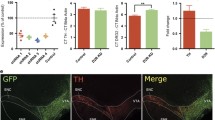
Abstract
Rationale
Increased activity of prefrontal D1 dopamine receptors (D1R) is involved in reward-related behavior found in bipolar disorder and drug addiction. While the effects of elevated D1R are known, depressive-like behaviors also occur in these disorders after reward-seeking ends.
Objectives
The goal is to characterize how termination of D1R overexpression influences depressive-like behaviors.
Methods
An inducible (Tet.On), lentiviral vector was used to manipulate the expression of the DRD1 gene in glutamate neurons within the prefrontal cortex in male, adult rats. Sexual activity and sucrose preference were studied in both D1R elevated ON and relatively reduced OFF states. Following termination of the D1R ON state, depressive-like behavior was determined in the OFF state. Expression of the transcriptional regulator, cyclic AMP-responsive element-binding protein (CREB), was used as an indication of downstream effects in the nucleus accumbens (NA).
Results
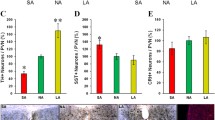
ON D1R expression increased sexual activity that returned to baseline in the OFF state. Sucrose preferences increased ~6 % in ON state but fell 11 % below control levels when OFF. Consistent with a depressive-like phenotype, D1R OFF decreased activity by 40 %, impaired the ability to control (43 %) and motivation to escape shock (27 % more impaired) relative to dsRed OFF. CREB increased 29 % in the NA in the D1R OFF state relative to the ON state.
Conclusions
This novel approach demonstrates that elevated D1R expression increased hedonic behavior, whereas the termination of D1R overexpression often resulted in depressive-like behavior. These observations support a role for D1R expression cycling in bipolar-associated behaviors and addiction.





Similar content being viewed by others
References
Ågmo A (1997) Male rat sexual behavior. Brain Res Protocol 1:203–209. doi:10.1016/S1385-299X(96)00036-0
Amat J, Baratta MV, Paul E et al (2005) Medial prefrontal cortex determines how stressor controllability affects behavior and dorsal raphe nucleus. Nat Neurosci 8:365–371. doi:10.1038/nn1399
Andersen SL (2015) Exposure to early adversity: points of cross-species translation that can lead to improved understanding of depression. Dev Psychopathol 27:477–491. doi:10.1017/S0954579415000103
Banasr M, Duman RS (2008) Glial loss in the prefrontal cortex is sufficient to induce depressive-like behaviors. Biol Psychiatry 64:863–870. doi:10.1016/j.biopsych.2008.06.008
Barr AM, Fiorino DF, Phillips AG (1999) Effects of withdrawal from an escalating dose schedule of d-amphetamine on sexual behavior in the male rat. Pharmacol Biochem Behav 64:597–604
Barrot M, Olivier JDA, Perrotti LI et al (2002) CREB activity in the NA shell controls gating of behavioral responses to emotional stimuli. Proc Natl Acad Sci U S A 99:11435–11440. doi:10.1073/pnas.172091899
Barrot M, Wallace DL, Bolaños CA et al (2005) Regulation of anxiety and initiation of sexual behavior by CREB in the NA. Proc Natl Acad Sci U S A 102:8357–8362. doi:10.1073/pnas.0500587102
Basco MR, Celis-de Hoyos CE (2012) Biopsychosocial model of hypersexuality in adolescent girls with bipolar disorder: strategies for intervention. J Child Adolesc Psychiatr Nurs Off Publ Assoc Child Adolesc Psychiatr Nurses Inc 25:42–50. doi:10.1111/j.1744-6171.2011.00312.x
Berk M, Dodd S, Kauer-Sant’anna M, et al (2007) Dopamine dysregulation syndrome: implications for a dopamine hypothesis of bipolar disorder. Acta Psychiatr Scand Suppl 41–9. doi: 10.1111/j.1600-0447.2007.01058.x
Berridge KC, Kringelbach ML (2013) Neuroscience of affect: brain mechanisms of pleasure and displeasure. Curr Opin Neurobiol 23:294–303. doi:10.1016/j.conb.2013.01.017
Bolla K, Ernst M, Kiehl K et al (2004) Prefrontal cortical dysfunction in abstinent cocaine abusers. J Neuropsychiatry Clin Neurosci 16:456–464. doi:10.1176/appi.neuropsych.16.4.456
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brenhouse HC, Sonntag KC, Andersen SL (2008) Transient D1 dopamine receptor expression on prefrontal cortex projection neurons: relationship to enhanced motivational salience of drug cues in adolescence. J Neurosci Off J Soc Neurosci 28:2375–2382. doi:10.1523/JNEUROSCI.5064-07.2008
Brenhouse H, Lukkes J, Andersen S (2013) Early life adversity alters the developmental profiles of addiction-related prefrontal cortex circuitry. Brain Sci 3:143–158
Carlezon WA, Duman RS, Nestler EJ (2005) The many faces of CREB. Trends Neurosci 28:436–445. doi:10.1016/j.tins.2005.06.005
Chandra R, Francis TC, Konkalmatt P, Amgalan A, Gancarz AM, Dietz DM, Lobo MK (2015) Opposing role for Egr3 in NA cell subtypes in cocaine action. J Neurosci 35:7927–7937. doi:10.1523/JNEUROSCI.0548-15.2015
Correa M, Carlson BB, Wisniecki A, Salamone JD (2002) NA dopamine and work requirements on interval schedules. Behav Brain Res 137:179–187
DelBello MP, Adler CM, Cerullo MA, et al. (2008) Bipolar disorder. In: Squire LR (ed). Elsevier, Amsterdam
Dell’Osso B, Ketter TA, Cremaschi L et al (2013) Assessing the roles of stimulants/stimulant-like drugs and dopamine-agonists in the treatment of bipolar depression. Curr Psychiatry Rep 15:1–12. doi:10.1007/s11920-013-0378-z
Der-Avakian A, Markou A (2012) The neurobiology of anhedonia and other reward-related deficits. Trends Neurosci 35:68–77. doi:10.1016/j.tins.2011.11.005
Dinieri JA, Nemeth CL, Parsegian A, Carle T, Gurevich VV, Gurevich E, Neve RL, Nestler EJ, Carlezon WA Jr (2009) Altered sensitivity to rewarding and aversive drugs in mice with inducible disruption of cAMP response element-binding protein function within the NA. J Neurosci 29:1855–1859. doi:10.1523/JNEUROSCI.5104-08.2009s
Donahue RJ, Muschamp JW, Russo SJ et al (2014) Effects of striatal ΔFosB overexpression and ketamine on social defeat stress-induced anhedonia in mice. Biol Psychiatry. doi:10.1016/j.biopsych.2013.12.014
Dvorak RD, Wray TB, Kuvaas NJ, Kilwein TM (2013) Mania and sexual risk: associations with behavioral self-regulation. J Affect Disord 150:1076–1081. doi:10.1016/j.jad.2013.04.023
Eisch AJ, Bolanos CA, de Wit J et al (2003) Brain-derived neurotrophic factor in the ventral midbrain-NA pathway: a role in depression. Biol Psychiatry 54:994–1005
Floresco SB, Phillips AG (2001) Delay-dependent modulation of memory retrieval by infusion of a dopamine D1 agonist into the rat medial prefrontal cortex. Behav Neurosci 115:934–939
Freund N, Thompson BS, Denormandie J et al (2013) Windows of vulnerability: maternal separation, age, and fluoxetine on adolescent depressive-like behavior in rats. Neuroscience 249:88–97. doi:10.1016/j.neuroscience.2013.06.066
Goldman-Rakic PS (1999) The “psychic” neuron of the cerebral cortex. Ann N Y Acad Sci 868:13–26. doi:10.1111/j.1749-6632.1999.tb11270.x
Goldstein RZ, Volkow ND (2002) Drug addiction and its underlying neurobiological basis: neuroimaging evidence for the involvement of the frontal cortex. Am J Psychiatry 159:1642–1652
Gonul AS, Coburn K, Kula M (2009) Cerebral blood flow, metabolic, receptor, and transporter changes in bipolar disorder: the role of PET and SPECT studies. Int Rev Psychiatry Abingdon Engl 21:323–335. doi:10.1080/09540260902962131
Hains AB, Arnsten AFT (2008) Molecular mechanisms of stress-induced prefrontal cortical impairment: implications for mental illness. Learn Mem 15:551–564. doi:10.1101/lm.921708
John CS, Smith KL, Van’t Veer A et al (2012) Blockade of astrocytic glutamate uptake in the prefrontal cortex induces anhedonia. Neuropsychopharmacology 37:2467–2475. doi:10.1038/npp.2012.105
Kalivas PW, McFarland K (2003) Brain circuitry and the reinstatement of cocaine-seeking behavior. Psychopharmacology (Berlin) 168:44–56. doi:10.1007/s00213-003-1393-2
Kalivas PW, Volkow N, Seamans J (2005) Unmanageable motivation in addiction: a pathology in prefrontal-accumbens glutamate transmission. Neuron 45:647–650. doi:10.1016/j.neuron.2005.02.005
Kato T, Kubota M, Kasahara T (2007) Animal models of bipolar disorder. Neurosci Biobehav Rev 31:832–842. doi:10.1016/j.neubiorev.2007.03.003
Kennedy SH, Rizvi S (2009) Sexual dysfunction, depression, and the impact of antidepressants. J Clin Psychopharmacol 29:157–164. doi:10.1097/JCP.0b013e31819c76e9
Koob G, Kreek MJ (2007) Stress, dysregulation of drug reward pathways, and the transition to drug dependence. Am J Psychiatry 164:1149–1159. doi:10.1176/appi.ajp.2007.05030503
Kram ML, Kramer GL, Ronan PJ et al (2002) Dopamine receptors and learned helplessness in the rat: an autoradiographic study. Prog Neuropsychopharmacol Biol Psychiatry 26:639–645
Kroener S, Lavin A (2010) Altered dopamine modulation of inhibition in the prefrontal cortex of cocaine-sensitized rats. Neuropsychopharmacology 35:2292–2304. doi:10.1038/npp.2010.107
Leussis MP, Freund N, Brenhouse HC et al (2012) Depressive-like behavior in adolescents after maternal separation: sex differences, controllability, and GABA. Dev Neurosci 34:210–217. doi:10.1159/000339162
Loos M, Pattij T, Janssen MCW et al (2010) Dopamine receptor D1/D5 gene expression in the medial prefrontal cortex predicts impulsive choice in rats. Cereb Cortex 20:1064–1070. doi:10.1093/cercor/bhp167
McFarland K, Lapish CC, Kalivas PW (2003) Prefrontal glutamate release into the core of the NA mediates cocaine-induced reinstatement of drug-seeking behavior. J Neurosci Off J Soc Neurosci 23:3531–3537
Minton GO, Young AH, McQuade R et al (2009) Profound changes in dopaminergic neurotransmission in the prefrontal cortex in response to flattening of the diurnal glucocorticoid rhythm: implications for bipolar disorder. Neuropsychopharmacology 34:2265–2274. doi:10.1038/npp.2009.53
Moscarello JM, LeDoux JE (2013) Active avoidance learning requires prefrontal suppression of amygdala-mediated defensive reactions. J Neurosci 33:3815–3823. doi:10.1523/JNEUROSCI.2596-12.2013
Murphy DL, Brodie HKH, Goodwin FK, Bunney WE (1971) Regular induction of hypomania by L-Dopa in “bipolar” manic-depressive patients. Nature 229:135–136. doi:10.1038/229135a0
Muschamp JW, Carlezon WA (2013) Roles of NA CREB and dynorphin in dysregulation of motivation. Cold Spring Harb Perspect Med 3:a012005. doi:10.1101/cshperspect.a012005
Muschamp JW, Veer AV, Parsegian A et al (2011) Activation of CREB in the NA shell produces anhedonia and resistance to extinction of fear in rats. J Neurosci 31:3095–3103. doi:10.1523/JNEUROSCI.5973-10.2011
Nestler EJ, Hyman SE (2010) Animal models of neuropsychiatric disorders. Nat Neurosci 13:1161–1169
Nirenberg MJ (2013) Dopamine agonist withdrawal syndrome: implications for patient care. Drugs Aging 30:587–592. doi:10.1007/s40266-013-0090-z
Olson VG, Zabetian CP, Bolanos CA, Edwards S, Barrot M, Eisch AJ, Hughes T, Self DW, Neve RL, Nestler EJ (2005) Regulation of drug reward by cAMP response element-binding protein: evidence for two functionally distinct subregions of the ventral tegmental area. J Neurosci 25:5553–5562. doi:10.1523/JNEUROSCI.0345-05.2005
Overstreet DH (2012) Modeling depression in animal models. Methods Mol Biol Clifton NJ 829:125–144. doi:10.1007/978-1-61779-458-2_7
Pantazopoulos H, Stone D, Walsh J, Benes FM (2004) Differences in the cellular distribution of D1 receptor mRNA in the hippocampus of bipolars and schizophrenics. Synapse 54:147–155. doi:10.1002/syn.20076
Paspalas CD, Wang M, Arnsten AFT (2013) Constellation of HCN channels and cAMP regulating proteins in dendritic spines of the primate prefrontal cortex: potential substrate for working memory deficits in schizophrenia. Cereb Cortex 23:1643–1654. doi:10.1093/cercor/bhs152
Paxinos G, Watson CR, Emson PC (1980) AChE-stained horizontal sections of the rat brain in stereotaxic coordinates. J Neurosci Methods 3:129–149
Pliakas AM, Carlson RR, Neve RL et al (2001) Altered responsiveness to cocaine and increased immobility in the forced swim test associated with elevated cAMP response element-binding protein expression in NA. J Neurosci 21:7397–7403
Ponce G, Quiñones-Lombraña A, Martín-Palanco NG et al (2015) The addiction-related gene Ankk1 is oppositely regulated by D1R- and D2R-like dopamine receptors. Neurotox Res. doi:10.1007/s12640-015-9545-9
Powell TR, Fernandes C, Schalkwyk LC (2011) Depression-related behavioral tests. Wiley
Pryce CR, Azzinnari D, Spinelli S et al (2011) Helplessness: a systematic translational review of theory and evidence for its relevance to understanding and treating depression. Pharmacol Ther 132:242–267. doi:10.1016/j.pharmthera.2011.06.006
Richard JM, Berridge KC (2013) Prefrontal cortex modulates desire and dread generated by NA glutamate disruption. Biol Psychiatry 73:360–370. doi:10.1016/j.biopsych.2012.08.009
Robbins TW (2005) Controlling stress: how the brain protects itself from depression. Nat Neurosci 8:261–262. doi:10.1038/nn0305-261
Salamone JD, Cousins MS, Bucher S (1994) Anhedonia or anergia? Effects of haloperidol and NA dopamine depletion on instrumental response selection in a T-maze cost/benefit procedure. Behav Brain Res 65:221–229
Sonntag KC, Brenhouse HC, Freund N et al (2014) Viral over-expression of D1 dopamine receptors in the prefrontal cortex increase high-risk behaviors in adults: comparison with adolescents. Psychopharmacology (Berlin). doi:10.1007/s00213-013-3399-8
Stoker AK, Markou A (2011) Withdrawal from chronic cocaine administration induces deficits in brain reward function in C57BL/6J mice. Behav Brain Res 223:176–181. doi:10.1016/j.bbr.2011.04.042
Suhara T, Nakayama K, Inoue O et al (1992) D1 dopamine receptor binding in mood disorders measured by positron emission tomography. Psychopharmacology (Berlin) 106:14–18
Tanda K, Nishi A, Matsuo N et al (2009) Abnormal social behavior, hyperactivity, impaired remote spatial memory, and increased D1-mediated dopaminergic signaling in neuronal nitric oxide synthase knockout mice. Mol Brain 2:19. doi:10.1186/1756-6606-2-19
Tye KM, Mirzabekov JJ, Warden MR et al (2013) Dopamine neurons modulate neural encoding and expression of depression-related behaviour. Nature 493:537–541. doi:10.1038/nature11740
Wallace DL, Han M-H, Graham DL et al (2009) CREB regulation of NA excitability mediates social isolation-induced behavioral deficits. Nat Neurosci 12:200–209. doi:10.1038/nn.2257
Weintraub D (2008) Dopamine and impulse control disorders in Parkinson’s disease. Ann Neurol 64:S93–S100. doi:10.1002/ana.21454
Willner P, Towell A, Sampson D et al (1987) Reduction of sucrose preference by chronic unpredictable mild stress, and its restoration by a tricyclic antidepressant. Psychopharmacology (Berlin) 93:358–364. doi:10.1007/BF00187257
Xing G, Zhang L, Russell S, Post R (2006) Reduction of dopamine-related transcription factors Nurr1 and NGFI-B in the prefrontal cortex in schizophrenia and bipolar disorders. Schizophr Res 84:36–56. doi:10.1016/j.schres.2005.11.006
Yao J, Pan Y, Ding M et al (2013) Meta-analysis shows dopamine receptor D1 gene polymorphism is associated with bipolar disorder but not with schizophrenia. Psychiatry Res 210:1324–1325. doi:10.1016/j.psychres.2013.08.031
Young JW, van Enkhuizen J, Winstanley CA, Geyer MA (2011) Increased risk-taking behavior in dopamine transporter knockdown mice: further support for a mouse model of mania. J Psychopharmacol 25:934–943. doi:10.1177/0269881111400646
Zhong P, Liu X, Zhang Z, Hu Y, Liu SJ, Lezama-Ruiz M, Joksimovic M, Liu QS (2014) Cyclin-dependent kinase 5 in the ventral tegmental area regulates depression-related behaviors. J Neurosci 34:6352–6366. doi:10.1523/JNEUROSCI.3673-13.2014
Acknowledgments
We want to thank the Leopoldina Fellowship Program LPDS-40, the NARSAD Young Investigator Grant 2012, the van Ameringen Foundation (to NF), DA-026485 and DA-015403 and the Simches Family (to SLA) for support, Dr. David Sibley (NIH) for providing the D1 receptor plasmid, Dr. Karl Deisseroth (Stanford University) for the CamKIIα promotor, Heather Brenhouse for her help with conducting the experiments and Nellie Mary for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The experiments were conducted in accordance with the principles of animal care as set forth in the Guide for the Care and Use of Laboratory Animals (NIH), and were approved by the Institutional Animal Care and Use Committee at McLean Hospital.
Financial disclosure
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Freund, N., Thompson, B.S., Sonntag, K. et al. When the party is over: depressive-like states in rats following termination of cortical D1 receptor overexpression. Psychopharmacology 233, 1191–1201 (2016). https://doi.org/10.1007/s00213-015-4200-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-015-4200-y