Abstract
Summary
This article describes the development of a model for postmenopausal osteoporosis (PMO) based on Swedish data that is easily adaptable to other countries.
Introduction
The aims of the study were to develop and validate a model to describe the current/future burden of PMO in different national settings.
Methods
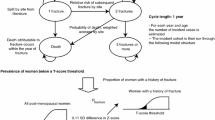
For validation purposes, the model was developed using Swedish data and provides estimates from 1990. For each year of the study, the “incident cohort” (women experiencing a first osteoporotic fracture) was identified and run through a Markov model using 1-year cycles until 2020. Health states were based on the number of fractures and death. Fracture by site (hip, vertebral, and non-hip non-vertebral) was tracked for each health state. Transition probabilities reflected site-specific risk of death and subsequent fractures. Bone mineral density (BMD) was included as a model output; model inputs included population size and life tables from 1970 to 2020, incidence of fracture, relative risk of subsequent fractures based on prior fracture, relative risk of death following a fracture by site, and BMD by age (mean and standard deviation).
Results
Model predictions averaged across age groups estimated the incidence of hip, vertebral, and other osteoporotic fractures within a 5% margin of error versus published data. In Sweden, the number of osteoporotic fractures is expected to rise by 11.5% between 2009 and 2020, with a shift towards more vertebral fractures and multiple fractures.
Conclusion
The current PMO disease model is easily adaptable to other countries, providing a consistent measure of present and future burden of PMO in different settings.





Similar content being viewed by others
References
Kanis JA, on behalf of the World Organization Scientific Group (2007) Assessment of osteoporosis at the primary health care level. Technical report. World Health Organization Collaborating Centre for Metabolic Bone Diseases. University of Sheffield, UK
Johnell O, Kanis JA (2006) An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int 17:1726–1733
World Health Organization (2004) Burden of disease in DALYs by cause, sex and mortality stratum in WHO regions, estimates for 2002, The world health report 2004: changing history. World Health Organization, Geneva, pp 126–131
Giannakouris K (2008) Ageing characterises the demographic perspectives of the European societies. Eurostat, Statistics in Focus issue number 72/2008
Melton LJ 3rd, Therneau TM, Larson DR (1998) Long-term trends in hip fracture prevalence: the influence of hip fracture incidence and survival. Osteoporos Int 8:68–74
Parker MJ, Anand JK (1991) What is the true mortality of hip fractures? Public Health 105:443–446
Kanis JA, Oden A, Johnell O, De Laet C, Jonsson B, Oglesby AK (2003) The components of excess mortality after hip fracture. Bone 32:468–473
Kanis JA, Oden A, Johnell O, De Laet C, Jonsson B (2004) Excess mortality after hospitalisation for vertebral fracture. Osteoporos Int 15:108–112
Klotzbuecher CM, Ross PD, Lansman PB, Abbott TA 3rd, Berger M (2000) Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res 15:721–739
Johnell O, Kanis JA, Oden A, Sernbo I, Redlund-Johnell I, Petterson C, De Laet C et al (2004) Fracture risk following an osteoporotic fracture. Osteoporos Int 15:175–179
Statistics Sweden, Demographic Reports: The future population of Sweden 2006–2050. http://www.scb.se/statistik/_publikationer/BE0401_2006I50_BR_BE51BR0602ENG.pdf. Accessed 17 September 2009
Kanis JA, Oden A, Johnell O, Jonsson B, de Laet C, Dawson A (2001) The burden of osteoporotic fractures: a method for setting intervention thresholds. Osteoporos Int 12:417–427
Kanis JA, Johnell O, Oden A, Sembo I, Redlund-Johnell I, Dawson A, De Laet C et al (2000) Long-term risk of osteoporotic fracture in Malmö. Osteoporos Int 11:669–674
Johnell O, Kanis JA, Odén A, Sernbo I, Redlund-Johnell I, Petterson C, De Laet C et al (2004) Mortality after osteoporotic fractures. Osteoporos Int 15:38–42
Barrett JA, Baron JA, Beach ML (2003) Mortality and pulmonary embolism after fracture in the elderly. Osteoporos Int 14:889–894
Marshall D, Johnell O, Wedel H (1996) Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. BMJ 312:1254–1259
Kanis JA, Johnell O, De Laet C, Johansson H, Oden A, Delmas P, Eisman J et al (2004) A meta-analysis of previous fracture and subsequent fracture risk. Bone 35:375–382
Looker AC, Wahner HW, Dunn WL, Calvo MS, Harris TB, Heyse SP, Johnston CC Jr et al (1998) Updated data on proximal femur bone mineral levels of US adults. Osteoporos Int 8:468–489
Löfman O, Larsson L, Ross I, Toss G, Berglund K (1997) Bone mineral density in normal Swedish women. Bone 20:167–174
Karlsson MK, Gärdsell P, Johnell O, Nilsson BE, Akesson K, Obrant KJ (1993) Bone mineral normative data in Malmö, Sweden. Comparison with reference data and hip fracture incidence in other ethnic groups. Acta Orthop Scand 64:168–172
Melton LJ, Crowson CS, O’Fallon WM (1999) Fracture incidence in Olmsted County, Minnesota: comparison of urban and with rural rates and changes in urban rates over time. Osteoporos Int 9:29–37
Cooper C, Campion G, Melton LJ 3rd (1992) Hip fractures in the elderly: a world-wide projection. Osteoporos Int 2:285–289
Gullberg B, Johnell O, Kanis JA (1997) World-wide projections for hip fracture. Osteoporos Int 7:407–413
Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson A (2007) Incidence and economic burden of osteoporosis-related fractures in the United States, 2005–2025. J Bone Miner Res 22:465–475
Schwenkglenks M, Lippuner K, Hauselmann HJ, Szucs TD (2005) A model of osteoporosis impact in Switzerland 2000–2020. Osteoporos Int 16:659–671
Melton LJ 3rd, Kanis JA, Johnell O (2005) Potential impact of osteoporosis treatment on hip fracture trends. J Bone Miner Res 20:895–897
Icks A, Haastert B, Wildner M, Becker C, Meyer G (2008) Trend of hip fracture incidence in Germany 1995–2004: a population-based study. Osteoporos Int 19:1139–1145
Kannus P, Niemi S, Parkkari J, Palvanen M, Vuori I, Järvinen M (2006) Nationwide decline in incidence of hip fracture. J Bone Miner Res 21:1836–1838
Brauer CA, Coca-Perraillon M, Cutler DM, Rosen AB (2009) Incidence and mortality of hip fractures in the United States. JAMA 302:1573–1579
Leslie WD, O’Donnell S, Jean S, Lagacé C, Walsh P, Bancej C, Morin S et al (2009) Trends in hip fracture rates in Canada. JAMA 302:883–889
Gullberg B, Duppe H, Nilsson B, Redlund-Johnell I, Sernbo I, Obrant K, Johnell O (1993) Incidence of hip fractures in Malmö, Sweden (1950–1991). Bone 14(Suppl 1):S23–S29
Rogmark C, Sernbo I, Johnell O, Nilsson JA (1999) Incidence of hip fractures in Malmö, Sweden, 1992–1995. A trend-break. Acta Orthop Scand 70:19–22
Acknowledgements
The study was supported by the International Osteoporosis Foundation and funded by Amgen (Europe) GmbH. The authors thank Dr. Matthew Gitlin and Sean Robbins from Amgen (Europe) GmbH for their involvement in this project, and Professor Alistair McGuire from the London School of Economics for his advice on the model development. Formatting/styling support was funded by Amgen (Europe) GmbH and GlaxoSmithKline.
Conflicts of interest
None.
Author information
Authors and Affiliations
Consortia
Corresponding author
Electronic supplementary material
Model parameters
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gauthier, A., Kanis, J.A., Martin, M. et al. Development and validation of a disease model for postmenopausal osteoporosis. Osteoporos Int 22, 771–780 (2011). https://doi.org/10.1007/s00198-010-1358-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-010-1358-3