Abstract
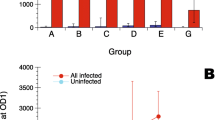
Erysipelothrix rhusiopathiae is the causative agent of erysipelas, a disease of many mammalian and avian species, mainly swine and turkeys. In cetaceans, erysipelas is considered to be the most common infection in juvenile individuals, which have not been vaccinated. Moreover, the disease manifest in both forms, the dermatologic and the acute septicemic forms, has been reported in various species of dolphins and whales. It is difficult to diagnose erysipelas by currently available approaches. Moreover, it is mainly based on culture methods and also PCR methods, which are currently being developed. At the present stage, prophylactic approaches are based on antibiotic therapy and vaccination mostly with porcine erysipelas vaccines. In the present study, an Indirect Immuno Fluorescence method for the detection of dolphin antibodies levels against E. rhusiopathiae was developed and applied in two different groups of captive bottlenose dolphins (Tursiops truncatus) from Loro Parque (Tenerife, Canary Islands, Spain) and L’Oceanogràfic de Valencia (Valencia, Spain) in order to check the tittering levels of antibodies after application of porcine erysipelas vaccines in the studied dolphins.
Similar content being viewed by others
References
Bernal-Guadarrama MJ, Salichs J, Almunia J, García-Parraga D, Fernández-Gallardo N, Santana-Morales MÁ, Pacheco V, Afonso-Lehmann RN, Déniz D, Lorenzo- Morales J, Valladares B, Martínez-Carretero E (2014) Development of an indirect immunofluorescence technique for the diagnosis of toxoplasmosis in bottlenose dolphins. Parasitol Res 113:451–455
Chastel C, Masure O, Balouet G, Laban P, Lucas A (1975) The student, the cetacean and swine-fever: a minor epidemic after dissection of a globicephale. Nouv Presse Med 4:1803–1805
Dierauf LA and Gulland FMD (2001). CRC Handbook of marine mammal medicine. (2nd Edn), CRC Press, New York, USA, pp: 895–906, 703–727
Dunn JL, Buck JD, Robeck TR (2001) Bacterial diseases of cetaceans and pinnipeds. In: Dierauf LA, Gulland FMD (eds) Handbook of marine mammal medicine: health, disease and rehabilitation, 2nd edn. CRC Press, Boca Raton, pp 312–319
Groschup MH, Timoney JF (1990) Modified Feist broth as a serum-free alternative for enhanced production of protective antigen of Erysipelothrix rhusiopathiae. J Clin Microbiol 28:2573–2575
Higgins R (2000) Bacteria and fungi of marine mammals: a review. Can Vet J 41:105–116
Hunt TD, Ziccardi MH, Gulland FMD, Yochem PK, Hird DW, Rowles T, Mazet JAK (2008) Health risks for marine mammal workers. Dis Aquat Org 81:81–92
Kinsel MJ, Boehm JR, Harris B, Murnane RD (1997) Fatal Erysipelothrix rhusiopathiae septicaemia in a captive Pacific white-sided dolphin. J Zoo Wildl Med 28:494–497
Lacave G, Cox E, Hermans J, Devriese L, Goddeeris BM (2001) Induction of cross- protection in mice against dolphin Erysipelothrix rhusiopathiae isolates with a swine commercial vaccine. Vet Microbiol 80:247–253
Lauckner G (1985) Diseases of Mammalia: Pinnipedia, Vol IV. In: Kinne O (ed) Diseases of marine animals. Biologische Anstalt Helgoland, Hamburg, pp 695–707
Leslie A, Gulland FMD (2001) CRC handbook of marine mammal medicine. CRC Press LLC, Boca Raton
Melero M, Rubio-Guerri C, Crespo JL, Arbelo M, Vela AI, García-Párraga D, Sierra E, Domínguez L, Sánchez-Vizcaíno JM (2011) First case of erysipelas in a free- ranging bottlenose dolphin (Tursiops truncatus) stranded in the Mediterranean Sea. Dis Aquat Organ. 97(2):167–170
Opriessnig T, Shen HG, Bender JS, Boehm JR, Halbur PG (2013) Erysipelothrix rhusiopathiae isolates recovered from fish, a harbour seal (Phoca vitulina) and the marine environment are capable of inducing characteristic cutaneous lesions in pigs. J Comp Pathol 148(4):365–372
Seibold HR, Neal JE (1956) Erysipelothrix septicaemia in the porpoise. J Am Vet Med Assoc 1956:537–539
Shimoji Y, Mori Y, Hyakutake K, Sekizaki T, Yokomizo Y (1998a) Use of an enrichment broth cultivation-PCR combination assay for rapid diagnosis of swine erysipelas. J Clin Microbiol 36:86–89
Shimoji Y, Mori Y, Sekizaki T, Shibahara T, Yokomizo Y (1998b) Construction and vaccine potential of acapsular mutants of Erysipelothrix rhusiopathiae: use of excision of Tn916 to inactivate a target gene. Infect Immun 66:3250–3254
Sitt T, Bowen L, Blanchard MT, Gershwin LJ, Byrne BA, Dold C, McBain J, Stott JL (2010) Cellular immune responses in cetaceans immunized with a porcine erysipelas vaccine. Vet Immunol Immunopathol 137(3–4):181–189
Soto A, Zapardiel J, Soriano F (1994) Evaluation of API Coryne system for identifying coryneform bacteria. J Clin Pathol 47:756–759
Swan RA, Lindsey MJ (1998) Treatment and control by vaccination of erysipelas in farmed emus (Dromaius novohollandiae). Aust Vet J 76:325–327
Thurman GD, Downes SJ, Fothergill MB, Goodwin NM, Hegarty MM (1983) Diagnosis and successful treatment of subacute erysipelas in a captive dolphin. J S Afr Vet Assoc 54:193–200
Wang Q, Chang BJ, Riley TV (2010) Erysipelothrix rhusiopathiae. Vet Microbiol 140(3–4):405–417
Wood RL (1992) Erysipelas. In: Leman AD, Straw BE, Mengeling WL, D’Allaire S, Taylor DJ (eds) Diseases of Swine, 7th edn. Iowa State Univ. Press, Ames, pp 475–486
Wood RL, Shuman RD (1981) Erysipelothrix infection. In: Davis JL, Karstad LH, Trainer DO (eds) Infectious diseases of wild mammals, 2nd edn. Iowa State University Press, Ames, pp 297–305
Yamazaki Y (2006) A multiplex polymerase chain reaction for discriminating Erysipelothrix rhusiopathiae from Erysipelothrix tonsillarum. J Vet Diagn Invest 18:384–387
Acknowledgments
The authors are grateful to Loro Parque and L’Oceanogràfic de Valencia dolphin training team for their help in this work and also to Cristine Jo Dreisörner and Andrea Cádiz Ortiz from Loro Parque Veterinary Clinic and the staff from L’Oceanogràfic de Valencia Veterinary Clinic; thanks to them, the collection of samples for the development of this work was possible. JLM was funded by a Grant RYC-2011-08863 from the Ramón y Cajal Subprogramme of the Spanish Ministerio de Economía y Competitividad. MRB was funded by Becas Fundación Cajacanarias para Postgraduados 2014. The authors are also grateful to Loro Parque Fundación for funding this research. Finally, we are grateful to the managing direction of Loro Parque for allowing us to undertake this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Erko Stackebrandt.
Rights and permissions
About this article
Cite this article
Bernal-Guadarrama, M.J., García-Parraga, D., Fernández-Gallardo, N. et al. Development of an indirect immunofluorescence technique for the evaluation of generated antibody titers against Erysipelothrix rhusiopathiae in captive bottlenose dolphins (Tursiops truncatus). Arch Microbiol 196, 785–790 (2014). https://doi.org/10.1007/s00203-014-1020-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-014-1020-0