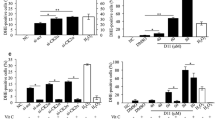
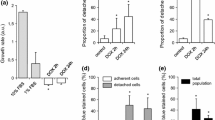
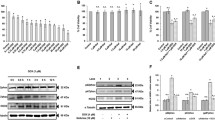
Abstract
The adaptor protein p66Shc modulates cellular redox status integrating oxidative stress with mitochondrial stress responses. Upon oxidative stress, p66Shc is translocated to mitochondria or mitochondria-associated membranes in a multi-step process, resulting in locally increased reactive oxygen species production. This signaling pathway is believed to be important in the context of drug-induced organ toxicity. The use of anthracyclines as anticancer agents is limited due to a dose-dependent and cumulative toxicity resulting in cardiomyopathy. Treatment with the anthracycline doxorubicin (DOX) results in a dose-dependent and cumulative cardiotoxicity which is mediated, at least in part, by increased oxidative stress. In the present study, we investigated for the first time whether p66Shc signaling is activated during DOX treatment of the rat cardiomyoblast H9c2 cell line. We further tested whether the transcriptional factor FoxO3a, which activates target genes responsible for apoptosis and cell cycle arrest, is also involved in p66Shc-dependent redox signaling pathway. Our results suggest that DOX treatment induces p66Shc protein up-regulation specifically in nuclear fractions. Increased nuclear expression of FoxO3a was also detected in H9c2 cells after DOX treatment. Treatment with the antioxidant and protein kinase C (PKC-β) inhibitor hispidin decreased DOX-induced activation of caspase 9 and p66Shc alterations. Taking together, we hypothesize that p66Shc signaling is involved in the activation of stress/toxicity responses elicited by the treatment of H9c2 cells with DOX. Hence, the selective inhibition of this redox pathway may be a promising therapeutic approach to circumvent DOX cardiotoxicity.






Similar content being viewed by others
References
Altieri P, Spallarossa P, Barisione C et al (2012) Inhibition of doxorubicin-induced senescence by PPARdelta activation agonists in cardiac muscle cells: cooperation between PPARdelta and Bcl6. PLoS One 7(9):e46126. doi:10.1371/journal.pone.0046126
Awadh Ali NA, Mothana RA, Lesnau A, Pilgrim H, Lindequist U (2003) Antiviral activity of Inonotus hispidus. Fitoterapia 74(5):483–485
Berthiaume JM, Wallace KB (2007) Persistent alterations to the gene expression profile of the heart subsequent to chronic doxorubicin treatment. Cardiovasc Toxicol 7(3):178–191. doi:10.1007/s12012-007-0026-0
Biggs WH 3rd, Meisenhelder J, Hunter T, Cavenee WK, Arden KC (1999) Protein kinase B/Akt-mediated phosphorylation promotes nuclear exclusion of the winged helix transcription factor FKHR1. Proc Natl Acad Sci USA 96(13):7421–7426
Bonora M, Wieckowski MR, Chinopoulos C et al (2015) Molecular mechanisms of cell death: central implication of ATP synthase in mitochondrial permeability transition. Oncogene 34(12):1475–1486. doi:10.1038/onc.2014.96
Brown JE, Zeiger SL, Hettinger JC et al (2010) Essential role of the redox-sensitive kinase p66shc in determining energetic and oxidative status and cell fate in neuronal preconditioning. J Neurosci 30(15):5242–5252. doi:10.1523/JNEUROSCI.6366-09.2010
Brunet A, Bonni A, Zigmond MJ et al (1999) Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96(6):857–868
Brunet A, Datta SR, Greenberg ME (2001) Transcription-dependent and -independent control of neuronal survival by the PI3K-Akt signaling pathway. Curr Opin Neurobiol 11(3):297–305
Calnan DR, Brunet A (2008) The FoxO code. Oncogene 27(16):2276–2288. doi:10.1038/onc.2008.21
Carvalho FS, Burgeiro A, Garcia R, Moreno AJ, Carvalho RA, Oliveira PJ (2014) Doxorubicin-induced cardiotoxicity: from bioenergetic failure and cell death to cardiomyopathy. Med Res Rev 34(1):106–135. doi:10.1002/med.21280
Chahdi A, Sorokin A (2008) Endothelin-1 couples betaPix to p66Shc: role of betaPix in cell proliferation through FOXO3a phosphorylation and p27kip1 down-regulation independently of Akt. Mol Biol Cell 19(6):2609–2619. doi:10.1091/mbc.E07-05-0424
De Angelis A, Piegari E, Cappetta D et al (2010) Anthracycline cardiomyopathy is mediated by depletion of the cardiac stem cell pool and is rescued by restoration of progenitor cell function. Circulation 121(2):276–292. doi:10.1161/CIRCULATIONAHA.109.895771
De Angelis A, Piegari E, Cappetta D et al (2015) SIRT1 activation rescues doxorubicin-induced loss of functional competence of human cardiac progenitor cells. Int J Cardiol 189:30–44. doi:10.1016/j.ijcard.2015.03.438
De Marchi E, Baldassari F, Bononi A, Wieckowski MR, Pinton P (2013) Oxidative stress in cardiovascular diseases and obesity: role of p66Shc and protein kinase C. Oxid Med Cell Longev 2013:564961. doi:10.1155/2013/564961
Dodd DA, Atkinson JB, Olson RD et al (1993) Doxorubicin cardiomyopathy is associated with a decrease in calcium release channel of the sarcoplasmic reticulum in a chronic rabbit model. J Clin Invest 91(4):1697–1705. doi:10.1172/JCI116379
Essers MA, Weijzen S, de Vries-Smits AM et al (2004) FOXO transcription factor activation by oxidative stress mediated by the small GTPase Ral and JNK. EMBO J 23(24):4802–4812. doi:10.1038/sj.emboj.7600476
Gambino V, De Michele G, Venezia O et al (2013) Oxidative stress activates a specific p53 transcriptional response that regulates cellular senescence and aging. Aging Cell 12(3):435–445. doi:10.1111/acel.12060
Gilda JE, Gomes AV (2013) Stain-Free total protein staining is a superior loading control to beta-actin for Western blots. Anal Biochem 440(2):186–188. doi:10.1016/j.ab.2013.05.027
Giorgio M, Migliaccio E, Orsini F et al (2005) Electron transfer between cytochrome c and p66Shc generates reactive oxygen species that trigger mitochondrial apoptosis. Cell 122(2):221–233. doi:10.1016/j.cell.2005.05.011
Gomes AR, Brosens JJ, Lam EW (2008) Resist or die: FOXO transcription factors determine the cellular response to chemotherapy. Cell Cycle 7(20):3133–3136
Gonindard C, Bergonzi C, Denier C et al (1997) Synthetic hispidin, a PKC inhibitor, is more cytotoxic toward cancer cells than normal cells in vitro. Cell Biol Toxicol 13(3):141–153
Greer EL, Brunet A (2005) FOXO transcription factors at the interface between longevity and tumor suppression. Oncogene 24(50):7410–7425. doi:10.1038/sj.onc.1209086
Guo R, Wu K, Chen J et al (2013) Exogenous hydrogen sulfide protects against doxorubicin-induced inflammation and cytotoxicity by inhibiting p38MAPK/NFkappaB pathway in H9c2 cardiac cells. Cell Physiol Biochem 32(6):1668–1680. doi:10.1159/000356602
Ho KK, McGuire VA, Koo CY et al (2012) Phosphorylation of FOXO3a on Ser-7 by p38 promotes its nuclear localization in response to doxorubicin. J Biol Chem 287(2):1545–1555. doi:10.1074/jbc.M111.284224
Houghton P, Fang R, Techatanawat I, Steventon G, Hylands PJ, Lee CC (2007) The sulphorhodamine (SRB) assay and other approaches to testing plant extracts and derived compounds for activities related to reputed anticancer activity. Methods 42(4):377–387. doi:10.1016/j.ymeth.2007.01.003
Jung JY, Lee IK, Seok SJ, Lee HJ, Kim YH, Yun BS (2008) Antioxidant polyphenols from the mycelial culture of the medicinal fungi Inonotus xeranticus and Phellinus linteus. J Appl Microbiol 104(6):1824–1832. doi:10.1111/j.1365-2672.2008.03737.x
Konorev EA, Vanamala S, Kalyanaraman B (2008) Differences in doxorubicin-induced apoptotic signaling in adult and immature cardiomyocytes. Free Radic Biol Med 45(12):1723–1728. doi:10.1016/j.freeradbiomed.2008.09.006
Lebiedzinska M, Duszynski J, Rizzuto R, Pinton P, Wieckowski MR (2009) Age-related changes in levels of p66Shc and serine 36-phosphorylated p66Shc in organs and mouse tissues. Arch Biochem Biophys 486(1):73–80. doi:10.1016/j.abb.2009.03.007
Lee V, Randhawa AK, Singal PK (1991) Adriamycin-induced myocardial dysfunction in vitro is mediated by free radicals. Am J Physiol 261(4 Pt 2):H989–H995
Luzi L, Confalonieri S, Di Fiore PP, Pelicci PG (2000) Evolution of Shc functions from nematode to human. Curr Opin Genet Dev 10(6):668–674
Mandziuk S, Baj T, Sieniawska E et al (2014) Protective effect of Mutellina purpurea polyphenolic compounds in doxorubicin-induced toxicity in H9c2 cardiomyocytes. Drug Chem Toxicol. doi:10.3109/01480545.2014.893443
Maruyama S, Shibata R, Ohashi K et al (2011) Adiponectin ameliorates doxorubicin-induced cardiotoxicity through Akt protein-dependent mechanism. J Biol Chem 286(37):32790–32800. doi:10.1074/jbc.M111.245985
Migliaccio E, Mele S, Salcini AE et al (1997) Opposite effects of the p52shc/p46shc and p66shc splicing isoforms on the EGF receptor-MAP kinase-fos signalling pathway. EMBO J 16(4):706–716. doi:10.1093/emboj/16.4.706
Migliaccio E, Giorgio M, Pelicci PG (2013) p53 and aging: role of p66Shc. Aging (Albany NY) 5(7):488–489
Momparler RL, Karon M, Siegel SE, Avila F (1976) Effect of adriamycin on DNA, RNA, and protein synthesis in cell-free systems and intact cells. Cancer Res 36(8):2891–2895
Moreira AC, Branco AF, Sampaio SF et al (2014) Mitochondrial apoptosis-inducing factor is involved in doxorubicin-induced toxicity on H9c2 cardiomyoblasts. Biochim Biophys Acta 1842(12 Pt A):2468–2478. doi:10.1016/j.bbadis.2014.09.015
Nemoto S, Finkel T (2002) Redox regulation of forkhead proteins through a p66shc-dependent signaling pathway. Science 295(5564):2450–2452. doi:10.1126/science.1069004
Orsini F, Migliaccio E, Moroni M et al (2004) The life span determinant p66Shc localizes to mitochondria where it associates with mitochondrial heat shock protein 70 and regulates trans-membrane potential. J Biol Chem 279(24):25689–25695. doi:10.1074/jbc.M401844200
Park IH, Chung SK, Lee KB et al (2004) An antioxidant hispidin from the mycelial cultures of Phellinus linteus. Arch Pharm Res 27(6):615–618
Patrussi L, Savino MT, Pellegrini M et al (2005) Cooperation and selectivity of the two Grb2 binding sites of p52Shc in T-cell antigen receptor signaling to Ras family GTPases and Myc-dependent survival. Oncogene 24(13):2218–2228. doi:10.1038/sj.onc.1208384
Pereira GC, Silva AM, Diogo CV, Carvalho FS, Monteiro P, Oliveira PJ (2011) Drug-induced cardiac mitochondrial toxicity and protection: from doxorubicin to carvedilol. Curr Pharm Des 17(20):2113–2129
Piegari E, De Angelis A, Cappetta D et al (2013) Doxorubicin induces senescence and impairs function of human cardiac progenitor cells. Basic Res Cardiol 108(2):334. doi:10.1007/s00395-013-0334-4
Raffaello A, Rizzuto R (2011) Mitochondrial longevity pathways. Biochim Biophys Acta 1813(1):260–268. doi:10.1016/j.bbamcr.2010.10.007
Romero-Calvo I, Ocon B, Martinez-Moya P et al (2010) Reversible Ponceau staining as a loading control alternative to actin in Western blots. Anal Biochem 401(2):318–320. doi:10.1016/j.ab.2010.02.036
Sardao VA, Oliveira PJ, Holy J, Oliveira CR, Wallace KB (2009a) Doxorubicin-induced mitochondrial dysfunction is secondary to nuclear p53 activation in H9c2 cardiomyoblasts. Cancer Chemother Pharmacol 64(4):811–827. doi:10.1007/s00280-009-0932-x
Sardao VA, Oliveira PJ, Holy J, Oliveira CR, Wallace KB (2009b) Morphological alterations induced by doxorubicin on H9c2 myoblasts: nuclear, mitochondrial, and cytoskeletal targets. Cell Biol Toxicol 25(3):227–243. doi:10.1007/s10565-008-9070-1
Savino C, Pelicci P, Giorgio M (2013) The P66Shc/mitochondrial permeability transition pore pathway determines neurodegeneration. Oxid Med Cell Longev 2013:719407. doi:10.1155/2013/719407
Singh SB, Jayasuriya H, Dewey R et al (2003) Isolation, structure, and HIV-1-integrase inhibitory activity of structurally diverse fungal metabolites. J Ind Microbiol Biot 30(12):721–731. doi:10.1007/s10295-003-0101-x
Solem LE, Henry TR, Wallace KB (1994) Disruption of mitochondrial calcium homeostasis following chronic doxorubicin administration. Toxicol Appl Pharmacol 129(2):214–222. doi:10.1006/taap.1994.1246
Spallarossa P, Altieri P, Aloi C et al (2009) Doxorubicin induces senescence or apoptosis in rat neonatal cardiomyocytes by regulating the expression levels of the telomere binding factors 1 and 2. Am J Physiol Heart Circ Physiol 297(6):H2169–H2181. doi:10.1152/ajpheart.00068.2009
Trinei M, Giorgio M, Cicalese A et al (2002) A p53-p66Shc signalling pathway controls intracellular redox status, levels of oxidation-damaged DNA and oxidative stress-induced apoptosis. Oncogene 21(24):3872–3878. doi:10.1038/sj.onc.1205513
Vindis C, Cerretti DP, Daniel TO, Huynh-Do U (2003) EphB1 recruits c-Src and p52Shc to activate MAPK/ERK and promote chemotaxis. J Cell Biol 162(4):661–671. doi:10.1083/jcb.200302073
Yoshida S, Masaki T, Feng H et al (2004) Enhanced expression of adaptor molecule p46 Shc in nuclei of hepatocellular carcinoma cells: study of LEC rats. Int J Oncol 25(4):1089–1096
Zhou S, Palmeira CM, Wallace KB (2001) Doxorubicin-induced persistent oxidative stress to cardiac myocytes. Toxicol Lett 121(3):151–157
Acknowledgments
This work is funded by FEDER funds through the Operational Programme Competitiveness Factors—COMPETE and national funds by FCT—Foundation for Science and Technology under the Project (PTDC/DTP-FTO/1180/2012) and strategic project UID/NEU/04539/2013. Also supported by QREN project 4832 with reference CENTRO-07-ST24-FEDER-002008 financed through FEDER. The FCT also sponsored IVN through FCT Researcher funding (IF/01316/2014). AW and MRW were supported by Polish Ministry of Science and Higher Education (W100/HFSC/2011) and Grant HFSP RGP0027/2011.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The manuscript does not contain clinical studies or patient data. The authors declare they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sampaio, S.F., Branco, A.F., Wojtala, A. et al. p66Shc signaling is involved in stress responses elicited by anthracycline treatment of rat cardiomyoblasts. Arch Toxicol 90, 1669–1684 (2016). https://doi.org/10.1007/s00204-015-1583-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-015-1583-9