Abstract
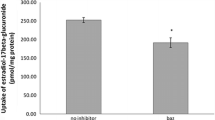
Soy isoflavones (IF) are phytoestrogens, which interact with estrogen receptors. They are extensively metabolized by glucuronosyltransferases and sulfotransferases, leading to the modulation of their estrogenic activity. It can be assumed that this biotransformation also has a crucial impact on the uptake of IF by active or passive cellular transport mechanisms, but little is known about the transport of IF phase II metabolites into the cell. Therefore, transport assays for phase II metabolites of daidzein (DAI) were carried out using HEK293 cell lines transfected with five human candidate carriers, i.e., organic anion transporter OAT4, sodium-dependent organic anion transporter (SOAT), Na+-taurocholate cotransporting polypeptide (NTCP), apical sodium-dependent bile acid transporter ASBT, and organic anion transporting polypeptide OATP2B1. Cellular uptake was monitored by UHPLC-DAD. DAI monosulfates were transported by the carriers NTCP and SOAT in a sodium-dependent manner, while OAT4-HEK293 cells revealed a partly sodium-dependent transport for these compounds. In contrast, DAI-7,4′-disulfate was only taken up by NTCP-HEK293 cells. DAI-7-glucuronide, but not DAI-4′-glucuronide, was transported exclusively by OATP2B1 in a sodium-independent manner. DAI-7-glucuronide-4′-sulfate, DAI-7-glucoside, and DAI were no substrate of any of the tested carriers. In addition, the inhibitory potency of the DAI metabolites toward estrone-sulfate (E1S) uptake of the above-mentioned carriers was determined. In conclusion, human SOAT, NTCP, OATP2B1, and OAT4 were identified as carriers for the DAI metabolites. Several metabolites were able to inhibit carrier-dependent E1S uptake. These findings might contribute to a better understanding of the bioactivity of IF especially in case of hormone-related cancers.





Similar content being viewed by others
Abbreviations
- ASBT:
-
Apical sodium-dependent bile acid transporter
- DAI:
-
Daidzein
- DHEAS:
-
Dehydroepiandrosterone sulfate
- DS:
-
Disulfate
- E1S:
-
Estrone-sulfate
- E2S:
-
17β-Estradiol-3-sulfate
- G:
-
Glucoside
- GA:
-
Glucuronide
- GEN:
-
Genistein
- GLY:
-
Glycitein
- IF:
-
Isoflavones
- NTCP:
-
Na+-taurocholate cotransporting polypeptide
- OAT:
-
Organic anion transporter
- OATP:
-
Organic anion transporting polypeptide
- S:
-
Sulfate
- SOAT:
-
Sodium-dependent organic anion transporter
- STS:
-
Steroid sulfatase
References
Al Sarakbi W, Mokbel R, Salhab M, Jiang WG, Reed MJ, Mokbel K (2006) The role of STS and OATP-B mRNA expression in predicting the clinical outcome in human breast cancer. Anticancer Res 26:4985–4990
Allred CD, Ju YH, Allred KF, Chang J, Helferich WG (2001) Dietary genistin stimulates growth of estrogen-dependent breast cancer tumors similar to that observed with genistein. Carcinogenesis 22:1667–1673
Bolca S, Urpi-Sarda M, Blondeel P, Roche N, Vanhaecke L, Possemiers S, Al-Maharik N, Botting N, De Keukeleire D, Bracke M, Heyerick A, Manach C, Depypere H (2010) Disposition of soy isoflavones in normal human breast tissue. Am J Clin Nutr 91:976–984
Brant SR, Yun CH, Donowitz M, Tse CM (1995) Cloning, tissue distribution, and functional analysis of the human Na+/N+ exchanger isoform, NHE3. Am J Physiol 269:C198–C206
Cha SH, Sekine T, Kusuhara H, Yu E, Kim JY, Kim DK, Sugiyama Y, Kanai Y, Endou H (2000) Molecular cloning and characterization of multispecific organic anion transporter 4 expressed in the placenta. J Biol Chem 275:4507–4512
Craddock AL, Love MW, Daniel RW, Kirby LC, Walters HC, Wong MH, Dawson PA (1998) Expression and transport properties of the human ileal and renal sodium-dependent bile acid transporter. Am J Physiol 274:G157–G169
Fietz D, Bakhaus K, Wapelhorst B, Grosser G, Gunther S, Alber J, Doring B, Kliesch S, Weidner W, Galuska CE, Hartmann MF, Wudy SA, Bergmann M, Geyer J (2013) Membrane transporters for sulfated steroids in the human testis—cellular localization, expression pattern and functional analysis. PLoS One 8:e62638
Geisler J (2003) Breast cancer tissue estrogens and their manipulation with aromatase inhibitors and inactivators. J Steroid Biochem Mol Biol 86:245–253
Geyer J, Wilke T, Petzinger E (2006) The solute carrier family SLC10: more than a family of bile acid transporters regarding function and phylogenetic relationships. Naunyn Schmiedebergs Arch Pharmacol 372:413–431
Geyer J, Doring B, Meerkamp K, Ugele B, Bakhiya N, Fernandes CF, Godoy JR, Glatt H, Petzinger E (2007) Cloning and functional characterization of human sodium-dependent organic anion transporter (SLC10A6). J Biol Chem 282:19728–19741
Grosser G, Fietz D, Gunther S, Bakhaus K, Schweigmann H, Ugele B, Brehm R, Petzinger E, Bergmann M, Geyer J (2013) Cloning and functional characterization of the mouse sodium-dependent organic anion transporter Soat (Slc10a6). J Steroid Biochem Mol Biol 138:90–99
Grube M, Kock K, Karner S, Reuther S, Ritter CA, Jedlitschky G, Kroemer HK (2006) Modification of OATP2B1-mediated transport by steroid hormones. Mol Pharmacol 70:1735–1741
Grube M, Reuther S, Meyer Zu Schwabedissen H, Kock K, Draber K, Ritter CA, Fusch C, Jedlitschky G, Kroemer HK (2007) Organic anion transporting polypeptide 2B1 and breast cancer resistance protein interact in the transepithelial transport of steroid sulfates in human placenta. Drug Metab Dispos 35:30–35
Hagenbuch B, Meier PJ (1994) Molecular cloning, chromosomal localization, and functional characterization of a human liver Na+/bile acid cotransporter. J Clin Investig 93:1326–1331
Hagos Y, Stein D, Ugele B, Burckhardt G, Bahn A (2007) Human renal organic anion transporter 4 operates as an asymmetric urate transporter. J Am Soc Nephrol 18:430–439
Hilakivi-Clarke L, Andrade JE, Helferich W (2010) Is soy consumption good or bad for the breast? J Nutr 140:2326S–2334S
Hosoda K, Furuta T, Ishii K (2011) Metabolism and disposition of isoflavone conjugated metabolites in humans after ingestion of kinako. Drug Metab Dispos 39:1762–1767
Imai Y, Tsukahara S, Asada S, Sugimoto Y (2004) Phytoestrogens/flavonoids reverse breast cancer resistance protein/ABCG2-mediated multidrug resistance. Cancer Res 64:4346–4352
Kijkuokool P, Parhar IS, Malaivijitnond S (2006) Genistein enhances N-nitrosomethylurea-induced rat mammary tumorigenesis. Cancer Lett 242:53–59
Kinjo J, Tsuchihashi R, Morito K, Hirose T, Aomori T, Nagao T, Okabe H, Nohara T, Masamune Y (2004) Interactions of phytoestrogens with estrogen receptors a and b (III). Estrogenic activities of soy isoflavone aglycones and their metabolites isolated from human urine. Biol Pharm Bull 27:185–188
Kobayashi D, Nozawa T, Imai K, Nezu J, Tsuji A, Tamai I (2003) Involvement of human organic anion transporting polypeptide OATP-B (SLC21A9) in pH-dependent transport across intestinal apical membrane. J Pharmacol Exp Ther 306:703–708
Koenen A, Kock K, Keiser M, Siegmund W, Kroemer HK, Grube M (2012) Steroid hormones specifically modify the activity of organic anion transporting polypeptides 2852. Eur J Pharm Sci 47:774–780
Kramer W, Stengelin S, Baringhaus KH, Enhsen A, Heuer H, Becker W, Corsiero D, Girbig F, Noll R, Weyland C (1999) Substrate specificity of the ileal and the hepatic Na(+)/bile acid cotransporters of the rabbit. I. Transport studies with membrane vesicles and cell lines expressing the cloned transporters. J Lipid Res 40:1604–1617
Kullak-Ublick GA, Ismair MG, Stieger B, Landmann L, Huber R, Pizzagalli F, Fattinger K, Meier PJ, Hagenbuch B (2001) Organic anion-transporting polypeptide B (OATP-B) and its functional comparison with three other OATPs of human liver. Gastroenterology 120:525–533
Lang K, Wagner C, Haddad G, Burnekova O, Geibel J (2003) Intracellular pH activates membrane-bound Na(+)/H(+) exchanger and vacuolar H(+)-ATPase in human embryonic kidney (HEK) cells. Cell Physiol Biochem 13:257–262
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Magee PJ, Rowland I (2012) Soy products in the management of breast cancer. Curr Opin Clin Nutr Metab Care 15:586–591
Meier PJ, Stieger B (2002) Bile salt transporters. Annu Rev Physiol 64:635–661
Mortensen A, Kulling SE, Schwartz H, Rowland I, Ruefer CE, Rimbach G, Cassidy A, Magee P, Millar J, Hall WL, Birkved FK, Sorensen IK, Sontag G (2009) Analytical and compositional aspects of isoflavones in food and their biological effects. Mol Nutr Food Res 53:S266–S309
Nishimura M, Naito S (2005) Tissue-specific mRNA expression profiles of human ATP-binding cassette and solute carrier transporter superfamilies. Drug Metab Pharmacokinet 20:452–477
Pasqualini JR, Chetrite GS (2005) Recent insight on the control of enzymes involved in estrogen formation and transformation in human breast cancer. J Steroid Biochem Mol Biol 93:221–236
Pizzagalli F, Varga Z, Huber RD, Folkers G, Meier PJ, St-Pierre MV (2003) Identification of steroid sulfate transport processes in the human mammary gland. J Clin Endocrinol Metab 88:3902–3912
Pugazhendhi D, Watson KA, Mills S, Botting N, Pope GS, Darbre PD (2008) Effect of sulphation on the oestrogen agonist activity of the phytoestrogens genistein and daidzein in MCF-7 human breast cancer cells. J Endocrinol 197:503–515
Reed MJ, Purohit A, Woo LW, Newman SP, Potter BV (2005) Steroid sulfatase: molecular biology, regulation, and inhibition. Endocr Rev 26:171–202
Ritchie MR, Cummings JH, Morton MS, Steel CM, Bolton-Smith C, Riches AC (2006) A newly constructed and validated isoflavone database for the assessment of total genistein and daidzein intake. Br J Nutr 95:204–213
Roth M, Obaidat A, Hagenbuch B (2012) OATPs, OATs and OCTs: the organic anion and cation transporters of the SLCO and SLC22A gene superfamilies. Br J Pharmacol 165:1260–1287
Rüfer CE, Bub A, Moseneder J, Winterhalter P, Sturtz M, Kulling SE (2008) Pharmacokinetics of the soybean isoflavone daidzein in its aglycone and glucoside form: a randomized, double-blind, crossover study. Am J Clin Nutr 87:1314–1323
Santner SJ, Feil PD, Santen RJ (1984) In situ estrogen production via the estrone sulfatase pathway in breast tumors: relative importance versus the aromatase pathway. J Clin Endocrinol Metab 59:29–33
Schroeder A, Eckhardt U, Stieger B, Tynes R, Schteingart CD, Hofmann AF, Meier PJ, Hagenbuch B (1998) Substrate specificity of the rat liver Na(+)-bile salt cotransporter in Xenopus laevis oocytes and in CHO cells. Am J Physiol 274:G370–G375
Schweigmann H, Sanchez-Guijo A, Ugele B, Hartmann K, Hartmann MF, Bergmann M, Pfarrer C, Doring B, Wudy SA, Petzinger E, Geyer J, Grosser G (2014) Transport of the placental estriol precursor 16alpha-hydroxy-dehydroepiandrosterone sulfate (16alpha-OH-DHEAS) by stably transfected OAT4-, SOAT-, and NTCP-HEK293 cells. J Steroid Biochem Mol Biol 143:259–265. doi:10.1016/j.jsbmb.2014.03.013
Selcer KW, Kabler H, Sarap J, Xiao Z, Li PK (2002) Inhibition of steryl sulfatase activity in LNCaP human prostate cancer cells. Steroids 67:821–826
Sfakianos J, Coward L, Kirk M, Barnes S (1997) Intestinal uptake and biliary excretion of the isoflavone genistein in rats. J Nutr 127:1260–1268
Shneider BL, Dawson PA, Christie DM, Hardikar W, Wong MH, Suchy FJ (1995) Cloning and molecular characterization of the ontogeny of a rat ileal sodium-dependent bile acid transporter. J Clin Investig 95:745–754
Soukup ST, Al-Maharik N, Botting N, Kulling SE (2014) Quantification of soy isoflavones and their conjugative metabolites in plasma and urine: an automated and validated UHPLC-MS/MS method for use in large-scale studies. Anal Bioanal Chem 406:6007–6020
St-Pierre MV, Hagenbuch B, Ugele B, Meier PJ, Stallmach T (2002) Characterization of an organic anion-transporting polypeptide (OATP-B) in human placenta. J Clin Endocrinol Metab 87:1856–1863
Tamai I, Nezu J, Uchino H, Sai Y, Oku A, Shimane M, Tsuji A (2000) Molecular identification and characterization of novel members of the human organic anion transporter (OATP) family. Biochem Biophys Res Commun 273:251–260
Trauner M, Boyer JL (2003) Bile salt transporters: molecular characterization, function, and regulation. Physiol Rev 83:633–671
Ugele B, Bahn A, Rex-Haffner M (2008) Functional differences in steroid sulfate uptake of organic anion transporter 4 (OAT4) and organic anion transporting polypeptide 2B1 (OATP2B1) in human placenta. J Steroid Biochem Mol Biol 111:1–6
Wietrzyk J, Mazurkiewicz M, Madej J, Dzimira S, Grynkiewicz G, Radzikowski C, Opolski A (2004) Genistein alone or combined with cyclophosphamide may stimulate 16/C transplantable mouse mammary cancer growth. Med Sci Monitor 10:BR414–BR419
Wong MH, Oelkers P, Dawson PA (1995) Identification of a mutation in the ileal sodium-dependent bile acid transporter gene that abolishes transport activity. J Biol Chem 270:27228–27234
Wong CC, Botting NP, Orfila C, Al-Maharik N, Williamson G (2011) Flavonoid conjugates interact with organic anion transporters (OATs) and attenuate cytotoxicity of adefovir mediated by organic anion transporter 1 (OAT1/SLC22A6). Biochem Pharmacol 81:942–949
Zamora-Ros R, Ferrari P, González C, Tjønneland A, Olsen A, Bredsdorff L, Overvad K, Touillaud M, Perquier F, Fagherazzi G, Lukanova A, Tikk K, Aleksandrova K, Boeing H, Trichopoulou A, Trichopoulos D, Dilis V, Masala G, Sieri S, Mattiello A, Tumino R, Ricceri F, Bueno-de-Mesquita HB, Peeters PM, Weiderpass E, Skeie G, Engeset D, Menéndez V, Travier N, Molina-Montes E, Amiano P, Chirlaque M-D, Barricarte A, Wallström P, Sonestedt E, Sund M, Landberg R, Khaw K-T, Wareham N, Travis R, Scalbert A, Ward H, Riboli E, Romieu I (2013) Dietary flavonoid and lignan intake and breast cancer risk according to menopause and hormone receptor status in the European Prospective Investigation into Cancer and Nutrition (EPIC) Study. Breast Cancer Res Treat 139:163–176
Zhang Y, Kang H, Li B, Zhang R (2012) Positive effects of soy isoflavone food on survival of breast cancer patients in China. Asian Pac J Cancer Prev 13:479–482
Acknowledgments
We want to thank Klaus Schuh for his skillful technical support. Parts of the work were funded by the German Research Foundation (DFG), Grant KU-1079/10-1. The project is part of the collaborative research project entitled IsoCross “Isoflavones: Cross-species comparison on metabolism, estrogen sensitivity, epigenetics, and carcinogenesis”.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grosser, G., Döring, B., Ugele, B. et al. Transport of the soy isoflavone daidzein and its conjugative metabolites by the carriers SOAT, NTCP, OAT4, and OATP2B1. Arch Toxicol 89, 2253–2263 (2015). https://doi.org/10.1007/s00204-014-1379-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-014-1379-3