withdrawal and hyperalgesia in mice. Neuropharmacology 138:72–79
Carstens E, Anderson KA, Simons CT, Carstens MI, Jinks SL (2001) Analgesia induced by chronic nicotine infusion in rats: differences by gender and pain test. Psychopharmacology 157(1):40–45
Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL (1994) Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 53(1):55–63
Cippitelli A, Astarita G, Duranti A, Caprioli G, Ubaldi M, Stopponi S, Kallupi M, Sagratini G, Rodriguez de Fonseca F, Piomelli D, Ciccocioppo R (2011) Endocannabinoid regulation of acute and protracted nicotine withdrawal: effect of FAAH inhibition. PLoS One 6(11):e28142
Cohen C, Pickworth WB, Henningfield JE (1991) Cigarette smoking and addiction. Clin Chest Med 12(4):701–710
Cohen A, Treweek J, Edwards S, Leao RM, Schulteis G, Koob GF, George O (2015) Extended access to nicotine leads to a CRF1 receptor dependent increase in anxiety-like behavior and hyperalgesia in rats. Addict Biol 20(1):56–68
Cryan JF, Bruijnzeel AW, Skjei KL, Markou A (2003) Bupropion enhances brain reward function and reverses the affective and somatic aspects of nicotine withdrawal in the rat. Psychopharmacology 168(3):347–358
Damaj MI, Kao W, Martin BR (2003) Characterization of spontaneous and precipitated nicotine withdrawal in the mouse. J Pharmacol Exp Ther 307(2):526–534
Damaj MI, Grabus SD, Navarro HA, Vann RE, Warner JA, King LS, Wiley JL, Blough BE, Lukas RJ, Carroll FI (2010) Effects of hydroxymetabolites of bupropion on nicotine dependence behavior in mice. J Pharmacol Exp Ther 334(3):1087–1095
Devinsky O, Cilio MR, Cross H, Fernandez-Ruiz J, French J, Hill C, Katz R, Di Marzo V, Jutras-Aswad D, Notcutt WG, Martinez-Orgado J, Robson PJ, Rohrback BG, Thiele E, Whalley B, Friedman D (2014) Cannabidiol: pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia 55(6):791–802
Gonzalez S, Cebeira M, Fernandez-Ruiz J (2005) Cannabinoid tolerance and dependence: a review of studies in laboratory animals. Pharmacol Biochem Behav 81(2):300–318
Gonzalez-Cuevas G, Martin-Fardon R, Kerr TM, Stouffer DG, Parsons LH, Hammell DC, Banks SL, Stinchcomb AL, Weiss F (2018) Unique treatment potential of cannabidiol for the prevention of relapse to drug use: preclinical proof of principle. Neuropsychopharmacology 43(10):2036–2045
Gourlay SG, Benowitz NL (1997) Arteriovenous differences in plasma concentration of nicotine and catecholamines and related cardiovascular effects after smoking, nicotine nasal spray, and intravenous nicotine. Clin Pharmacol Ther 62(4):453–463
Hamouda AK, Jackson A, Bagdas D, Imad Damaj M (2018) Reversal of nicotine withdrawal signs through positive allosteric modulation of alpha4beta2 nicotinic acetylcholine receptors in male mice. Nicotine Tob Res 20(7):903–907
Hiler M, Breland A, Spindle T, Maloney S, Lipato T, Karaoghlanian N, Shihadeh A, Lopez A, Ramoa C, Eissenberg T (2017) Electronic cigarette user plasma nicotine concentration, puff topography, heart rate, and subjective effects: influence of liquid nicotine concentration and user experience. Exp Clin Psychopharmacol 25(5):380–392
Hindocha C, Freeman TP, Grabski M, Crudgington H, Davies AC, Stroud JB, Das RK, Lawn W, Morgan CJA, Curran HV (2018a) The effects of cannabidiol on impulsivity and memory during abstinence in cigarette dependent smokers. Sci Rep 8(1):7568
Hindocha C, Freeman TP, Grabski M, Stroud JB, Crudgington H, Davies AC, Das RK, Lawn W, Morgan, CJA, Curran HV (2018b) Cannabidiol reverses attentional bias to cigarette cues in a human experimental model of tobacco withdrawal. Addiction 113(9):1696–1705
Hubbard JA, Smith BE, Sobolesky PM, Kim S, Hoffman MA, Stone J, Huestis MA, Grelotti DJ, Grant I, Marcotte TD, Fitzgerald RL (2020) Validation of a liquid chromatography tandem mass spectrometry (LC-MS/MS) method to detect cannabinoids in whole blood and breath. Clin Chem Lab Med 58(5):673–681
Igari M, Alexander JC, Ji Y, Qi X, Papke RL, Bruijnzeel AW (2014) Varenicline and cytisine diminish the dysphoric-like state associated with spontaneous nicotine withdrawal in rats. Neuropsychopharmacology 39(2):455–465
Jackson A, Silk S, Buhidma Y, Shoaib M (2017) Varenicline, the clinically effective smoking cessation agent, restores probabilistic response reversal performance during withdrawal from nicotine. Addict Biol 22(5):1316–1328
Kathmann M, Flau K, Redmer A, Trankle C, Schlicker E (2006) Cannabidiol is an allosteric modulator at mu- and delta-opioid receptors. Naunyn Schmiedebergs Arch Pharmacol 372(5):354–361
Khan SZ, Farooq A, Masood M, Shahid A, Khan IU, Nisar H, Fatima I (2020) Smokeless tobacco use and risk of oral cavity cancer. Turk J Med Sci 50(1):291–297
Kallupi M, Xue S, Zhou B, Janda KD, George O (2018) An enzymatic approach reverses nicotine dependence, decreases compulsive-like intake, and prevents relapse. Sci Adv 4(10):eeat4751
Kallupi M, de Guglielmo G, Larrosa E, George O (2019) Exposure to passive nicotine vapor in male adolescent rats produces a withdrawal-like state and facilitates nicotine self-administration during adulthood. Eur Neuropsychopharmacology 29(11):1227–1234
Larsen C, Shahinas J (2020) Dosage, efficacy and safety of cannabidiol administration in adults: a systematic review of human trials. J Clin Med Res 12(3):129–141
Leischow SJ, Stitzer ML (1991) Smoking cessation and weight gain. Br J Addict 86(5):577–581
Liput DJ, Hammell DC, Stinchcomb AL, Nixon K (2013) Transdermal delivery of cannabidiol attenuates binge alcohol-induced neurodegeneration in a rodent model of an alcohol use disorder. Pharmacol Biochem Behav 111:120–127
Mahgoub M, Keun-Hang SY, Sydorenko V, Ashoor A, Kabbani N, Al Kury L, Sadek B, Howarth CF, Isaev D, Galadari S, Oz M (2013) Effects of cannabidiol on the function of alpha7-nicotinic acetylcholine receptors. Eur J Pharmacol 720(1–3):310–319
Malin DH, Goyarzu P (2009) Rodent models of nicotine withdrawal syndrome. Handb Exp Pharmacol (192): 401–434
Malin DH, Lake JR, Newlin-Maultsby P, Roberts LK, Lanier JG, Carter VA, Cunningham JS, Wilson OB (1992) Rodent model of nicotine abstinence syndrome. Pharmacol Biochem Behav 43(3):779–784
Malin DH, Lake JR, Smith TD, Khambati HN, Meyers-Paal RL, Montellano AL, Jennings RE, Erwin DS, Presley SE, Perales BA (2006) Bupropion attenuates nicotine abstinence syndrome in the rat. Psychopharmacology 184(3–4):494–503
Markou A, Paterson NE (2001) The nicotinic antagonist methyllycaconitine has differential effects on nicotine self-administration and nicotine withdrawal in the rat. Nicotine Tob Res 3(4):361–373
McLaughlin I, Dani JA, De Biasi M (2015) Nicotine withdrawal. Curr Top Behav Neurosci 24:99–123
Mooney ME, Sofuoglu M (2006) Bupropion for the treatment of nicotine withdrawal and craving. Expert Rev Neurother 6(7):965–981
Morgan CJ, Das RK, Joye A, Curran HV, Kamboj SK (2013) Cannabidiol reduces cigarette consumption in tobacco smokers: preliminary findings. Addict Behav 38(9):2433–2436
Pahng AR, Edwards S (2018) Measuring pain avoidance-like behavior in drug-dependent rats. Curr Protoc Neurosci 85(1):e53
Perkins KA (1993) Weight gain following smoking cessation. J Consult Clin Psychol 61(5):768–777
Prochaska JJ, Benowitz NL (2016) The past, present, and future of nicotine addiction therapy. Annu Rev Med 67:467–486
Russell MA, Feyerabend C, Cole PV (1976) Plasma nicotine levels after cigarette smoking and chewing nicotine gum. Br Med J 1(6017):1043–1046
Russo EB, Burnett A, Hall B, Parker KK (2005) Agonistic properties of cannabidiol at 5-HT1a receptors. Neurochem Res 30(8):1037–1043
Shoaib M, Stolerman IP (1999) Plasma nicotine and cotinine levels following intravenous nicotine self-administration in rats. Psychopharmacology 143(3):318–321
Sloan ME, Gowin JL, Ramchandani VA, Hurd YL, Le Foll B (2017) The endocannabinoid system as a target for addiction treatment: trials and tribulations. Neuropharmacology 124:73–83
Sobolesky PM, Smith BE, Hubbard JA, Stone J, Marcotte TD, Grelotti DJ, Grant I, Fitzgerald RL (2019) Validation of a liquid chromatography-tandem mass spectrometry method for analyzing cannabinoids in oral fluid. Clin Chim Acta 491:30–38
Stitzer ML, Gross J (1988) Smoking relapse: the role of pharmacological and behavioral factors. Prog Clin Biol Res 261:163–184
Stout SM, Cimino NM (2014) Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: a systematic review. Drug Metab Rev 46(1):86–95
Thomas A, Baillie GL, Phillips AM, Razdan RK, Ross RA, Pertwee RG (2007) Cannabidiol displays unexpectedly high potency as an antagonist of CB1 and CB2 receptor agonists in vitro. Br J Pharmacol 150(5):613–623
Viudez-Martinez A, Garcia-Gutierrez MS, Navarron CM, Morales-Calero MI, Navarrete F, Torres-Suarez AI, Manzanares J (2018) Cannabidiol reduces ethanol consumption, motivation and relapse in mice. Addict Biol 23(1):154–164
Viudez-Martinez A, Garcia-Gutierrez MS, Medrano-Relinque J, Navarron CM, Navarrete F, Manzanares J (2019) Cannabidiol does not display drug abuse potential in mice behavior. Acta Pharmacol Sin 40(3):358–364
Warren GW, Alberg AJ, Kraft AS, Cummings KM (2014) The 2014 Surgeon General’s report: “The health consequences of smoking–50 years of progress”: a paradigm shift in cancer care. Cancer 120(13):1914–1916
West R, Baker CL, Cappelleri JC, Bushmakin AG (2008) Effect of varenicline and bupropion SR on craving, nicotine withdrawal symptoms, and rewarding effects of smoking during a quit attempt. Psychopharmacology 197(3):371–377
Xue S, Kallupi M, Zhou B, Smith LC, Miranda PO, George O, Janda KD (2018) An enzymatic advance in nicotine cessation therapy. Chem Commun (Camb) 54(14):1686–1689
Zuardi AW (2008) Cannabidiol: from an inactive cannabinoid to a drug with wide spectrum of action. Braz J Psychiatry 30(3):271–280
Acknowledgements
The authors wish to express their thanks to Molly Brennan, Sharona Sedighim, Hassiba Beldjoud, and Nathan Velarde for providing help with the study. The authors aknowledge the Preclinical Addiction Research Consortium at UCSD for supporting this project.
Funding
This work was supported by the National Institute on Drug Abuse (grant no. 1F31DA047113-01 to L.C.S.), National Institute on Alcohol Abuse and Alcoholism (grant no. AA022977 and AA006420 to O.G.), Tobacco-Related Disease Research Program (grant no. 27IR-0047 to O.G.), Tobacco-Related Disease Research Program (grant no. T31KT1859 to M.K.), and CV science (O.G.).
Author information
Authors and Affiliations
Contributions
MK, OG, KH, and JD designed the experiment; LCS and LT performed the behavioral experiments, and nicotine and cotinine blood extraction. BB and LLGC performed the behavioral experiments, blood withdrawal, and data analysis. LCS and MK analyzed the data and prepared the manuscript and figures. YS performed LCMS analysis of nicotine and cotinine serum levels. JDM supervised and interpreted nicotine and cotinine serum level determinations; RS, MH, and RF developed the isotope dilution LC/MS/MS assay for quantifying CBD in blood specimens; MK and OG edited the manuscript and figures. All authors approve the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Joseph Dowling and Kate Hanham are CEO and Acting Sr. VP of Development of CV Sciences, Inc.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Smith, L.C., Tieu, L., Suhandynata, R.T. et al. Cannabidiol reduces withdrawal symptoms in nicotine-dependent rats. Psychopharmacology 238, 2201–2211 (2021). https://doi.org/10.1007/s00213-021-05845-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-021-05845-4