Abstract
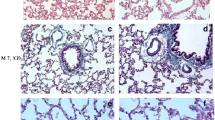
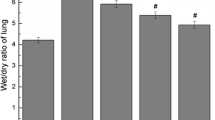
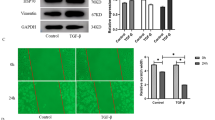
Pulmonary fibrosis is a progressive lung disorder with high mortality rate and limited successful treatment. This study was designed to assess the potential anti-oxidant and anti-fibrotic effects of aliskiren (Alsk) during bleomycin (BLM)-induced pulmonary fibrosis. Male Wistar rats were used as control untreated or treated with the following: a single dose of 2.5 mg/kg of BLM endotracheally and BLM and Alsk (either low dose 30 mg/kg/day or high dose 60 mg/kg/day), and another group was given Alsk 60 mg/kg/day alone. Alsk was given by gavage. Alsk anti-oxidant and anti-fibrotic effects were assessed. BLM significantly increased relative lung weight and the levels of lactate dehydrogenase and total and differential leucocytic count in bronchoalveolar lavage that was significantly ameliorated by high-dose Alsk treatment. As markers of oxidative stress, BLM caused a significant increase in the levels of lipid peroxides and nitric oxide accompanied with a significant decrease of superoxide dismutase and glutathione transferase enzymes. High-dose Alsk treatment restored these markers toward normal values. Alsk counteracted the overexpression of advanced glycation end products, matrix metalloproteinase-9 (MMP-9), and tissue inhibitor of metalloproteinases-1 in lung tissue induced by BLM. Fibrosis assessed by measuring hydroxyproline content, which markedly increased in the BLM group, was also significantly reduced by Alsk. These were confirmed by histopathological and immunohistochemical examination which revealed that Alsk attenuates signs of pulmonary fibrosis and decreased the overexpressed MMP-9 and transforming growth factor β1. Collectively, these findings indicate that Alsk has a potential anti-fibrotic effect beside its anti-oxidant activity.





Similar content being viewed by others
References
Ali S, Mann DA (2004) Signal transduction via the NF-kappa B pathway: a targeted treatment modality for infection, inflammation and repair. Cell Biochem Funct 22:67–79
Allred DC, Clark GM, Elledge R, et al. (1993) Association of p53 protein expression with tumor cell proliferation rate and clinical outcome in node-negative breast cancer. J Natl Cancer Inst 85:200–206
Ashcroft T, Simpson JM, Timbrell V (1988) Simple method of estimating severity of pulmonary fibrosis on a numerical scale. J Clin Pathol 41(4):467–470
Asker SA, Mazroa SA, Boshra V, et al. (2015) Biochemical and histological impact of direct renin inhibition byaliskiren on myofibroblasts activation and differentiation inbleomycin induced pulmonary fibrosis in adult mice. Tissue Cell. doi:10.1016/j.tice.2015.05.001
Bataller R, Sancho-Bru P, Gine’s P, et al. (2005) Liver fibrogenesis: a new role for the renin-angiotensin system. Antioxid Redox Signal 7:1346–1355
Bergeron A, Soler P, Kambouchner M, et al. (2003) Cytokine profiles in idiopathic pulmonary fibrosis suggest an important role for TGF-b and IL-10. Eur Respir J 22:69–76
Bhattacharyya P, Acharya D, Roychowdhury S (2007) Role of matrix metalloproteinases in the pathophysiology of idiopathic pulmonary fibrosis. Lung India 24:61–65
Bohlender JM, Franke S, Stein G, et al. (2005) Advanced glycation end products and the kidney. Am J Physiol Renal Physiol 289:F645–F659
Bonniaud P, Margetts PJ, Ask K, et al. (2005) TGF-beta and Smad3 signaling link inflammation to chronic fibro genesis. J Immunol 175:5390–5395
Boyaci H, Maral H, Turan G, et al. (2006) Effects of erdosteine on bleomycin-induced lung fibrosis in rats. Mol Cell Biochem 281(1–2):129–137
Broekelmann TJ, Limper AH, Colby TV, et al. (1991) Transforming growth factor beta 1 is present at sites of extracellular matrix gene expression in human pulmonary fibrosis. Proc Natl Acad Sci U S A 88:6642–6646
Chaudhary NI, Schnapp A, Park JE (2006) Pharmacologic differentiation of inflammation and fibrosis in the rat bleomycin model. Am J Respir Crit Care Med 173:769–776
Chen L, Wang T, Wang X, et al. (2009) Blockade of advanced glycation end product formation attenuates bleomycin-induced pulmonary fibrosis in rats. Respir Res 10:55. doi:10.1186/1465-9921-10-55
Cheresh P, Kim S, Tulasiram S, et al. (2013) Oxidative stress and pulmonary fibrosis. Biochim Biophys Acta 1832:1028–1040
Cherniack RM, Colby TV, Flint A, et al. (1991) Quantitative assessment of lung pathology in idiopathic fibrosis. Am J Respir Dis 144:892–900
Chung KF (2005) Inflammatory mediators in chronic obstructive pulmonary disease. Curr Drug Targets Inflamm Allergy 4:619–625
Coker RK, Laurent GJ, Jeffery PK, et al. (2001) Localization of transforming growth factor beta1 and beta3 mRNA transcripts in normal and fibrotic human lung. Thorax 56:549–556
Coker RK, Laurent GJ, Shahzeidi S, et al. (1997) Transforming growth factors-beta 1, -beta 2, and -beta 3 stimulate fibroblast procollagen production in vitro but are differentially expressed during bleomycin-induced lung fibrosis. Am J Pathol 150:981–991
Cooper SA, Whaley-Connell A, Habibi J, et al. (2007) Renin–angiotensin–aldosterone system and oxidative stress in cardiovascular insulin resistance. Am J Physiol Heart Circ Physiol 293:H2009–H2023
Cragg GM (1998) Paclitaxel (Taxol): a success story with valuable lessons for natural product drug discovery and development. Med Res Rev 18:315–331
Daba MA, Abdel-Aziz AH, Moustafa AM, et al. (2002) Effects of L carnitine and Ginkgo biloba extract (EGb 761) in experimental bleomycin-induced lung fibrosis. Pharmacol Res 45(6):461–467
Daniil ZD, Papaqeorqiou E, Koutsokera K, et al. (2008) Serum levels of oxidative stress as a marker of disease severity in idiopathic pulmonary fibrosis. Pulm Pharmacol Ther 21(1):26–31
El-Dakhakhny M (1965) Studies on the Egyptian Nigella sativa L IV Some pharmacological properties of the seeds’ active principle in comparison to its dihydro compound and its polymer. Arzneimittelforschung 15:1227–1229
El-Demerdash E (2011) Anti-inflammatory and anti-fibrotic effects of methyl palmitate. Toxicol Appl Pharmacol 254:238–244
El-Khatib AS (2002) Possible modulatory role of nitric oxide in lung toxicity induced in rats by chronic administration of bleomycin. Chemotherapy 48(4–5):244–251
El-Medany A, Hagar HH, Moursi M, et al. (2005) Attenuation of bleomycin induced lung fibrosis in rats by mesna. Eur J Pharmacol 509(1):61–70
Englert JM, Hanford LE, Kaminski N, et al. (2008) A role for the receptor for advanced glycation end products in idiopathic pulmonary fibrosis. Am J Pathol 172:583–591. doi:10.2353/ajpath.2008.070569
Faner R, Rojas M, Macnee W, et al. (2012) Abnormal lung aging in chronic obstructive pulmonary disease and idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 186:306–313
Filderman AE, Genovese LA, Lazo JS (1988) Alterations in pulmonary protective enzymes following systemic bleomycin treatment in mice. Biochem Pharmacol 37(6):1111–1116
Gauldie J (2002) Inflammatory mechanisms are a minor component of the pathogenesis of idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 165:1205–1206
Gazdhar A, Fachinger P, Van Leer C, et al. (2007) Gene transfer of hepatocyte growth factor by electroporation reduces bleomycin-induced lung fibrosis. Am J Physiol: Lung Cell Mol Physiol 292:L529–L536
Gilani AH, Aziz N, Khurram IM, et al. (2001) Bronchodilator, spasmolytic and calcium antagonist activities of Nigella sativa seeds (Kalonji): a traditional herbal product with multiple medicinal uses. J Pak Med Assoc 51:115–120
Gross O, Girgert R, Rubel D, et al. (2011) Renal protective effects of aliskiren beyond its antihypertensive property in a mouse model of progressive fibrosis. Am J Hypertens 24:355–361
Halliwell B (2011) Free radicals and antioxidants—quo vadis? Trends Pharmacol Sci 32:125–130
Hartner A, Porst M, Klanke B, et al. (2006) Angiotensin II formation in the kidney and nephrosclerosis in Ren-2 hypertensive rats. Nephrol Dial Transplant 21:1778–1785
Hay J, Shahzeidi S, Laurent G (1991) Mechanisms of bleomycin-induced lung damage. Arch Toxicol 65:81–94
Hudson BI, Bucciarelli LG, Wendt T, et al. (2003) Blockade of receptor for advanced glycation endproducts: a new target for therapeutic intervention in diabetic complications and inflammatory disorders. Arch Biochem Biophys 419(1):80–88
Hyde DM, Giri SN (1990) Polyinosinic–polycytidylic acid, an interferon inducer, ameliorates bleomycin-induced lung fibrosis in mice. Exp Lung Res 16:533–546
Inomata M, Nishioka Y, Azuma A (2015) Nintedanib: evidence for its therapeutic potential in idiopathic pulmonary fibrosis. Core Evid 10:89–98
Izbicki G, Segel MJ, Christensen TG, et al. (2002) Time course of bleomycin-induced lung fibrosis. Int J Exp Pathol 83:111–119
Jensen C, Herold P, Brunner HR (2008) Aliskiren: the first renin inhibitor for clinical treatment. Nat Rev Drug Discov 7:399–410
John M, Oltmanns U, Fietze I, et al. (2002) Increased production of matrix metalloproteinase-2 in alveolar macrophages and regulation by interleukin-10 in patients with acute pulmonary sarcoidosis. Exp Lung Res 28:55–68
Ke CY, Lee CC, Lee CJ, et al. (2010) Aliskiren ameliorates chlorhexidine digluconate induced peritoneal fibrosis in rats. Eur J Clin Investig 40(4):301–309
Keane MP, Strieter RM (2002) The importance of balanced pro-inflammatory and anti-inflammatory mechanisms in diffuse lung disease. Respir Res 3:5
Kim JY, Choeng HC, Ahn C, et al. (2009) Early and late changes of MMP-2 and MMP-9 in bleomycin-induced pulmonary fibrosis. Yonsei Med J 50:68–77
Kinnula VL, Crapo JD (2003) Superoxide dismutases in the lung and human lung diseases. Am J Respir Crit Care Med 167:1600–1619
Kinnula VL, Fattman CL, Tan RJ, et al. (2005) Oxidative stress in pulmonary fibrosis: a possible role for redox modulatory therapy. Am J Respir Crit Care Med 172(4):417–422
Kliment CR, Oury TD (2010) Oxidative stress, extracellular matrix targets, and idiopathic pulmonary fibrosis. Free Rad Biol Med 49:707–717
Kunugi S, Fukuda Y, Ishizaki M, et al. (2001) Role of MMP-2 in alveolar epithelial cell repair after bleomycin administration in rabbits. Lab Investig 81:1309–1318
Latta VD, Cecchettini A, Del Ry S, et al. (2015) Bleomycin in the setting of lung fibrosis induction: from biological mechanisms to counteractions. Pharmacol Res. doi:10.1016/j. phrs.2015.04.012
Lemjabbar H, Gosset P, Lechapt-Zalcman E, et al. (1999) Overexpression of alveolar macrophage gelatinase B (MMP-9) in patients with idiopathic pulmonary fibrosis: effects of steroid and immunosuppressive treatment. Am J Respir Cell Mol Biol 20:903–913
Li X, Rayford H, Uhal BD (2003) Essential roles for angiotensin receptor AT1a in bleomycin-induced apoptosis and lung fibrosis in mice. Am J Pathol 163:2523–2530
Liang X, Tian Q, Wei Z, et al. (2011) Effect of feining on bleomycin induced pulmonary injuries in rats. J Ethno Pharmacol 134(3):971–976
Liu RM, Gaston Pravia KA (2010) Oxidative stress and glutathione in TGF-beta-mediated fibrogenesis. Free Radic Biol Med 48:1–15
Liu G, Cheresh P, Kamp DW (2013) Molecular basis of asbestos-induced lung disease. Annu Rev Pathol Mech Dis 8:161–187
Liu RM, Vayalil PK, Ballinger C, et al. (2012) Transforming growth factor beta suppresses glutamate-cysteine ligase gene expression and induces oxidative stress in a lung fibrosis model. Free Rad Biol Med 53:554–563
Lohwasser C, Neureiter D, Weigle B, et al. (2006) The receptor for advanced glycation end products is highly expressed in the skin and upregulated by advanced glycation end products and tumor necrosis factor-alpha. J Invest Dermatol 126(2):291–299
Marshall RP, Gohlke P, Chambers RC, et al. (2004) Angiotensin II and the fibro proliferative response to acute lung injury. Am J Physiol Lung Cell Mol Physiol 286:L156–L164
Matsuse T, Ohga E, Teramoto S, et al. (1998) Immunohistochemical localization of advanced glycation end products in pulmonary fibrosis. J Clin Pathol 51(7):515–519
McKeown S, Richter AG, O’Kane C, et al. (2009) MMP expression and abnormal lung permeability are important determinants of outcome in IPF. Eur Respir J 33:77–84
Montano M, Ramos C, Gonzalez G, et al. (1989) Lung collagenase inhibitors and spontaneous and latent collagenase activity in idiopathic pulmonary fibrosis and hypersensitivity pneumonitis. Chest 96:1115–1119
Montes E, Ruiz V, Checa M, et al. (2012) Renin is an angiotensin-independent profibrotic mediator: role in pulmonary fibrosis. Eur Respir J 39:141–148
Nelson KK, Melendez JA (2004) Mitochondrial redox control of matrix metalloproteinases. Free Radic Biol Med 37(6):768–784
Ohashi S, Abe H, Takahashi T, et al. (2004) End products increase collagen-specific chaperone protein in mouse diabetic nephropathy. J Biol Chem 279(19):19816–19823
Oury TD, Thakker K, Menache M, et al. (2001) Attenuation of bleomycin-induced pulmonary fibrosis by a catalytic antioxidant metalloporphyrin. Am J Respir Cell Mol Biol 25:164–169
Paino IM, Miranda JC, Marzocchi-Machado CM, et al. (2011) Phagocytosis and nitric oxide levels in rheumatic inflammatory states in elderly women. J Clin Lab Anal 25:47–51
Pardo A, Selman M (2012) Role of matrix metaloproteases in idiopathic pulmonary fibrosis. Fibrogenesis Tissue Repair 5(1):S9
Pardo A, Ruiz V, Arreola JL, et al. (2003) Bleomycin-induced pulmonary fibrosis is attenuated in 훾-glutamyl transpeptidase deficient mice. Am J Respir Crit Care Med 167(6):925–932
Pardo A, Selman M, Ramirez R, et al. (1992) Production of collagenase and tissue inhibitor of metalloproteinases by fibroblasts derived from normal and fibrotic human lung. Chest 102:1085–1089
Perez-Ramos J, Segura-Valdez M, Vanda B, et al. (1999) Matrix metalloproteinases 2, 9, and 13, and tissue inhibitors of metalloproteinases 1 and 2 in experimental lung silicosis. Am J Respir Crit Care Med 160:1274–1282
Raghu G, Collard HR, Egan JJ, et al. (2011) ATS/ERS/JRS/ALAT Committee on Idiopathic Pulmonary Fibrosis An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med 183:788–824
Ramadan MF, Kroh LW, Morsel JT (2003) Radical scavenging activity of black cumin (Nigella sativa L.), coriander (Coriandrum sativum L.), and niger (Guizotia abyssinica Cass.) crude seed oils and oil fractions. J Agric Food Chem 51:6961–6969
Ramasamy R, Vannucci SJ, Yan SS, et al. (2005) Advanced glycation end products and RAGE: a common thread in aging, diabetes, neurodegeneration, and inflammation. Glycobiology 15(7):16R–28R
Rokhsara R, Maya M, Timothy E, et al. (2013) A review of current and novel therapies for idiopathic pulmonary fibrosis. J Thorac Dis 5(1)
Santuzzi CH, Tiradentes RV, Mengal V, et al. (2015) Combined aliskiren and L-arginine treatment has antihypertensive effects and prevents vascular endothelial dysfunction in a model of renovascular hypertension. Braz J Med Biol Res 48(1):65–76
Sasaki M, Kashima M, Ito T, et al. (2000) Differential regulation of metalloproteinase production, proliferation and chemotaxis of human lung fibroblasts by PDGF, interleukin-1beta and TNF-alpha. Mediat Inflamm 9:155–160
Schmidt AM, Yan SD, Wautier JL, et al. (1999) Activation of receptor for advanced glycation end products: a mechanism for chronic vascular dysfunction in diabetic vasculopathy and atherosclerosis. Circ Res 84(5):489–497
Selman M, King TE, Pardo A (2001) Idiopathic pulmonary fibrosis: prevailing evolving hypotheses about its pathogenesis and implications for therapy. Ann Intern Med 134:136–151
Serrano-Mollar A, Closa D, Prats N, et al. (2003) In vivo antioxidant treatment protects against bleomycin-induced lung damage in rats. Br J Pharmacol 138(6):1037–1048
Snider GL, Celli BR, Goldstein RH, et al. (1978) Chronic interstitial pulmonary fibrosis produced in hamsters by endotracheal bleomycin. Lung volumes volume–pressure relations, carbon monoxide uptake, and arterial blood gas studied. Am Rev Respir Dis 117:289–297
Sriram N, Kalayarasan S, Sudhandiran G (2009) Epigallocatechin-3-gallate augments antioxidant activities and inhibits inflammation during bleomycin-induced experimental pulmonary fibrosis through Nrf2-Keap1 signaling. Pulm Pharmacol Ther 22(3):221–236
Tan RJ, Fattman CL, Niehouse LM, et al. (2006) Matrix metalloproteinases promote inflammation and fibrosis in asbestos-induced lung injury in mice. Am J Respir Cell Mol Biol 35(3):289–297
Todd NW, Luzina IG, Atamas SP (2012) Molecular and cellular mechanisms of pulmonary Fibrosis. Fibrogenesis & Tissue Repair 5:11. treatment modality for infection, inflammation and repair. Cell Biochem Funct 22:67–79
Tzurel A, Segel J, Or R, et al. (2002) Halofuginone does not reduce fibrosis in bleomycin-induced lung injury. Life Sci 7(14):1599–1606
Ulrich P, Cerami A (2001) Protein glycation, diabetes, and aging. Recent Prog Horm Res 56:1–21
Virdis A, Ghiadoni L, Qasem A, et al. (2012) Effect of aliskiren treatment on endothelium dependent vasodilation and aortic stiffness in essential hypertensive patients. Eur Heart J 33:1530–1538
Wang Q, Giri SN, Hyde DM, et al. (1991) Amelioration of bleomycin-induced pulmonary fibrosis in hamsters by combined treatment with taurine and niacin. Biochem Pharmacol 42(5):1115–1122
Woessner JF (1994) The family of matrix metalloproteinase family. Ann N Y Acad Sci 732:11–21
Wollin L, Maillet I, Quesniaux V, et al. (2014) Antifibrotic and anti-inflammatory activity of the tyrosine kinase inhibitor nintedanib in experimental models of lung fibrosis. J Pharmacol Exp Ther 349:209–220
Yara S, Kawakami K, Kudeken N, et al. (2001) FTS reduces bleomycin-induced cytokine and chemokine production and inhibits pulmonary fibrosis in mice. Clin Exp Immunol 124:77–85
Yu Q, Stamenkovic I (2000) Cell surface localized matrixmetalloproteinase-9 proteolytically activates TGF-beta and promotes tumor invasion and angiogenesis. Genes Dev 14:163–176
Zhang W, Han Y, Meng G, et al. (2014) Direct renin inhibition with aliskiren protects against myocardial ischemia/reperfusion injury by activating nitric oxide synthase signaling in spontaneously hypertensive rats. J Am Heart Assoc 3:e000606. doi:10.1161/JAHA.113.000606
Zhao L, Wang X, Chang Q, et al. (2010) Neferine, a bisbenzylisoquinline alkaloid attenuates bleomycin-induced pulmonary fibrosis. Eur J Pharmacol 627(1–3):304–312
Zhi H, Luptak I, Alreja G, et al. (2013) Effects of direct renin inhibition on myocardial fibrosis and cardiac fibroblast function. PLoS One 8(12):e81612. doi:10.1371/journal.pone.0081612
Zhou GH, Li C, Cai L (2004) Advanced glycation end products induce connective tissue growth factor-mediated renal fibrosis predominantly through transforming growth factor beta independent pathway. Am J Pathol 165(6):2033–2043
Acknowledgments
This research was supported by the Medical Research Service of Ain Shams University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Abuelezz, S.A., Hendawy, N. & Osman, W.M. Aliskiren attenuates bleomycin-induced pulmonary fibrosis in rats: focus on oxidative stress, advanced glycation end products, and matrix metalloproteinase-9. Naunyn-Schmiedeberg's Arch Pharmacol 389, 897–909 (2016). https://doi.org/10.1007/s00210-016-1253-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-016-1253-3