Abstract
Introduction and hypothesis
Urethral injection therapy (UIT) has been performed since the early 20th century and a variety of agents have been launched. In 2006, polyacrylamide hydrogel (PAGH) was introduced and is now widely used as an agent. The objective was to evaluate the efficacy of PAGH based on a national population over a 5-year period (2007–2011) and the influence of patient-related factors, surgeon experience, and department volume.
Methods
A retrospective cohort study was carried out based on data from the Danish Urogynaecological Database (DugaBase).
Results
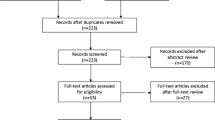
A total of 731 women were registered in the DugaBase. Cure was achieved in 75 out of 252 women (29.8%) and no leakage at all in 23 out of 252 (9.1%) at the 3-month follow-up. The mean total International Consultation on Incontinence Questionnaire-Short Form (ICIQ-SF) score decreased from 16 (SD 3.8) to 10.6 (SD 6.2; p < 0.001). UIT was performed at 16 departments, of which four high-volume departments performed 547 out of 814 UITs (67.2%). Women with severe UI had a decreased chance of cure (all ICIQ-SF scores), as did women on antimuscarinic drugs (adjusted OR 0.14; 95%, CI 0.04–0.41 “frequency”) and (adjusted OR 0.33; 95%, CI 0.13–0.82, “amount”). Women treated by a high-volume surgeon had a higher chance of cure (OR 4.51; 95% CI, 1.21–16.82, “frequency”) and a lower risk of 30-day hospital contacts (OR 0.27; 95% CI 0.09–0.76).
Conclusion
The study represented a cure for UIT among women in an everyday life setting. A surgeon learning curve for UIT was indicated, as was assigning interventions to fewer hands to improve the surgical training value and consequently the cure rate for women with UIT.


Similar content being viewed by others
References
Meyer L. 2 tilfælde af incontinentia urinae helbredte ved parafininjektioner. Ugeskr Laeger. 1904;104:5.
Kotb AF, Campeau L, Corcos J. Urethral bulking agents: techniques and outcomes. Curr Urol Rep. 2009;10(5):396–400.
Lose G, Mouritsen L, Nielsen JB. A new bulking agent (polyacrylamide hydrogel) for treating stress urinary incontinence in women. BJU Int. 2006;98(1):100–4. doi:10.1111/j.1464-410X.2006.06205.x.
Lose G, Sorensen HC, Axelsen SM, Falconer C, Lobodasch K, Safwat T. An open multicenter study of polyacrylamide hydrogel (Bulkamid®) for female stress and mixed urinary incontinence. Int Urogynecol J. 2010;21(12):1471–7. doi:10.1007/s00192-010-1214-1.
Martan A, Masata J, Svabik K, El-Haddad R, Hubka P. Transurethral injection of polyacrylamide hydrogel (Bulkamid®) for the treatment of female stress urinary incontinence and changes in the cure rate over time. Ceska Gynekol. 2013;78(6):554–9.
Beraru A, Droupy S, Wagner L, Soustelle L, Muyschondt C, Ben Naoum K, et al. Efficacy of periurethral injections of polyacrylamide hydrogel (Bulkamid(®)) and quality of life of patients with urinary incontinence due to sphincter deficiency (IUE-IS). Prog Urol. 2014;24(8):501–10. doi:10.1016/j.purol.2014.02.003.
Leone Roberti Maggiore U, Alessandri F, Medica M, Gabelli M, Venturini PL, Ferrero S. Outpatient periurethral injections of polyacrylamide hydrogel for the treatment of female stress urinary incontinence: effectiveness and safety. Arch Gynecol Obstet. 2013;288(1):131–7. doi:10.1007/s00404-013-2718-y.
Toozs-Hobson P, Al-Singary W, Fynes M, Tegerstedt G, Lose G. Two-year follow-up of an open-label multicenter study of polyacrylamide hydrogel (Bulkamid®) for female stress and stress-predominant mixed incontinence. Int Urogynecol J. 2012;23(10):1373–8. doi:10.1007/s00192-012-1761-8.
Trutnovsky G, Tamussino K, Greimel E, Bjelic-Radisic V. Quality of life after periurethral injection with polyacrylamide hydrogel for stress urinary incontinence. Int Urogynecol J. 2011;22(3):353–6. doi:10.1007/s00192-010-1296-9.
Vecchioli-Scaldazza CV, Smaali C, Morosetti C, Azizi B, Giannubilo W, Ferrara V. Polyacrylamide hydrogel (bulkamid®) in female patients of 80 or more years with urinary incontinence. Int Braz J Urol. 2014;40(1):37–43. doi:10.1590/s1677-5538.ibju.2014.01.06.
Sokol ER, Karram MM, Dmochowski R. Efficacy and safety of polyacrylamide hydrogel for the treatment of female stress incontinence: a randomized, prospective, multicenter North American study. J Urol. 2014;192(3):843–9. doi:10.1016/j.juro.2014.03.109.
Pai A, Al-Singary W. Durability, safety and efficacy of polyacrylamide hydrogel (Bulkamid(®)) in the management of stress and mixed urinary incontinence: three year follow up outcomes. Cent European J Urol. 2015;68(4):428–33. doi:10.5173/ceju.2015.647.
Zivanovic I, Rautenberg O, Lobodasch K, von Bunau G, Walser C, Viereck V. Urethral bulking for recurrent stress urinary incontinence after midurethral sling failure. Neurourol Urodyn. 2016. doi:10.1002/nau.23007.
Kasi AD, Pergialiotis V, Perrea DN, Khunda A, Doumouchtsis SK. Polyacrylamide hydrogel (Bulkamid®) for stress urinary incontinence in women: a systematic review of the literature. Int Urogynecol J. 2016;27(3):367–75. doi:10.1007/s00192-015-2781-y.
Monga AK, Robinson D, Stanton SL. Periurethral collagen injections for genuine stress incontinence: a 2-year follow-up. Br J Urol. 1995;76(2):156–60.
Shah PJR. Injectables and GSI. Urol News. 1997;2:7–11.
Guldberg R, Brostrom S, Hansen JK, Kaerlev L, Gradel KO, Norgard BM, et al. The Danish Urogynaecological Database: establishment, completeness and validity. Int Urogynecol J. 2012;6(24):983–90. doi:10.1007/s00192-012-1968-8.
Larsen MD, Lose G, Guldberg R, Gradel KO. Discrepancies between patient-reported outcome measures when assessing urinary incontinence or pelvic- prolapse surgery. Int Urogynecol J. 2015;4(27):537–43. doi:10.1007/s00192-015-2840-4.
Guldberg R, Kesmodel US, Hansen JK, Gradel KO, Brostrom S, Kaerlev L, et al. Patient reported outcome measures in women undergoing surgery for urinary incontinence and pelvic organ prolapse in Denmark, 2006–2011. Int Urogynecol J. 2013;24(7):1127–34. doi:10.1007/s00192-012-1979-5.
Schiøler G, Larsen OB (2005) Klassifikation af operationer. 2 edn. Copenhagen: Munksgaard
Danish Urogynaecological Database, annual report 2007. Accessed 12 August 2016
Schmidt M, Schmidt SA, Sandegaard JL, Ehrenstein V, Pedersen L, Sorensen HT. The Danish national patient registry: a review of content, data quality, and research potential. J Clin Epidemiol. 2015;7:449–90. doi:10.2147/clep.s91125.
Kjaergaard J, Clemmensen IH, Thomsen BL, Storm HH. Validity of diagnoses of and operations for nonmalignant gynecological conditions in the Danish national hospital registry. J Clin Epidemiol. 2002;55(2):137–42.
Kildemoes HW, Sorensen HT, Hallas J. The Danish national prescription registry. Scand J Public Health. 2011;39(7 Suppl):38–41. doi:10.1177/1403494810394717.
STROBE (2016) Strengthening the Reporting of Observational Studies in Epidemiology (STROBE). http://www.strobe-statement.org/index.php?id=available-checklists. Accessed 21 June 2016
International Classification of Diseases (ICD10). http://www.who.int/classifications/icd/ICD-10_2nd_ed_volume2.pdf. Accessed 31 August 2016
Castillo PA, Espaillat-Rijo LM, Davila GW. Outcome measures and definition of cure in female stress urinary incontinence surgery: a survey of recent publications. Int Urogynecol J. 2010;21(3):343–8. doi:10.1007/s00192-009-1032-5.
Kirchin V, Page T, Keegan PE, Atiemo K, Cody JD, McClinton S (2012) Urethral injection therapy for urinary incontinence in women. Cochrane Database Syst Rev 2:CD003881. doi:10.1002/14651858.CD003881.pub3
Freeman RM (2007) What’s a ‘cure’? Patient-centred outcomes of treatments for stress urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct 18(1):13–18 10.1007/s00192-006-0110-1
Statens Serum Institut. http://www.ssi.dk/Sundhedsdataogit/Sundhedsvaesenet%20i%20tal/Sundhedsveasenet%20generelt/Operationer/Operationer%20Regionsfordelt.aspx. Accessed 31 August 2016
Acknowledgements
Funding was provided by Aase og Ejnar Danielsens Fond and Edgar Schnohr og Hustru Gilberte Schnohr’s Fond. The Centre for Quality, Region of Southern Denmark, Middelfart, and the University of Southern Denmark financed the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the Danish Data Protection Agency (J.nr. 2012-41-0414). As the study did not include patient contact, it was not necessary to obtain approval from the Health Research Ethics Committee.
Conflicts of interest
Margrethe Foss Hansen incurred conference and travel expenses for attendance at the EUGA Leading Lights in Urogynaecology Congress, Warsaw, 2015, paid for by Astella. The other authors have no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Hansen, M.F., Lose, G., Kesmodel, U.S. et al. A national population-based cohort study of urethral injection therapy for female stress and mixed urinary incontinence: the Danish Urogynaecological Database, 2007–2011. Int Urogynecol J 28, 1309–1317 (2017). https://doi.org/10.1007/s00192-017-3265-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-017-3265-z