Abstract
Rationale
The synthetic phenethylamines are recreational drugs known to produce psychostimulant effects. However, their abuse potential has not been widely studied.
Objectives
Here, we investigated the rewarding and the hallucinatory effects of 2,5-dimetoxy-4-bromo-amphetamine hydrobromide (DOB) and para-methoxyamphetamine (PMA) in comparison with the classical 3,4-methylenedioxymethamphetamine (MDMA). In addition, the role of serotonin 5-HT2-like receptor on the abovementioned effects was evaluated.
Methods
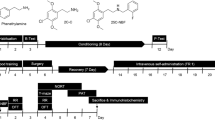
Zebrafish were intramuscularly (i.m.) treated with a wide range of doses of DOB (0.1–20 mg/kg), PMA (0.0005–2 mg/kg), or MDMA (0.5–160 mg/kg). Animals were submitted to a conditioned place preference (CPP) task, to investigation of the rewarding properties, and to the evaluation of hallucinatory behavior in terms of appearance of a trance-like behavior. The serotonin 5-HT2 subtype receptor antagonist ritanserin (0.025–2.5 mg/kg) in association with the maximal effective dose of MDMA, DOB, and PMA was given i.m., and the effect on CPP or hallucinatory behavior was evaluated.
Results
MDMA and its derivatives exhibited CPP in a biphasic fashion, being PMA the most potent. This effect was accompanied, for DOB (2 mg/kg) and PMA (0.1 mg/kg), by a trance-like hallucinatory behavior. MDMA at a high dose as 160 mg/kg did not induce any hallucinatory behavior. Ritanserin significantly blocked the rewarding and hallucinatory effects suggesting the involvement of serotonin 5HT2 subtype receptor.
Conclusion
Collectively, these findings demonstrate for the first time that the rewarding properties of DOB and PMA are accompanied by hallucinatory behavior through a serotonergic system and reinforce zebrafish as an emerging experimental model for screening new hallucinogens.





Similar content being viewed by others
References
Aguilar MA, Rodríguez-Arias M, Miñarro J (2009) Neurobiological mechanisms of the reinstatement of drug-conditioned place preference. Brain Res Rev 59:253–277
Araújo AM, Carvalho F, Bastos Mde L, Guedes de Pinho P, Carvalho M (2015) The hallucinogenic world of tryptamines: an updated review. Arch Toxicol 89:1151–1173
Balíková M (2005) Nonfatal and fatal DOB (2,5-dimethoxy-4-bromoamphetamine) overdose. Forensic Sci Int 153:85–91
Baylen CA, Rosenberg H (2006) A review of the acute subjective effects of MDMA/ecstasy. Addiction 101:933–947
Benneyworth MA, Smith RL, Barrett RJ, Sanders-Bush E (2005) Complex discriminative stimulus properties of (+)lysergic acid diethylamide (LSD) in C57Bl/6J mice. Psychopharmacology (Berl) 179:854–862
Bilsky EJ, Montegut MJ, Nichols ML, Reid LD (1998) CGS 10746B, a novel dopamine release inhibitor, blocks the establishment of cocaine and MDMA conditioned place preferences. Pharmacol Biochem Behav 59:215–220
Boehmler W, Obrecht-Pflumio S, Canfield V, Thisse C, Thisse B, Levenson R (2004) Evolution and expression of D2 and D3 dopamine receptor genes in zebrafish. Dev Dyn 230:481–493
Braida D, Limonta V, Pegorini S, Zani A, Guerini-Rocco C, Gori E, Sala M (2007) Hallucinatory and rewarding effect of salvinorin A in zebrafish: kappa-opioid and CB1-cannabinoid receptor involvement. Psychopharmacology (Berl) 190:441–448
Braida D, Donzelli A, Martucci R, Capurro V, Busnelli M, Chini B, Sala M (2012) Neurohypophyseal hormones manipulation modulate social and anxiety-related behavior in zebrafish. Psychopharmacology (Berl) 220:319–323
Brennan KA, Carati C, Lea RA, Fitzmaurice PS, Schenk S (2009) Effect of D1-like and D2-like receptor antagonists on methamphetamine and 3,4-methylenedioxymethamphetamine self-administration in rats. Behav Pharmacol 20:688–694
Cadoni C, Solinas M, Pisanu A, Zernig G, Acquas E, Di Chiara G (2005) Effect of 3,4-methylendioxymethamphetamine (MDMA, “ecstasy”) on dopamine transmission in the nucleus accumbens shell and core. Brain Res 1055:143–148
Cheng KC, Xin X, Clark DP, La Riviere P (2011) Whole-animal imaging, gene function, and the Zebrafish Phenome Project. Curr Opin Genet Dev 21:620–629
Collier AD, Khan KM, Caramillo EM, Mohn RS, Echevarria DJ (2014) Zebrafish and conditioned place preference: a translational model of drug reward. Prog Neuropsychopharmacol Biol Psychiatry 55:16–25
Cunningham CL, Noble D (1992) Methamphetamine-induced conditioned place preference or aversion depending on dose and presence of drug. Ann NY Acad Sci 654:431–633
Daniela E, Brennan K, Gittings D, Hely L, Schenk S (2004) Effect of SCH 23390 on (+/−)-3,4-methylenedioxymethamphetamine hyperactivity and self-administration in rats. Pharmacol Biochem Behav 77:745–750
Daws LC, Irvine RJ, Callaghan PD, Toop NP, White JM, Bochner F (2000) Differential behavioural and neurochemical effects of para-methoxy-amphetamine and 3,4-methylenedioxy methamphetamine in the rat. Prog Neuropsychopharmacol Biol Psychiatry 24:955–977
Daza-Losada M, Ribeiro Do Couto B, Manzanedo C, Aguilar MA, Rodríguez-Arias M, Miñarro J (2007) Rewarding effects and reinstatement of MDMA-induced CPP in adolescent mice. Neuropsychopharmacology 32:1750–1759
de la Torre R, Farré M, Roset PN, Pizarro N, Abanades S, Segura M, Segura J, Camí J (2004) Human pharmacology of MDMA: pharmacokinetics, metabolism, and disposition. Ther Drug Monit 26:137–144
Diller AJ, Rocha A, Cardon AL, Valles R, Wellman PJ, Nation JR (2007) The effects of concurrent administration of +/−3,4-methylenedioxymethamphetamine and cocaine on conditioned place preference in the adult male rat. Pharmacol Biochem Behav 88:165–170
Dumont GJ, Verkes RJ (2006) A review of acute effects of 3,4-methylenedioxymethamphetamine in healthy volunteers. J Psychopharmacol 20:176–187
Escobedo I, O’Shea E, Orio L, Sanchez V, Segura M, de la Torre R, Farre M, Green AR, Colado MI (2005) A comparative study on the acute and long-term effects of MDMA and 3,4-dihydroxymethamphetamine (HHMA) on brain monoamine levels after i.p. or striatal administration in mice. Br J Pharmacol 144:231–241
European Monitoring Centre for Drugs and Drug Addiction (2003) Report on the risk assessment of PMMA in the framework of the joint action on new synthetic drugs. Office for Official Publications of the European Communities, Luxembourg
Fantegrossi WE, Simoneau J, Cohen MS, Zimmerman SM, Henson CM, Rice KC, Woods JH (2010) Interaction of 5-HT2A and 5-HT2C receptors in R(−)-2,5-dimethoxy-4-iodoamphetamine-elicited head twitch behavior in mice. J Pharmacol Exp Ther 335:728–734
Fantegrossi WE, Gannon BM, Zimmerman SM, Rice KC (2013) In vivo effects of abused ‘bath salt’ constituent 3,4-methylenedioxypyrovalerone (MDPV) in mice: drug discrimination, thermoregulation, and locomotor activity. Neuropsychopharmacology 38:563–573
García-Pardo MP, Escobar-Valero C, Rodríguez-Arias M, Miñarro J, Aguilar MA (2015) Involvement of NMDA glutamate receptors in the acquisition and reinstatement of the conditioned place preference induced by MDMA. Behav Pharmacol 26:411–417
Gołembiowska K, Jurczak A, Kamińska K, Noworyta-Sokołowska K, Górska A (2016) Effect of some psychoactive drugs used as ‘legal highs’ on brain neurotransmitters. Neurotox Res 29:394–407
Green AR, Mechan AO, Elliott JM, O’Shea E, Colado MI (2003) The pharmacology and clinical pharmacology of 3,4-methylenedioxymethamphetamine (MDMA, “ecstasy”). Pharmacol Rev 55:463–508
Harris DS, Baggott M, Mendelson JH, Mendelson JE, Jones RT (2002) Subjective and hormonal effects of 3,4-methylenedioxymethamphetamine (MDMA) in humans. Psychopharmacology (Berl) 162:396–405
Hill SL, Thomas SH (2011) Clinical toxicology of newer recreational drugs. Clin Toxicol (Phila) 49:705–719
Huang PK, Aarde SM, Angrish D, Houseknecht KL, Dickerson TJ, Taffe MA (2012) Contrasting effects of d-methamphetamine, 3,4-methylenedioxymethamphetamine, 3,4-methylenedioxy-pyrovalerone, and 4-methylmethcathinone on wheel activity in rats. Drug Alcohol Depend 126:168–175
Kankaanpää A, Meririnne E, Lillsunde P, Seppälä T (1998) The acute effects of amphetamine derivatives on extracellular serotonin and dopamine levels in rat nucleus accumbens. Pharmacol Biochem Behav 59:1003–1009
Koob GF, Le Moal M (2001) Drug addiction, dysregulation of reward, and allostasis. Neuropsychopharmacology 24:97–129
Kravitz AV, Tye LD, Kreitzer AC (2012) Distinct roles for direct and indirect pathway striatal neurons in reinforcement. Nat Neurosci 15:816–818
Lamb RJ, Griffiths RR (1987) Self-injection of d,1-3,4-methylenedioxy methamphetamine (MDMA) in the baboon. Psychopharmacology (Berl) 91:268–272
Lawn W, Barratt M, Williams M, Horne A, Winstock A (2014) The NBOMe hallucinogenic drug series: patterns of use, characteristics of users and self-reported effects in a large international sample. J Psychopharmacol 28:780–788
Li P, Shah S, Huang L, Carr AL, Gao Y, Thisse C, Thisse B, Li L (2007) Cloning and spatial and temporal expression of the zebrafish dopamine D1 receptor. Dev Dyn 236:1339–1346
Liechti M (2015) Novel psychoactive substances (designer drugs): overview and pharmacology of modulators of monoamine signaling. Swiss Med Weekly 145:w14043
Monte AP, Waldman SR, Marona-Lewicka D, Wainscott DB, Nelson DL, Sanders-Bush E, Nichols DE (1997) Dihydrobenzofuran analogues of hallucinogens. 4. Mescaline derivatives. J Med Chem 40:2997–3008
Müller CP, Homberg JR (2015) Serotonin revisited. Behav Brain Res 277:1–2
Neelkantan N, Mikhaylova A, Stewart AM, Arnold R, Gjeloshi V, Kondaveeti D, Poudel MK, Kalueff AV (2013) Perspectives on zebrafish models of hallucinogenic drugs and related psychotropic compounds. ACS Chem 4:1137–1150
Nichols DE (2004) Hallucinogens. Pharmacol Ther 101:131–181
Nichols DE (2016) Psychedelics. Pharmacol Rev 68:264–355
O’Shea E, Escobedo I, Orio L, Sanchez V, Navarro M, Green AR, Colado MI (2005) Elevation of ambient room temperature has differential effects on MDMA-induced 5-HT and dopamine release in striatum and nucleus accumbens of rats. Neuropsychopharmacology 30:1312–1323
Owens MJ, Knight DL, Ritchie JC, Nemeroff CB (1991) The 5-hydroxy-tryptamine 2 agonist, (±)-1-(2,5-dimethoxy-4-bromophenyl)-2-aminopropane stimulates the hypothalamic-pituitary-adrenal (HPA) axis. II. Biochemical and physiological evidence for the development of tolerance after chronic administration. J Pharmacol Exp Ther 256:787–794
Parrott AC (2013) Human psychobiology of MDMA or ‘Ecstasy’: an overview of 25 years of empirical research. Hum Psychopharmacol 28:289–307
Parrott AC, Gibbs A, Scholey AB, King R, Owens K, Swann P, Ogden E, Stough C (2011a) MDMA and methamphetamine: some paradoxical negative and positive mood changes in an acute dose laboratory study. Psychopharmacology 215:527–536
Parrott AC, Evans LJ, Howells J, Robart R (2011b) Cocaine versus Ecstasy/ MDMA: comparative effects on mood and cognition in recreational users. Open Addict J 4:36–37
Ponzoni L, Braida D, Pucci L, Andrea D, Fasoli F, Manfredi I, Papke RL, Stokes C, Cannazza G, Clementi F, Gotti C, Sala M (2014) The cytisine derivatives, CC4 and CC26, reduce nicotine-induced conditioned place preference in zebrafish by acting on heteromeric neuronal nicotinic acetylcholine receptors. Psychopharmacology (Berl) 231:4681–4693
Ribeiro Do Couto B, Daza-Losada M, Rodríguez-Arias M, Nadal R, Guerri C, Summavielle T, Miñarro J, Aguilar MA (2012) Adolescent pre-exposure to ethanol and 3,4-methylenedioxymethylamphetamine (MDMA) increases conditioned rewarding effects of MDMA and drug-induced reinstatement. Addict Biol 17:588–600
Robertson JL, Savin NE, Preisler HK, Russell RM (1992) Pesticide bioassays with arthropods. CRC Press, Boca Raton
Robledo P, Mendizabal V, Ortuño J, de la Torre R, Kieffer BL, Maldonado R (2004) The rewarding properties of MDMA are preserved in mice lacking mu-opioid receptors. Eur J Neurosci 20:853–858
Rodríguez-Arias M, Manzanedo C, Roger-Sánchez C, Do Couto BR, Aguilar MA, Miñarro J (2010) Effect of adolescent exposure to WIN 55212–2 on the acquisition and reinstatement of MDMA-induced conditioned place preference. Prog Neuropsychopharmacol Biol Psychiatry 34:166–171
Roger-Sánchez C, Rodríguez-Arias M, Miñarro J, Aguilar MA (2013a) Effects of risperidone on the acquisition and reinstatement of the conditioned place preference induced by MDMA. Brain Res Bull 98:36–43
Roger-Sánchez C, Rodríguez-Arias M, Miñarro J, Aguilar MA (2013b) Involvement of 5-hydroxytryptamine 5-HT3 serotonergic receptors in the acquisition and reinstatement of the conditioned place preference induced by MDMA. Eur J Pharmacol 714:132–141
Roger-Sánchez C, Aguilar MA, Manzanedo C, Miñarro J, Rodríguez-Arias M (2013c) Neurochemical substrates of MDMA reward: effects of the inhibition of serotonin reuptake on the acquisition and reinstatement of MDMA-induced CPP. Curr Pharm Des 19:7050–7064
Schenk S, Gittings D, Colussi-Mas J (2011) Dopaminergic mechanisms of reinstatement of MDMA-seeking behaviour in rats. Br J Pharmacol 162:1770–1780
Schneider H, Fritzky L, Williams J, Heumann C, Yochum M, Pattar K, Noppert G, Mock V, Hawley E (2012) Cloning and expression of a zebrafish 5-HT2C receptor gene. Gene 502:108–117
Stewart A, Riehl R, Wong K, Green J, Cosgrove J, Vollmer K, Kyzar E, Hart P, Allain A, Cachat J, Gaikwad S, Hook M, Rhymes K, Newman A, Utterback E, Chang K, Kalueff AV (2011) Behavioral effects of MDMA (“Ecstasy”) on adult zebrafish. Behav Pharmacol 22:275–280
Streisinger G (2000) The zebrafish book. Oregon Press, Eugene
Tran S, Nowicki M, Muraleetharan A, Gerlai R (2015) Differential effects of dopamine D1 and D 2/3 receptor antagonism on motor responses. Psychopharmacology (Berl) 232:795–806
Tzschentke TM (2007) Measuring reward with the conditioned place preference (CPP) paradigm: update of the last decade. Addict Biol 12:227–462
United Nations Office on Drugs and Crime (2015) World drug report. United Nations publication, Vienna
Vandewater SA, Creehan KM, Taffe MA (2015) Intravenous self-administration of entactogen-class stimulants in male rats. Neuropharmacology 99:538–545
Vidal-Infer A, Roger-Sánchez C, Daza-Losada M, Aguilar MA, Miñarro J, Rodríguez-Arias M (2012) Role of the dopaminergic system in the acquisition, expression and reinstatement of MDMA-induced conditioned place preference in adolescent mice. PLoS One 7(8), e43107
Winkelman M (1991) Therapeutic effects of hallucinogens. Anthropol Conscious 2:15–19
Winter JC (1984) The stimulus properties of p-methoxyamphetamine: a nonessential serotonergic component. Pharmacol Biochem Behav 20:201–203
Acknowledgments
This research was partially supported by Fondazione Zardi-Gori (Milan, Italy).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The experiments were performed in compliance with the recommendations of the European Community Council Directive No. 86/609/EEC and the subsequent Italian Law on the Protection of animals used for experimental and other scientific reasons. The experimental protocol was approved by the Italian Governmental Decree No. 18/2013. All efforts were made to minimize the number of animals used and their discomfort.
Conflict of interest
The authors do not declare any conflict of interest.
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s00213-016-4433-4.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Video S1
Trance-like effect following PMA (0.1 mg/kg) evaluated within 5 min after treatment. (MP4 4162 kb)
Rights and permissions
About this article
Cite this article
Ponzoni, L., Daniela, B. & Sala, M. Abuse potential of methylenedioxymethamphetamine (MDMA) and its derivatives in zebrafish: role of serotonin 5HT2-type receptors. Psychopharmacology 233, 3031–3039 (2016). https://doi.org/10.1007/s00213-016-4352-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-016-4352-4