Abstract
Rationale
Allopregnanolone effects on mood in postmenopausal women are unclear thus far.
Objectives
Allopregnanolone is a neuroactive steroid with contradictory effects. Anaesthetic, sedative, and anxiolytic as well as aggressive and anxiogenic properties have been reported. The aim of this study is to compare severity of negative mood between women receiving different serum allopregnanolone concentrations during progesterone treatment.
Materials and methods
A randomized, placebo-controlled, double-blind, crossover study of postmenopausal women (n=43) treated with 2 mg estradiol daily during four treatment cycles. Oral micronized progesterone at 30, 60, and 200 mg/day, and placebo were added sequentially to each cycle. Participants kept daily symptom ratings using a validated rating scale. Blood samples for progesterone and allopregnanolone analyses were collected during each treatment cycle.
Results
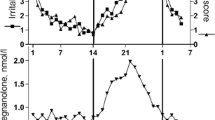
During progesterone treatment, women had significantly higher negative mood scores when allopregnanolone serum concentration was in the range of 1.5–2 nmol/l compared to lower and higher concentrations. In addition, women displayed a significant increase in negative mood during the progesterone treatment period, compared to the estradiol-only period when 30 mg progesterone daily was used. On the other hand, treatment with higher doses of progesterone had no influence on negative mood.
Conclusions
Mood effects during progesterone treatment seem to be related to allopregnanolone concentration, and a bimodal association between allopregnanolone and adverse mood is evident.




Similar content being viewed by others
References
Andreen L, Bixo M, Nyberg S, Sundstrom-Poromaa I, Backstrom T (2003) Progesterone effects during sequential hormone replacement therapy. Eur J Endocrinol 148:571–577
Andreen L, Spigset O, Andersson A, Nyberg S, Backstrom T (2006) Pharmacokinetics of progesterone and its metabolites allopregnanolone and pregnanolone after oral administration of low-dose progesterone. Maturitas, 2006 Jan 4; [Epub ahead of print]
Andreen L, Sundstrom-Poromaa I, Bixo M, Andersson A, Nyberg S, Backstrom T (2005) Relationship between allopregnanolone and negative mood in postmenopausal women taking sequential hormone replacement therapy with vaginal progesterone. Psychoneuroendocrinology 30:212–224
Backstrom T, Sanders D, Leask R, Davidson D, Warner P, Bancroft J (1983) Mood, sexuality, hormones, and the menstrual cycle. II. Hormone levels and their relationship to the premenstrual syndrome. Psychosom Med 45:503–507
Backstrom T, Zetterlund B, Blom S, Romano M (1984) Effects of intravenous progesterone infusions on the epileptic discharge frequency in women with partial epilepsy. Acta Neurol Scand 69:240–248
Beauchamp MH, Ormerod BK, Jhamandas K, Boegman RJ, Beninger RJ (2000) Neurosteroids and reward: allopregnanolone produces a conditioned place aversion in rats. Pharmacol Biochem Behav 67:29–35
Ben-Porath DD, Taylor SP (2002) The effects of diazepam (valium) and aggressive disposition on human aggression: an experimental investigation. Addict Behav 27:167–177
Bitran D, Shiekh M, McLeod M (1995) Anxiolytic effect of progesterone is mediated by the neurosteroid allopregnanolone at brain GABAA receptors. J Neuroendocrinol 7:171–177
Bixo M, Allard P, Backstrom T, Mjorndal T, Nyberg S, Spigset O, Sundstrom-Poromaa I (2001) Binding of [3H]paroxetine to serotonin uptake sites and of [3H]lysergic acid diethylamide to 5-HT2A receptors in platelets from women with premenstrual dysphoric disorder during gonadotropin releasing hormone treatment. Psychoneuroendocrinology 26:551–564
Bjorn I, Backstrom T (1999) Drug related negative side-effects is a common reason for poor compliance in hormone replacement therapy. Maturitas 32:77–86
Bjorn I, Sundstrom-Poromaa I, Bixo M, Nyberg S, Backstrom T (2003) Increase of estrogen dose deteriorates mood during progestin phase in sequential hormonal therapy. J Clin Endocrinol Metab 88(5):2026–2030
Bjorn I, Bixo M, Nojd KS, Collberg P, Nyberg S, Sundstrom-Poromaa I, Backstrom T (2002) The impact of different doses of medroxyprogesterone acetate on mood symptoms in sequential hormonal therapy. Gynecol Endocrinol 16:1–8
Bjorn I, Bixo M, Nojd KS, Nyberg S, Backstrom T (2000) Negative mood changes during hormone replacement therapy: a comparison between two progestogens. Am J Obstet Gynecol 183:1419–1426
Carl P, Hogskilde S, Nielsen JW, Sorensen MB, Lindholm M, Karlen B, Backstrom T (1990) Pregnanolone emulsion. A preliminary pharmacokinetic and pharmacodynamic study of a new intravenous anaesthetic agent. Anaesthesia 45:189–197
Cherek DR, Spiga R, Egli M (1992) Effects of response requirement and alcohol on human aggressive responding. J Exp Anal Behav 58:577–587
Czlonkowska AI, Krzascik P, Sienkiewicz-Jarosz H, Siemiatkowski M, Szyndler J, Maciejak P, Bidzinski A, Plaznik A (2001) Tolerance to the anticonvulsant activity of midazolam and allopregnanolone in a model of picrotoxin seizures. Eur J Pharmacol 425:121–127
de Lignieres B, Dennerstein L, Backstrom T (1995) Influence of route of administration on progesterone metabolism. Maturitas 21:251–257
Dougherty DM, Cherek DR, Bennett RH (1996) The effects of alcohol on the aggressive responding of women. J Stud Alcohol 57:178–186
Fish EW, DeBold JF, Miczek KA (2005) Escalated aggression as a reward: corticosterone and GABA(A) receptor positive modulators in mice. Psychopharmacology (Berl) 182:116–127
Fish EW, Faccidomo S, DeBold JF, Miczek KA (2001) Alcohol, allopregnanolone and aggression in mice. Psychopharmacology (Berl) 153:473–483
Freeman EW, Purdy RH, Coutifaris C, Rickels K, Paul SM (1993) Anxiolytic metabolites of progesterone: correlation with mood and performance measures following oral progesterone administration to healthy female volunteers. Neuroendocrinology 58:478–484
Genazzani AR, Petraglia F, Bernardi F, Casarosa E, Salvestroni C, Tonetti A, Nappi RE, Luisi S, Palumbo M, Purdy RH, Luisi M (1998) Circulating levels of allopregnanolone in humans: gender, age, and endocrine influences. J Clin Endocrinol Metab 83:2099–2103
Gourley SL, Debold JF, Yin W, Cook J, Miczek KA (2005) Benzodiazepines and heightened aggressive behavior in rats: reduction by GABA(A)/alpha(1) receptor antagonists. Psychopharmacology (Berl) 178:232–240
Greendale GA, Reboussin BA, Hogan P, Barnabei VM, Shumaker S, Johnson S, Barrett-Connor E (1998) Symptom relief and side effects of postmenopausal hormones: results from the Postmenopausal Estrogen/Progestin Interventions Trial. Obstet Gynecol 92:982–988
Gulinello M, Gong QH, Li X, Smith SS (2001) Short-term exposure to a neuroactive steroid increases alpha4 GABA(A) receptor subunit levels in association with increased anxiety in the female rat. Brain Res 910:55–66
Hammarback S, Backstrom T, Holst J, von Schoultz B, Lyrenas S (1985) Cyclical mood changes as in the premenstrual tension syndrome during sequential estrogen-progestagen postmenopausal replacement therapy. Acta Obstet Gynecol Scand 64:393–397
Holst J, Backstrom T, Hammarback S, von Schoultz B (1989) Progestogen addition during oestrogen replacement therapy—effects on vasomotor symptoms and mood. Maturitas 11:13–20
Johansson IM, Birzniece V, Lindblad C, Olsson T, Backstrom T (2002) Allopregnanolone inhibits learning in the Morris water maze. Brain Res 934:125–131
Kirkham C, Hahn PM, Van Vugt DA, Carmichael JA, Reid RL (1991) A randomized, double-blind, placebo-controlled, cross-over trial to assess the side effects of medroxyprogesterone acetate in hormone replacement therapy. Obstet Gynecol 78:93–97
Kurthen M, Linke DB, Reuter BM, Hufnagel A, Elger CE (1991) Severe negative emotional reactions in intracarotid sodium amytal procedures: further evidence for hemispheric asymmetries? Cortex 27:333–337
Landgren S, Selstam G (1995) Interaction between 17 beta-oestradiol and 3 alpha-hydroxy-5 alpha-pregnane-20-one in the control of neuronal excitability in slices from the CA1 hippocampus in vitro of guinea-pigs and rats. Acta Physiol Scand 154:165–176
Landgren S, Wang MD, Backstrom T, Johansson S (1998) Interaction between 3 alpha-hydroxy-5 alpha-pregnan-20-one and carbachol in the control of neuronal excitability in hippocampal slices of female rats in defined phases of the oestrus. Acta Physiol Scand 162:77–88
Lee GP, Loring DW, Meador KJ, Flanigin HF, Brooks BS (1988) Severe behavioral complications following intracarotid sodium amobarbital injection: implications for hemispheric asymmetry of emotion. Neurology 38:1233–1236
Magos AL, Brewster E, Singh R, O‘Dowd T, Brincat M, Studd JW (1986) The effects of norethisterone in postmenopausal women on oestrogen replacement therapy: a model for the premenstrual syndrome. Br J Obstet Gynaecol 93:1290–1296
Majewska MD, Harrison NL, Schwartz RD, Barker JL, Paul SM (1986) Steroid hormone metabolites are barbiturate-like modulators of the GABA receptor. Science 232:1004–1007
Masia SL, Perrine K, Westbrook L, Alper K, Devinsky O (2000) Emotional outbursts and post-traumatic stress disorder during intracarotid amobarbital procedure. Neurology 54:1691–1693
Miczek KA, DeBold JF, van Erp AM, Tornatzky W (1997) Alcohol, GABAA-benzodiazepine receptor complex, and aggression. Recent Dev Alcohol 13:139–171
Miczek KA, Fish EW, De Bold JF (2003) Neurosteroids, GABAA receptors, and escalated aggressive behavior. Horm Behav 44:242–257
Palmer AA, Moyer MR, Crabbe JC, Phillips TJ (2002) Initial sensitivity, tolerance and cross-tolerance to allopregnanolone- and ethanol-induced hypothermia in selected mouse lines. Psychopharmacology (Berl) 162:313–322
Pinna G, Costa E, Guidotti A (2005) Changes in brain testosterone and allopregnanolone biosynthesis elicit aggressive behavior. Proc Natl Acad Sci U S A 102:2135–2140
Pinna G, Dong E, Matsumoto K, Costa E, Guidotti A (2003) In socially isolated mice, the reversal of brain allopregnanolone down-regulation mediates the anti-aggressive action of fluoxetine. Proc Natl Acad Sci U S A 100:2035–2040
Purdy RH, Moore PH Jr, Rao PN, Hagino N, Yamaguchi T, Schmidt P, Rubinow DR, Morrow AL, Paul SM (1990) Radioimmunoassay of 3 alpha-hydroxy-5 alpha-pregnan-20-one in rat and human plasma. Steroids 55:290–296
Sanders D, Warner P, Backstrom T, Bancroft J (1983) Mood, sexuality, hormones and the menstrual cycle. I. Changes in mood and physical state: description of subjects and method. Psychosom Med 45:487–501
Schmidt PJ, Nieman LK, Danaceau MA, Adams LF, Rubinow DR (1998) Differential behavioral effects of gonadal steroids in women with and in those without premenstrual syndrome. N Engl J Med 338:209–216
Schmidt PJ, Purdy RH, Moore PH Jr, Paul SM, Rubinow DR (1994) Circulating levels of anxiolytic steroids in the luteal phase in women with premenstrual syndrome and in control subjects. J Clin Endocrinol Metab 79:1256–1260
Seippel L, Backstrom T (1998) Luteal-phase estradiol relates to symptom severity in patients with premenstrual syndrome. J Clin Endocrinol Metab 83:1988–1992
Smith SS, Gong QH, Hsu FC, Markowitz RS, ffrench-Mullen JM, Li X (1998) GABA(A) receptor alpha4 subunit suppression prevents withdrawal properties of an endogenous steroid. Nature 392: 926–930
Spitzer RL, Williams JB, Kroenke K, Linzer M, deGruy FV 3rd, Hahn SR, Brody D, Johnson JG (1994) Utility of a new procedure for diagnosing mental disorders in primary care. The PRIME-MD 1000 study. JAMA 272:1749–1756
Sundstrom I, Andersson A, Nyberg S, Ashbrook D, Purdy RH, Backstrom T (1998) Patients with premenstrual syndrome have a different sensitivity to a neuroactive steroid during the menstrual cycle compared to control subjects. Neuroendocrinology 67:126–138
Sundstrom I, Nyberg S, Bixo M, Hammarback S, Backstrom T (1999) Treatment of premenstrual syndrome with gonadotropin-releasing hormone agonist in a low dose regimen. Acta Obstet Gynecol Scand 78:891–899
Timby E, Balgard M, Nyberg S, Spigset O, Andersson A, Porankiewicz-Asplund J, Purdy RH, Zhu D, Backstrom T, Poromaa IS (2005) Pharmacokinetic and behavioral effects of allopregnanolone in healthy women. Psychopharmacology (Berl) 2005 Sept 21, pp 1–11 [Epub ahead of print]
Turkmen S, Lofgren M, Birzniece V, Backstrom T, Johansson I-M (2006) Tolerance development to Morris water maze test impairments induced by acute allopregnanolone. Neuroscience (in press) 2006 Jan 31 [Epub ahead of print]
Wang M, Seippel L, Purdy RH, Backstrom T (1996) Relationship between symptom severity and steroid variation in women with premenstrual syndrome: study on serum pregnenolone, pregnenolone sulfate, 5 alpha-pregnane-3,20-dione and 3 alpha-hydroxy-5 alpha- pregnan-20-one. J Clin Endocrinol Metab 81:1076–1082
Wenzel RR, Bartel T, Eggebrecht H, Philipp T, Erbel R (2002) Central-nervous side effects of midazolam during transesophageal echocardiography. J Am Soc Echocardiogr 15:1297–1300
Wieland S, Lan NC, Mirasedeghi S, Gee KW (1991) Anxiolytic activity of the progesterone metabolite 5 alpha-pregnan-3 alpha-o1-20-one. Brain Res 565:263–268
Wihlback AC, Nyberg S, Backstrom T, Bixo M, Sundstrom-Poromaa I (2005) Estradiol and the addition of progesterone increase the sensitivity to a neurosteroid in postmenopausal women. Psychoneuroendocrinology 30:38–50
Yoshimura H, Ogawa N (1989) Acute and chronic effects of psychotropic drugs on maternal aggression in mice. Psychopharmacology (Berl) 97:339–342
Zhu D, Birzniece V, Backstrom T, Wahlstrom G (2004) Dynamic aspects of acute tolerance to allopregnanolone evaluated using anaesthesia threshold in male rats. Br J Anaesth 93:560–567
Acknowledgements
This work was supported by the Swedish Medical Research Council (Proj. 4X-11198), Umeå sjukvård, spjutspetsanslag, Visare Norr Norra Regionen and by a grant from the EU Regional Funds, Objective 1. Agneta Andersson is acknowledged for skillful technical assistance, Margatetha Jänes and Ingrid Otterbring for skillful technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Andréen, L., Sundström-Poromaa, I., Bixo, M. et al. Allopregnanolone concentration and mood—a bimodal association in postmenopausal women treated with oral progesterone. Psychopharmacology 187, 209–221 (2006). https://doi.org/10.1007/s00213-006-0417-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-006-0417-0