Abstract
Summary
The effect of strontium ranelate (SR) on human osteoblast differentiation was tested. SR induced osteoblastic proliferation, in vitro mineralization, and increased the expression of osteocyte markers. SR also elicited an osteoprotegerin (OPG) secretory response. We conclude that SR promotes the osteoblast maturation and osteocyte differentiation while promoting an additional antiresorptive effect.
Introduction
SR is a new treatment for osteoporosis that reduces the risk of hip and vertebral fractures in postmenopausal women. This study sought to investigate the extent, to which SR modulates human osteoblast differentiation.
Methods
Adult human primary osteoblasts (NHBC) were exposed to SR under mineralizing conditions in long-term cultures. Osteoblast differentiation status was investigated by cell-surface phenotypic analysis. Expression of genes associated with osteoblast/osteocyte differentiation was examined using real-time RT-PCR. Secreted OPG was assayed by enzyme-linked immunosorbent assay.
Results
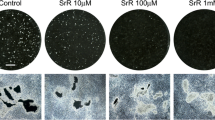
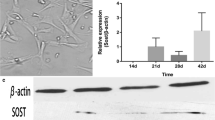
SR significantly increased osteoblast replication. SR time- and dose-dependently induced an osteocyte-like phenotype, as determined by cell surface alkaline phosphatase and STRO-1 expression. SR at 5 mM or greater dramatically increased in vitro mineralization. In parallel, mRNA levels of dentin matrix protein (DMP)-1 and sclerostin were higher under SR treatment, strongly suggestive of the presence of osteocytes. SR also increased the OPG/RANKL ratio throughout the culture period, consistent with an effect to inhibit osteoblast-induced osteoclastogenesis.
Conclusions
This study suggests that SR can promote osteoblast maturation and an osteocyte-like phenotype. Coupled with its effect on the OPG/RANKL system, these findings are consistent with in vivo effects in patients receiving SR for the treatment of osteoporosis.






Similar content being viewed by others
References
Meunier PJ, Roux C, Seeman E, Ortolani S, Badurski JE, Spector TD, Cannata J, Balogh A, Lemmel EM, Pors-Nielsen S, Rizzoli R, Genant HK, Reginster JY (2004) The effects of strontium ranelate on the risk of vertebral fracture in women with postmenopausal osteoporosis. N Engl J Med 350(5):459–468
Reginster JY, Seeman E, De Vernejoul MC, Adami S, Compston J, Phenekos C, Devogelaer JP, Curiel MD, Sawicki A, Goemaere S, Sorensen OH, Felsenberg D, Meunier PJ (2005) Strontium ranelate reduces the risk of nonvertebral fractures in postmenopausal women with osteoporosis: Treatment of Peripheral Osteoporosis (TROPOS) study. J Clin Endocrinol Metab 90(5):2816–2822
Marie PJ, Hott M, Modrowski D, De Pollak C, Guillemain J, Deloffre P, Tsouderos Y (1993) An uncoupling agent containing strontium prevents bone loss by depressing bone resorption and maintaining bone formation in estrogen-deficient rats. J Bone Miner Res 8(5):607–615
Hott M, Deloffre P, Tsouderos Y, Marie PJ (2003) S12911-2 reduces bone loss induced by short-term immobilization in rats. Bone 33(1):115–123
Ammann P, Shen V, Robin B, Mauras Y, Bonjour JP, Rizzoli R (2004) Strontium ranelate improves bone resistance by increasing bone mass and improving architecture in intact female rats. J Bone Miner Res 19(12):2012–2020
Ammann P, Badoud I, Barraud S, Dayer R, Rizzoli R (2007) Strontium ranelate treatment improves trabecular and cortical intrinsic bone tissue quality, a determinant of bone strength. J Bone Miner Res 22(9):1419–1425
Delannoy P, Bazot D, Marie PJ (2002) Long-term treatment with strontium ranelate increases vertebral bone mass without deleterious effect in mice. Metabolism 51(7):906–911
Baron R, Tsouderos Y (2002) In vitro effects of S12911-2 on osteoclast function and bone marrow macrophage differentiation. Eur J Pharmacol 450(1):11–17
Takahashi N, Sasaki T, Tsouderos Y, Suda T (2003) S 12911-2 inhibits osteoclastic bone resorption in vitro. J Bone Miner Res 18(6):1082–1087
Canalis E, Hott M, Deloffre P, Tsouderos Y, Marie PJ (1996) The divalent strontium salt S12911 enhances bone cell replication and bone formation in vitro. Bone 18(6):517–523
Bonnelye E, Chabadel A, Saltel F, Jurdic P (2008) Dual effect of strontium ranelate: stimulation of osteoblast differentiation and inhibition of osteoclast formation and resorption in vitro. Bone 42(1):129–138
Bonewald LF (2007) Osteocytes as dynamic, multifunctional cells. Ann N Y Acad Sci 1116:281–290
Seeman E (2006) Osteocytes—martyrs for integrity of bone strength. Osteoporos Int 17(10):1443–1448
O’Brien CA, Jia D, Plotkin LI, Bellido T, Powers CC, Stewart SA, Manolagas SC, Weinstein RS (2004) Glucocorticoids act directly on osteoblasts and osteocytes to induce their apoptosis and reduce bone formation and strength. Endocrinology 145(4):1835–1841
Clark WD, Smith EL, Linn KA, Paul-Murphy JR, Muir P, Cook ME (2005) Osteocyte apoptosis and osteoclast presence in chicken radii 0–4 days following osteotomy. Calcif Tissue Int 77(5):327–336
Suva LJ, Gaddy D, Perrien DS, Thomas RL, Findlay DM (2005) Regulation of bone mass by mechanical loading: microarchitecture and genetics. Curr Osteoporos Rep 3(2):46–51
Qiu S, Rao DS, Palnitkar S, Parfitt AM (2003) Reduced iliac cancellous osteocyte density in patients with osteoporotic vertebral fracture. J Bone Miner Res 18(9):1657–1663
Mullender MG, Tan SD, Vico L, Alexandre C, Klein-Nulend J (2005) Differences in osteocyte density and bone histomorphometry between men and women and between healthy and osteoporotic subjects. Calcif Tissue Int 77(5):291–296
Frank JD, Ryan M, Kalscheur VL, Ruaux-Mason CP, Hozak RR, Muir P (2002) Aging and accumulation of microdamage in canine bone. Bone 30(1):201–206
Plotkin LI, Mathov I, Aguirre JI, Parfitt AM, Manolagas SC, Bellido T (2005) Mechanical stimulation prevents osteocyte apoptosis: requirement of integrins, Src kinases, and ERKs. Am J Physiol Cell Physiol 289(3):C633–C643
Aguirre JI, Plotkin LI, Stewart SA, Weinstein RS, Parfitt AM, Manolagas SC, Bellido T (2006) Osteocyte apoptosis is induced by weightlessness in mice and precedes osteoclast recruitment and bone loss. J Bone Miner Res 21(4):605–615
Dodd JS, Raleigh JA, Gross TS (1999) Osteocyte hypoxia: a novel mechanotransduction pathway. Am J Physiol 277(3 Pt 1):C598–C602
James J, Steijn-Myagkaya GL (1986) Death of osteocytes. Electron microscopy after in vitro ischaemia. J Bone Joint Surg Br 68(4):620–624
Eberhardt AW, Yeager-Jones A, Blair HC (2001) Regional trabecular bone matrix degeneration and osteocyte death in femora of glucocorticoid-treated rabbits. Endocrinology 142(3):1333–1340
Catto M (1977) Ischaemia of bone. J Clin Pathol Suppl (R Coll Pathol) 11:78–93
Gu G, Hentunen TA, Nars M, Harkonen PL, Vaananen HK (2005) Estrogen protects primary osteocytes against glucocorticoid-induced apoptosis. Apoptosis 10(3):583–595
Poole KE, van Bezooijen RL, Loveridge N, Hamersma H, Papapoulos SE, Lowik CW, Reeve J (2005) Sclerostin is a delayed secreted product of osteocytes that inhibits bone formation. Faseb J 19(13):1842–1844
Gronthos S, Zannettino AC, Graves SE, Ohta S, Hay SJ, Simmons PJ (1999) Differential cell surface expression of the STRO-1 and alkaline phosphatase antigens on discrete developmental stages in primary cultures of human bone cells. J Bone Miner Res 14(1):47–56
Stewart K, Walsh S, Screen J, Jefferiss CM, Chainey J, Jordan GR, Beresford JN (1999) Further characterization of cells expressing STRO-1 in cultures of adult human bone marrow stromal cells. J Bone Miner Res 14(8):1345–1356
Kalajzic I, Braut A, Guo D, Jiang X, Kronenberg MS, Mina M, Harris MA, Harris SE, Rowe DW (2004) Dentin matrix protein 1 expression during osteoblastic differentiation, generation of an osteocyte GFP-transgene. Bone 35(1):74–82
Feng JQ, Ward LM, Liu S, Lu Y, Xie Y, Yuan B, Yu X, Rauch F, Davis SI, Zhang S, Rios H, Drezner MK, Quarles LD, Bonewald LF, White KE (2006) Loss of DMP1 causes rickets and osteomalacia and identifies a role for osteocytes in mineral metabolism. Nat Genet 38(11):1310–1315
Gu G, Nars M, Hentunen TA, Metsikko K, Vaananen HK (2006) Isolated primary osteocytes express functional gap junctions in vitro. Cell Tissue Res 323(2):263–271
Toyosawa S, Shintani S, Fujiwara T, Ooshima T, Sato A, Ijuhin N, Komori T (2001) Dentin matrix protein 1 is predominantly expressed in chicken and rat osteocytes but not in osteoblasts. J Bone Miner Res 16(11):2017–2026
Winkler DG, Sutherland MK, Geoghegan JC, Yu C, Hayes T, Skonier JE, Shpektor D, Jonas M, Kovacevich BR, Staehling-Hampton K, Appleby M, Brunkow ME, Latham JA (2003) Osteocyte control of bone formation via sclerostin, a novel BMP antagonist. Embo J 22(23):6267–6276
van Bezooijen RL, ten Dijke P, Papapoulos SE, Lowik CW (2005) SOST/sclerostin, an osteocyte-derived negative regulator of bone formation. Cytokine Growth Factor Rev 16(3):319–327
Atkins GJ, Kostakis P, Pan B, Farrugia A, Gronthos S, Evdokiou A, Harrison K, Findlay DM, Zannettino AC (2003) RANKL expression is related to the differentiation state of human osteoblasts. J Bone Miner Res 18(6):1088–1098
Atkins GJ, Bouralexis S, Evdokiou A, Hay S, Labrinidis A, Zannettino AC, Haynes DR, Findlay DM (2002) Human osteoblasts are resistant to Apo2L/TRAIL-mediated apoptosis. Bone 31(4):448–456
Findlay DM, Raggatt LJ, Bouralexis S, Hay S, Atkins GJ, Evdokiou A (2002) Calcitonin decreases the adherence and survival of HEK-293 cells by a caspase-independent mechanism. J Endocrinol 175(3):715–725
Atkins GJ, Anderson PH, Findlay DM, Welldon KJ, Vincent C, Zannettino AC, O’Loughlin PD, Morris HA (2007) Metabolism of vitamin D(3) in human osteoblasts: evidence for autocrine and paracrine activities of 1alpha,25-dihydroxyvitamin D(3). Bone 40(6):1517–1528
Lyons AB, Hasbold J, Hodgkin PD (2001) Flow cytometric analysis of cell division history using dilution of carboxyfluorescein diacetate succinimidyl ester, a stably integrated fluorescent probe. Methods Cell Biol 63:375–398
Findlay DM, Welldon K, Atkins GJ, Howie DW, Zannettino AC, Bobyn D (2004) The proliferation and phenotypic expression of human osteoblasts on tantalum metal. Biomaterials 25(12):2215–2227
Lyons AB, Parish CR (1994) Determination of lymphocyte division by flow cytometry. J Immunol Methods 171(1):131–137
Thomas GP, Baker SU, Eisman JA, Gardiner EM (2001) Changing RANKL/OPG mRNA expression in differentiating murine primary osteoblasts. J Endocrinol 170(2):451–460
Silvestrini G, Ballanti P, Patacchioli F, Leopizzi M, Gualtieri N, Monnazzi P, Tremante E, Sardella D, Bonucci E (2005) Detection of osteoprotegerin (OPG) and its ligand (RANKL) mRNA and protein in femur and tibia of the rat. J Mol Histol 36(1):59–67
Zhao S, Zhang YK, Harris S, Ahuja SS, Bonewald LF (2002) MLO-Y4 osteocyte-like cells support osteoclast formation and activation. J Bone Miner Res 17(11):2068–2079
Shiotani A, Shibasaki Y, Sasaki T (2001) Localization of receptor activator of NFkappaB ligand, RANKL, in periodontal tissues during experimental movement of rat molars. J Electron Microsc (Tokyo) 50(4):365–369
Kurata K, Heino TJ, Higaki H, Vaananen HK (2006) Bone marrow cell differentiation induced by mechanically damaged osteocytes in 3D gel-embedded culture. J Bone Miner Res 21(4):616–625
Arlot ME, Jiang Y, Genant HK, Zhao J, Burt-Pichat B, Roux JP, Delmas PD, Meunier PJ (2008) Histomorphometric and microCT analysis of bone biopsies from postmenopausal osteoporotic women treated with strontium ranelate. J Bone Miner Res 23(2):215–222
Aarden EM, Wassenaar AM, Alblas MJ, Nijweide PJ (1996) Immunocytochemical demonstration of extracellular matrix proteins in isolated osteocytes. Histochem Cell Biol 106(5):495–501
Mikuni-Takagaki Y, Kakai Y, Satoyoshi M, Kawano E, Suzuki Y, Kawase T, Saito S (1995) Matrix mineralization and the differentiation of osteocyte-like cells in culture. J Bone Miner Res 10(2):231–242
Robling AG, Niziolek PJ, Baldridge LA, Condon KW, Allen MR, Alam I, Mantila SM, Gluhak-Heinrich J, Bellido TM, Harris SE, Turner CH (2008) Mechanical stimulation of bone in vivo reduces osteocyte expression of sost/sclerostin. J Biol Chem 283(9):5866–5875
Zhang K, Barragan-Adjemian C, Ye L, Kotha S, Dallas M, Lu Y, Zhao S, Harris M, Harris SE, Feng JQ, Bonewald LF (2006) E11/gp38 selective expression in osteocytes: regulation by mechanical strain and role in dendrite elongation. Mol Cell Biol 26(12):4539–4552
Nampei A, Hashimoto J, Hayashida K, Tsuboi H, Shi K, Tsuji I, Miyashita H, Yamada T, Matsukawa N, Matsumoto M, Morimoto S, Ogihara T, Ochi T, Yoshikawa H (2004) Matrix extracellular phosphoglycoprotein (MEPE) is highly expressed in osteocytes in human bone. J Bone Miner Metab 22(3):176–184
Choudhary S, Halbout P, Alander C, Raisz L, Pilbeam C (2007) Strontium ranelate promotes osteoblastic differentiation and mineralization of murine bone marrow stromal cells: involvement of prostaglandins. J Bone Miner Res 22(7):1002–1010
Ikeda T, Utsuyama M, Hirokawa K (2001) Expression profiles of receptor activator of nuclear factor kappaB ligand, receptor activator of nuclear factor kappaB, and osteoprotegerin messenger RNA in aged and ovariectomized rat bones. J Bone Miner Res 16(8):1416–1425
Acknowledgements
This study was funded in part by Servier, Courbevoie, France. This work was also supported by the National Health and Medical Research Council of Australia (NHMRC). GJA was supported by a NHMRC R Douglas Wright Fellowship and by Osteoporosis Australia. The authors are grateful to the surgeons and nursing staff of the Department of Orthopaedics and Trauma of the Royal Adelaide Hospital for their kind assistance in the provision of bone samples at surgery.
Conflicts of interest
This study was funded in part by Servier. PH is an employee of Servier. The authors state that they had full access to all of the data in the study. By so doing, they accept complete responsibility for the integrity of the data and the accuracy of the data analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Atkins, G.J., Welldon, K.J., Halbout, P. et al. Strontium ranelate treatment of human primary osteoblasts promotes an osteocyte-like phenotype while eliciting an osteoprotegerin response. Osteoporos Int 20, 653–664 (2009). https://doi.org/10.1007/s00198-008-0728-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-008-0728-6