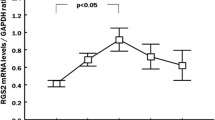
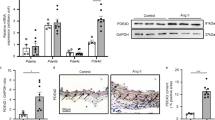
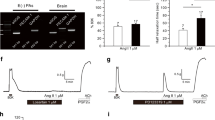
Abstract
Our previous study demonstrated that adenine (6-amino-6H-purine) relaxed contracted rat aorta rings in an endothelial-independent manner. Although adenine receptors (AdeRs) are expressed in diverse tissues, aortic AdeR expression has not been ascertained. Thus, the aims of this study were to clarify the expression of AdeR in rat vascular smooth muscle cells (VSMCs) and to investigate the adenine-induced vasorelaxation mechanism(s). VSMCs were isolated from 8-week-old male Wistar-Kyoto rats and used in this study. Phosphorylation of myosin light chain (p-MLC) was measured by western blot. AdeR mRNA was detected by RT-PCR. Intracellular Ca2+ concentration ([Ca2+]i) was measured by using Fura-2/AM. Vasorelaxant adenine (10–100 μM) significantly reduced p-MLC by angiotensin II (Ang II, 10 μM) in VSMCs (P < 0.05). We confirmed the expression of aortic AdeR mRNA and the activation of PKA in VSMCs through stimulation of AdeR by adenine by ELISA. Intracellular Ca2+ concentration ([Ca2+]i) measurement demonstrated that adenine inhibits Ang II- and m-3M3FBS (PLC agonist)-induced [Ca2+]i elevation. In AdeR-knockdown VSMCs, PKA activation and p-MLC reduction by adenine were completely abolished. These results firstly demonstrated that vasorelaxant adenine can suppress Ca2+ contraction signaling pathways via aortic AdeR/PKA activation in VSMCs.







Similar content being viewed by others
References
Archer SL, Huang JM, Hampl V, et al. (1994) Nitric oxide and cGMP cause vasorelaxation by activation of a charybdotoxin-sensitive K channel by cGMP-dependent protein kinase. Proc Natl Acad Sci U S A 91:7583–7587. doi:10.1073/pnas.91.16.7583
Bender E, Buist A, Jurzak M, et al. (2002) Characterization of an orphan G protein-coupled receptor localized in the dorsal root ganglia reveals adenine as a signaling molecule. Proc Natl Acad Sci U S A 99:8573–8578. doi:10.1073/pnas.122016499
Fimia GM, Sassone-Corsi P (2001) Cyclic AMP signalling. J Cell Sci 114:1971–1972
Godfraind T (1994) Calcium antagonists and vasodilation. Pharmacol Ther 64:37–75. doi:10.1016/0163-7258(94)90033-7
Grynkiewicz G, Poenie M, Tsien RY (1985) A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem 260:3440–3450. doi: 3838314
Huang J, Zhou H, Mahavadi S, et al. (2007) Inhibition of Galphaq-dependent PLC-beta1 activity by PKG and PKA is mediated by phosphorylation of RGS4 and GRK2. Am J Phys Cell Phys 292:C200–C208. doi:10.1152/ajpcell.00103.2006
Intengan HD, Schiffrin EL (2001) Vascular remodeling in hypertension: roles of apoptosis, inflammation, and fibrosis. Hypertension 38:581–587. doi:10.1161/hy09t1.096249
Kelton JG, Blajchman MA (1980) Prostaglandin I2 (prostacyclin). Can Med Assoc J 122:175–179
Kishore BK, Zhang Y, Gevorgyan H, et al. (2013) Cellular localization of adenine receptors in the rat kidney and their functional significance in the inner medullary collecting duct. Am J Phys Renal Phys 305:F1298–F1305. doi:10.1152/ajprenal.00254.2013
Knospe M, Müller CE, Rosa P, et al. (2013) The rat adenine receptor: pharmacological characterization and mutagenesis studies to investigate its putative ligand binding site. Purinergic Signal 9:367–381. doi:10.1007/s11302-013-9355-6
Kobayashi Y, Fukuda T, Tanaka M, Matsui T (2012) The anti-atherosclerotic di-peptide, Trp-His, inhibits the phosphorylation of voltage-dependent L-type Ca2+ channels in rat vascular smooth muscle cells. FEBS Open Bio 2:83–88. doi:10.1016/j.fob.2012.04.005
Kumrungsee T, Saiki T, Akiyama S, et al. (2014) Inhibition of calcium-calmodulin complex formation by vasorelaxant basic dipeptides demonstrated by in vitro and in silico analyses. Biochim Biophys Acta, Gen Subj 1840:3073–3078. doi:10.1016/j.bbagen.2014.07.020
Lee T, Karl R, Moosmang S, et al. (2006) Calmodulin kinase II is involved in voltage-dependent facilitation of the L-type Ca v1.2 Calcium Channel. J Biol Chem 281:25560–25567. doi:10.1074/jbc.M508661200
Matthews EA, Dickenson AH (2004) Effects of spinally administered adenine on dorsal horn neuronal responses in a rat model of inflammation. Neurosci Lett 356:211–214. doi:10.1016/j.neulet.2003.11.057
Mehta PK, Griendling KK (2007) Angiotensin II cell signaling: physiological and pathological effects in the cardiovascular system. Am J Phys Cell Phys 292:C82–C97. doi:10.1152/ajpcell.00287.2006
Murphy K, Gerzanich V, Zhou HUI, et al. (2003) Adenosine-A2a receptor down-regulates cerebral smooth muscle L-type Ca 2ϩ channel activity via protein tyrosine phosphatase, not cAMP-dependent protein kinase. Mol Pharmacol 64:640–649. doi:10.1124/mol.64.3.640
Pollock DM, Keith TL, Highsmith RF (1995) Endothelin receptors and calcium signaling. FASEB J 9:1196–1204
Somlyo AP, Somlyo AV (1994) Signal transduction and regulation in smooth muscle. Nature 372:231–236. doi:10.1038/372231a0
Tanaka M, Watanabe S, Wang Z, et al. (2009) His-Arg-Trp potently attenuates contracted tension of thoracic aorta of Sprague-Dawley rats through the suppression of extracellular Ca2+ influx. Peptides 30:1502–1507. doi:10.1016/j.peptides.2009.05.012
Thimm D, Knospe M, Abdelrahman A, et al. (2013) Characterization of new G protein-coupled adenine receptors in mouse and hamster. Purinergic Signal 9:415–426. doi:10.1007/s11302-013-9360-9
Thimm D, Schiedel AC, Peti-Peterdi J, et al. (2015) The nucleobase adenine as a signalling molecule in the kidney. Acta Physiol (Oxford) 213:808–818. doi:10.1111/apha.12452
Timmermans PB, Wong PC, Chiu AT, et al. (1993) Angiotensin II receptors and angiotensin II receptor antagonists. Pharmacol Rev 45:205–251
Tokuda S, Fukuda T, Kobayashi Y, et al. (2012) Effect of the uncharged imidazolium moiety in adenine on endothelium-independent relaxation in the contracted thoracic aorta of Sprague-Dawley rats. Biosci Biotechnol Biochem 76:828–830. doi:10.1271/bbb.110806
Wang Z, Watanabe S, Kobayashi Y, et al. (2010) Trp-His, a vasorelaxant di-peptide, can inhibit extracellular Ca2+ entry to rat vascular smooth muscle cells through blockade of dihydropyridine-like L-type Ca2+ channels. Peptides 31:2060–2066. doi:10.1016/j.peptides.2010.07.013
Watanabe S, Yoshimi Y, Ikekita M (2003) Neuroprotective effect of adenine on Purkinje cell survival in rat cerebellar primary cultures. J Neurosci Res 74:754–759. doi:10.1002/jnr.10790
Watanabe A, Sohail MA, Gautam S, et al. (2012) Adenine induces differentiation of rat hepatic stellate cells. Dig Dis Sci 57:2371–2378. doi:10.1007/s10620-012-2183-7
Wengert M, Adão-Novaes J, Assaife-Lopes N, et al. (2007) Adenine-induced inhibition of Na+-ATPase activity: evidence for involvement of the Gi protein-coupled receptor in the cAMP signaling pathway. Arch Biochem Biophys 467:261–267. doi:10.1016/j.abb.2007.08.018
Acknowledgment
This study was supported in part by a grand-in-aid from the Ministry of Education, Science, Sports and Culture of Japan (No. 22248014) to TM and by the Japan Society for the Promotion of Science Research Fellowship for Young Scientists to TF (No. 14 J03039).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 197 kb)
Rights and permissions
About this article
Cite this article
Fukuda, T., Kuroda, T., Kono, M. et al. Adenine attenuates the Ca2+ contraction-signaling pathway via adenine receptor-mediated signaling in rat vascular smooth muscle cells. Naunyn-Schmiedeberg's Arch Pharmacol 389, 999–1007 (2016). https://doi.org/10.1007/s00210-016-1264-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-016-1264-0