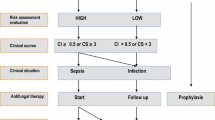
Abstract
Introduction
The term invasive candidiasis (IC) refers to both bloodstream and deep-seated invasive infections, such as peritonitis, caused by Candida species. Several guidelines on the management of candidemia and invasive infection due to Candida species have recently been published, but none of them focuses specifically on critically ill patients admitted to intensive care units (ICUs).
Material and Methods
In the absence of available scientific evidence, the resulting recommendations are based solely on epidemiological and clinical evidence in conjunction with expert opinion. The task force used the GRADE (Grading of Recommendations Assessment, Development, and Evaluation) approach to evaluate the recommendations and assign levels of evidence. The recommendations and their strength were decided by consensus and, if necessary, by vote (modified Delphi process). Descriptive statistics were used to analyze the results of the Delphi process. Statements obtaining > 80% agreement were considered to have achieved consensus.
Conclusions
The heterogeneity of this patient population necessitated the creation of a mixed working group comprising experts in clinical microbiology, infectious diseases and intensive care medicine, all chosen on the basis of their expertise in the management of IC and/or research methodology. The working group’s main goal was to provide clinicians with clear and practical recommendations to optimize microbiological diagnosis and treatment of IC. The Systemic Inflammation and Sepsis and Infection sections of the European Society of Intensive Care Medicine (ESICM) and the Critically Ill Patients Study Group of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) therefore decided to develop a set of recommendations for application in non-immunocompromised critically ill patients.

Similar content being viewed by others
References
O’Leary R-A, Einav S, Leone M et al (2018) Management of invasive candidiasis and candidaemia in critically ill adults: expert opinion of the European Society of Anaesthesia Intensive Care Scientific Subcommittee. J Hosp Infect 1:1. https://doi.org/10.1016/j.jhin.2017.11.020
Baldesi O, Bailly S, Ruckly S et al (2017) ICU-acquired candidaemia in France: epidemiology and temporal trends, 2004-2013—a study from the REA-RAISIN network. J Infect 75:59–67. https://doi.org/10.1016/j.jinf.2017.03.011
Pfaller MA, Andes DR, Diekema DJ et al (2014) Epidemiology and outcomes of invasive candidiasis due to non-albicans species of Candida in 2496 patients: data from the Prospective Antifungal Therapy (PATH) registry 2004–2008. PLoS One 9:e101510. https://doi.org/10.1371/journal.pone.0101510
Colombo AL, Guimarães T, Sukienik T et al (2014) Prognostic factors and historical trends in the epidemiology of candidemia in critically ill patients: an analysis of five multicenter studies sequentially conducted over a 9-year period. Intensive Care Med 40:1489–1498. https://doi.org/10.1007/s00134-014-3400-y
Pappas PG, Kauffman CA, Andes DR et al (2016) Clinical practice guideline for the management of candidiasis: 2016 update by the infectious diseases society of America. Clin Infect Dis 62:e1–e50. https://doi.org/10.1093/cid/civ933
Martin-Loeches I, Diaz E, Valles J et al (2014) Risks for multidrug-resistant pathogens in the ICU. Curr Opin Crit Care 20:516–524. https://doi.org/10.1097/MCC.0000000000000124
Delaloye J, Calandra T (2014) Invasive candidiasis as a cause of sepsis in the critically ill patient. Virulence 5:161–169. https://doi.org/10.4161/viru.26187
Calandra T, Roberts JA, Antonelli M et al (2016) Diagnosis and management of invasive candidiasis in the ICU: an updated approach to an old enemy. Crit Care 20:125. https://doi.org/10.1186/s13054-016-1313-6
Vincent J-LJ-L, Martin-Loeches I, Annane D (2016) What patient data should be collected in this randomized controlled trial in sepsis? Intensive Care Med 42:2011–2013. https://doi.org/10.1007/s00134-016-4560-8
Garey KW, Rege M, Pai MP et al (2006) Time to initiation of fluconazole therapy impacts mortality in patients with candidemia: a multi-institutional study. Clin Infect Dis 43:25–31. https://doi.org/10.1086/504810
Bassetti M, Ansaldi F, Diaz-Martin A et al (2014) A multicenter study of septic shock due to candidemia: outcomes and predictors of mortality. Intensive Care Med 40:839–845. https://doi.org/10.1007/s00134-014-3310-z
Paiva J-A, Charles P-E (2017) Biomarker-guided antifungal therapy in patients with suspected invasive candidiasis: ready for prime time? Intensive Care Med 43:1889–1891. https://doi.org/10.1007/s00134-017-4990-y
Cornely OA, Bassetti M, Calandra T et al (2012) ESCMID* guideline for the diagnosis and management of Candida diseases 2012: non-neutropenic adult patients. Clin Microbiol Infect 18(Suppl 7):19–37. https://doi.org/10.1111/1469-0691.12039
Tagliaferri E, Menichetti F (2015) Treatment of invasive candidiasis: between guidelines and daily clinical practice. Expert Rev Anti Infect Ther 13:685–689. https://doi.org/10.1586/14787210.2015.1029916
Echeverria PM, Kett DH, Azoulay E (2011) Candida prophylaxis and therapy in the ICU. Semin Respir Crit Care Med 32:159–173. https://doi.org/10.1055/s-0031-1275528
Bassetti M, Marchetti M, Chakrabarti A et al (2013) A research agenda on the management of intra-abdominal candidiasis: results from a consensus of multinational experts. Intensive Care Med 39:2092–2106. https://doi.org/10.1007/s00134-013-3109-3
Guyatt G, Gutterman D, Baumann MH et al (2006) Grading strength of recommendations and quality of evidence in clinical guidelines: report from an American college of chest physicians task force. Chest 129:174–181. https://doi.org/10.1378/chest.129.1.174
Helmer O (1967) Analysis of the future. The RAND corporation, Santa Monica, California. https://www.rand.org/pubs/papers/P3558.html. Accessed 26 Nov 2018.
Hsu J, Brożek JL, Terracciano L et al (2011) Application of GRADE: making evidence-based recommendations about diagnostic tests in clinical practice guidelines. Implement Sci 6:62. https://doi.org/10.1186/1748-5908-6-62
Schünemann HJ, Mustafa R, Brozek J et al (2016) GRADE Guidelines: 16. GRADE evidence to decision frameworks for tests in clinical practice and public health. J Clin Epidemiol 76:89–98. https://doi.org/10.1016/j.jclinepi.2016.01.032
Ullmann AJ, Cornely OA, Donnelly JP et al (2012) ESCMID* guideline for the diagnosis and management of Candida diseases 2012: developing European guidelines in clinical microbiology and infectious diseases. Clin Microbiol Infect 18(Suppl 7):1–8. https://doi.org/10.1111/1469-0691.12037
Shan Y-S, Sy ED, Wang S-T et al (2006) Early presumptive therapy with fluconazole for occult Candida infection after gastrointestinal surgery. World J Surg 30:119–126. https://doi.org/10.1007/s00268-005-7807-z
León C, Ostrosky-Zeichner L, Schuster M (2014) What’s new in the clinical and diagnostic management of invasive candidiasis in critically ill patients. Intensive Care Med 40:808–819. https://doi.org/10.1007/s00134-014-3281-0
Lopez-Cortes LE, Almirante B, Cuenca-Estrella M et al (2016) Empirical and targeted therapy of candidemia with fluconazole versus echinocandins: a propensity score-derived analysis of a population-based, multicentre prospective cohort. Clin Microbiol Infect 22:733.e1–733.e8. https://doi.org/10.1016/j.cmi.2016.05.008
Rodriguez A, Martin-Loeches I, Yebenes JC (2016) New definition of sepsis and septic shock: what does it give us? Med Intensiva. https://doi.org/10.1016/j.medin.2016.03.008
Pittet D, Monod M, Suter PM et al (1994) Candida colonization and subsequent infections in critically ill surgical patients. Ann Surg 220:751–758
Ostrosky-Zeichner L, Sable C, Sobel J et al (2007) Multicenter retrospective development and validation of a clinical prediction rule for nosocomial invasive candidiasis in the intensive care setting. Eur J Clin Microbiol Infect Dis 26:271–276. https://doi.org/10.1007/s10096-007-0270-z
Leon C, Ruiz-Santana S, Saavedra P et al (2006) A bedside scoring system (“Candida score”) for early antifungal treatment in nonneutropenic critically ill patients with Candida colonization. Crit Care Med 34:730–737. https://doi.org/10.1097/01.CCM.0000202208.37364.7D
Paphitou NI, Ostrosky-Zeichner L, Rex JH (2005) Rules for identifying patients at increased risk for candidal infections in the surgical intensive care unit: approach to developing practical criteria for systematic use in antifungal prophylaxis trials. Med Mycol 43:235–243
Dupont H, Bourichon A, Paugam-Burtz C et al (2003) Can yeast isolation in peritoneal fluid be predicted in intensive care unit patients with peritonitis? Crit Care Med 31:752–757. https://doi.org/10.1097/01.CCM.0000053525.49267.77
Michalopoulos AS, Geroulanos S, Mentzelopoulos SD (2003) Determinants of candidemia and candidemia-related death in cardiothoracic ICU patients. Chest 124:2244–2255
Guillamet CV, Vazquez R, Micek ST et al (2015) Development and validation of a clinical prediction rule for candidemia in hospitalized patients with severe sepsis and septic shock. J Crit Care 30:715–720. https://doi.org/10.1016/j.jcrc.2015.03.010
Hermsen ED, Zapapas MK, Maiefski M et al (2011) Validation and comparison of clinical prediction rules for invasive candidiasis in intensive care unit patients: a matched case-control study. Crit Care 15:R198. https://doi.org/10.1186/cc10366
Leroy G, Lambiotte F, Thévenin D et al (2011) Evaluation of “Candida score” in critically ill patients: a prospective, multicenter, observational, cohort study. Ann Intensive Care 1:50. https://doi.org/10.1186/2110-5820-1-50
Montravers P, Perrigault PF, Timsit JF et al (2017) Antifungal therapy for patients with proven or suspected Candida peritonitis: Amarcand2, a prospective cohort study in French intensive care units. Clin Microbiol Infect 23:117.e1–117.e8. https://doi.org/10.1016/j.cmi.2016.10.001
Clancy CJ, Nguyen MH (2013) Finding the “missing 50%” of invasive candidiasis: how nonculture diagnostics will improve understanding of disease spectrum and transform patient care. Clin Infect Dis 56:1284–1292. https://doi.org/10.1093/cid/cit006
Martin-Loeches I, Garnacho-Montero J, Nseir S (2017) Focus on infection and sepsis 2017. Intensive Care Med 43:867–869. https://doi.org/10.1007/s00134-017-4787-z
Martin-Loeches I, Perner A, Loeches IM, Perner A (2016) Focus on infection and sepsis in intensive care patients. Intensive Care Med 42:491–493. https://doi.org/10.1007/s00134-016-4234-6
Kirby JE, Delaney M, Qian Q, Gold HS (2009) Optimal use of Myco/F lytic and standard BACTEC blood culture bottles for detection of yeast and mycobacteria. Arch Pathol Lab Med 133:93–96. https://doi.org/10.1043/1543-2165-133.1.93
Jekarl DW, Lee S-Y, Lee S et al (2012) Comparison of the Bactec Fx Plus, Mycosis IC/F, Mycosis/F Lytic blood culture media and the BacT/Alert 3D FA media for detection of Candida species in seeded blood culture specimens containing therapeutic peak levels of fluconazole. J Clin Lab Anal 26:412–419. https://doi.org/10.1002/jcla.21535
Köck R, Eiβing LC, Boschin MG et al (2013) Evaluation of bactec mycosis IC/F and plus aerobic/F blood culture bottles for detection of Candida in the presence of antifungal agents. J Clin Microbiol 51:3683–3687. https://doi.org/10.1128/JCM.02048-13
Idelevich EA, Grunewald CM, Wüllenweber J, Becker K (2014) Rapid identification and susceptibility testing of Candida spp. from positive blood cultures by combination of direct MALDI-TOF mass spectrometry and direct inoculation of Vitek 2. PLoS One 9:e114834. https://doi.org/10.1371/journal.pone.0114834
Stone NRH, Gorton RL, Barker K et al (2013) Evaluation of PNA-FISH yeast traffic light for rapid identification of yeast directly from positive blood cultures and assessment of clinical impact. J Clin Microbiol 51:1301–1302. https://doi.org/10.1128/JCM.00028-13
Guinea J, Zaragoza Ó, Escribano P et al (2014) Molecular identification and antifungal susceptibility of yeast isolates causing fungemia collected in a population-based study in Spain in 2010 and 2011. Antimicrob Agents Chemother 58:1529–1537. https://doi.org/10.1128/AAC.02155-13
Avni T, Leibovici L, Paul M (2011) PCR diagnosis of invasive candidiasis: systematic review and meta-analysis. J Clin Microbiol 49:665–670. https://doi.org/10.1128/JCM.01602-10
Lamoth F, Jaton K, Prod’hom G et al (2010) Multiplex blood PCR in combination with blood cultures for improvement of microbiological documentation of infection in febrile neutropenia. J Clin Microbiol 48:3510–3516. https://doi.org/10.1128/JCM.00147-10
Metzgar D, Frinder MW, Rothman RE et al (2016) The IRIDICA BAC BSI Assay: rapid, sensitive and culture-independent identification of bacteria and candida in blood. PLoS One 11:e0158186. https://doi.org/10.1371/journal.pone.0158186
León C, Ruiz-Santana S, Saavedra P et al (2016) Contribution of Candida biomarkers and DNA detection for the diagnosis of invasive candidiasis in ICU patients with severe abdominal conditions. Crit Care 20:149. https://doi.org/10.1186/s13054-016-1324-3
Ramos JT, Villar S, Bouza E et al (2017) Performance of a quantitative PCR-based assay and beta-d-glucan detection for diagnosis of invasive candidiasis in very-low-birth-weight preterm neonatal patients (CANDINEO Study). J Clin Microbiol 55:2752–2764. https://doi.org/10.1128/JCM.00496-17
Beyda ND, Alam MJ, Garey KW (2013) Comparison of the T2Dx instrument with T2Candida assay and automated blood culture in the detection of Candida species using seeded blood samples. Diagn Microbiol Infect Dis 77:324–326. https://doi.org/10.1016/j.diagmicrobio.2013.07.007
Mylonakis E, Clancy CJ, Ostrosky-Zeichner L et al (2015) T2 magnetic resonance assay for the rapid diagnosis of candidemia in whole blood: a clinical trial. Clin Infect Dis 60:892–899. https://doi.org/10.1093/cid/ciu959
Mikulska M, Calandra T, Sanguinetti M et al (2010) The use of mannan antigen and anti-mannan antibodies in the diagnosis of invasive candidiasis: recommendations from the Third European Conference on Infections in Leukemia. Crit Care 14:R222. https://doi.org/10.1186/cc9365
Martínez-Jiménez MC, Muñoz P, Valerio M et al (2015) Candida biomarkers in patients with candidaemia and bacteraemia. J Antimicrob Chemother 70:2354–2361. https://doi.org/10.1093/jac/dkv090
Tissot F, Lamoth F, Hauser PM et al (2013) β-glucan antigenemia anticipates diagnosis of blood culture-negative intraabdominal candidiasis. Am J Respir Crit Care Med 188:1100–1109. https://doi.org/10.1164/rccm.201211-2069OC
De Pauw B, Walsh TJ, Donnelly JP et al (2008) Revised definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) C. Clin Infect Dis 46:1813–1821. https://doi.org/10.1086/588660
Persat F, Ranque S, Derouin F et al (2008) Contribution of the (1---3)-beta-d-glucan assay for diagnosis of invasive fungal infections. J Clin Microbiol 46:1009–1013. https://doi.org/10.1128/JCM.02091-07
León C, Ruiz-Santana S, Saavedra P et al (2012) Value of β-d-glucan and Candida albicans germ tube antibody for discriminating between Candida colonization and invasive candidiasis in patients with severe abdominal conditions. Intensive Care Med 38:1315–1325. https://doi.org/10.1007/s00134-012-2616-y
Posteraro B, Tumbarello M, De Pascale G et al (2016) (1,3)-β-d-Glucan-based antifungal treatment in critically ill adults at high risk of candidaemia: an observational study. J Antimicrob Chemother 71:2262–2269. https://doi.org/10.1093/jac/dkw112
Lahmer T, Rasch S, Schnappauf C et al (2016) Comparison of serum galactomannan and 1,3-beta-d-glucan determination for early detection of invasive pulmonary aspergillosis in critically ill patients with hematological malignancies and septic shock. Mycopathologia 181:505–511. https://doi.org/10.1007/s11046-016-0011-x
Coopersmith CM, de Backer D, Deutschman CS et al (2018) Surviving sepsis campaign: research priorities for sepsis and septic shock. Intensive Care Med. https://doi.org/10.1007/s00134-018-5175-z
Guilbart M, Zogheib E, Ntouba A et al (2016) Compliance with an empirical antimicrobial protocol improves the outcome of complicated intra-abdominal infections: a prospective observational study. Br J Anaesth 117:66–72. https://doi.org/10.1093/bja/aew117
Micek ST, Arnold H, Juang P et al (2014) Effects of empiric antifungal therapy for septic shock on time to appropriate therapy for Candida infection: a pilot study. Clin Ther 36:1226–1232. https://doi.org/10.1016/j.clinthera.2014.06.028
Garnacho-Montero J, Díaz-Martín A, García-Cabrera E et al (2013) Impact on hospital mortality of catheter removal and adequate antifungal therapy in Candida spp. bloodstream infections. J Antimicrob Chemother 68:206–213. https://doi.org/10.1093/jac/dks347
Pierce CG, Lopez-Ribot JL (2013) Candidiasis drug discovery and development: new approaches targeting virulence for discovering and identifying new drugs. Expert Opin Drug Discov 8:1117–1126. https://doi.org/10.1517/17460441.2013.807245
Playford EG, Webster AC, Sorrell TC, Craig JC (2006) Antifungal agents for preventing fungal infections in non-neutropenic critically ill and surgical patients: systematic review and meta-analysis of randomized clinical trials. J Antimicrob Chemother 57:628–638. https://doi.org/10.1093/jac/dki491
Shorr AF, Chung K, Jackson WL et al (2005) Fluconazole prophylaxis in critically ill surgical patients: a meta-analysis. Crit Care Med 33:1928–1935 (quiz 1936)
Vardakas KZ, Samonis G, Michalopoulos A et al (2006) Antifungal prophylaxis with azoles in high-risk, surgical intensive care unit patients: a meta-analysis of randomized, placebo-controlled trials. Crit Care Med 34:1216–1224. https://doi.org/10.1097/01.CCM.0000208357.05675.C3
Cruciani M, de Lalla F, Mengoli C (2005) Prophylaxis of Candida infections in adult trauma and surgical intensive care patients: a systematic review and meta-analysis. Intensive Care Med 31:1479–1487. https://doi.org/10.1007/s00134-005-2794-y
Playford EG, Webster AC, Sorrell TC, Craig JC (2006) Antifungal agents for preventing fungal infections in non-neutropenic critically ill patients. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.cd004920.pub2
Whaley SG, Berkow EL, Rybak JM et al (2016) Azole antifungal resistance in Candida albicans and emerging non-albicans Candida species. Front Microbiol 7:2173. https://doi.org/10.3389/fmicb.2016.02173
Pappas PG, Kauffman CA, Andes D et al (2009) Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis 48:503–535. https://doi.org/10.1086/596757
Bow EJ, Evans G, Fuller J et al (2010) Canadian clinical practice guidelines for invasive candidiasis in adults. Can J Infect Dis Med Microbiol 21:e122–e150
Pang Y-K, Ip M, You JHS (2017) Potential clinical and economic outcomes of active beta-d-glucan surveillance with preemptive therapy for invasive candidiasis at intensive care units: a decision model analysis. Eur J Clin Microbiol Infect Dis 36:187–194. https://doi.org/10.1007/s10096-016-2796-4
Azoulay E, Dupont HH, Tabah A et al (2012) Systemic antifungal therapy in critically ill patients without invasive fungal infection*. Crit Care Med 40:813–822. https://doi.org/10.1097/CCM.0b013e318236f297
Wissing H, Ballus J, Bingold TM et al (2013) Intensive care unit-related fluconazole use in Spain and Germany: patient characteristics and outcomes of a prospective multicenter longitudinal observational study. Infect Drug Resist 6:15–25. https://doi.org/10.2147/IDR.S38945
Piarroux R, Grenouillet F, Balvay P et al (2004) Assessment of preemptive treatment to prevent severe candidiasis in critically ill surgical patients. Crit Care Med 32:2443–2449
Bailly S, Bouadma L, Azoulay E et al (2015) Failure of empirical systemic antifungal therapy in mechanically ventilated critically ill patients. Am J Respir Crit Care Med 191:1139–1146. https://doi.org/10.1164/rccm.201409-1701OC
Leroy O, Bailly S, Gangneux J-P et al (2016) Systemic antifungal therapy for proven or suspected invasive candidiasis: the AmarCAND 2 study. Ann Intensive Care 6:2. https://doi.org/10.1186/s13613-015-0103-7
Golan Y, Wolf MP, Pauker SG et al (2005) Empirical anti-Candida therapy among selected patients in the intensive care unit: a cost-effectiveness analysis. Ann Intern Med 143:857–869
Schuster MG, Edwards JE, Sobel JD et al (2008) Empirical fluconazole versus placebo for intensive care unit patients: a randomized trial. Ann Intern Med 149:83–90
Timsit J-F, Azoulay E, Schwebel C et al (2016) Empirical micafungin treatment and survival without invasive fungal infection in adults with ICU-acquired sepsis, Candida colonization, and multiple organ failure: the EMPIRICUS randomized clinical trial. JAMA. https://doi.org/10.1001/jama.2016.14655
Sinnollareddy MG, Roberts MS, Lipman J et al (2016) In vivo microdialysis to determine subcutaneous interstitial fluid penetration and pharmacokinetics of fluconazole in intensive care unit patients with sepsis. Antimicrob Agents Chemother 60:827–832. https://doi.org/10.1128/AAC.02461-15
Sinnollareddy MG, Roberts JA, Lipman J et al (2015) Pharmacokinetic variability and exposures of fluconazole, anidulafungin, and caspofungin in intensive care unit patients: data from multinational Defining Antibiotic Levels in Intensive care unit (DALI) patients Study. Crit Care 19:33. https://doi.org/10.1186/s13054-015-0758-3
Perlin DS (2015) Echinocandin resistance in Candida. Clin Infect Dis 61(Suppl 6):S612–S617. https://doi.org/10.1093/cid/civ791
Pfaller MA, Diekema DJ, Ostrosky-Zeichner L et al (2008) Correlation of MIC with outcome for Candida species tested against caspofungin, anidulafungin, and micafungin: analysis and proposal for interpretive MIC breakpoints. J Clin Microbiol 46:2620–2629. https://doi.org/10.1128/JCM.00566-08
Puig-Asensio M, Pemán J, Zaragoza R et al (2014) Impact of therapeutic strategies on the prognosis of candidemia in the ICU. Crit Care Med 42:1423–1432. https://doi.org/10.1097/CCM.0000000000000221
Muilwijk EW, Schouten JA, van Leeuwen HJ et al (2014) Pharmacokinetics of caspofungin in ICU patients. J Antimicrob Chemother 69:3294–3299. https://doi.org/10.1093/jac/dku313
Lempers VJ, Schouten JA, Hunfeld NG et al (2015) Altered micafungin pharmacokinetics in intensive care unit patients. Antimicrob Agents Chemother 59:4403–4409. https://doi.org/10.1128/AAC.00623-15
Grau S, Luque S, Campillo N et al (2015) Plasma and peritoneal fluid population pharmacokinetics of micafungin in post-surgical patients with severe peritonitis. J Antimicrob Chemother 70:2854–2861. https://doi.org/10.1093/jac/dkv173
Dupont H, Massias L, Jung B et al (2017) Pharmacokinetic study of anidulafungin in ICU patients with intra-abdominal candidiasis. J Antimicrob Chemother 72:1429–1432. https://doi.org/10.1093/jac/dkw568
Ashley ESD, Lewis R, Lewis JS et al (2006) Pharmacology of systemic antifungal agents. Clin Infect Dis 43:S28–S39. https://doi.org/10.1086/504492
Mora-Duarte J, Betts R, Rotstein C et al (2002) Comparison of caspofungin and amphotericin B for invasive candidiasis. N Engl J Med 347:2020–2029. https://doi.org/10.1056/NEJMoa021585
Kuse E-R, Chetchotisakd P, da Cunha CA et al (2007) Micafungin versus liposomal amphotericin B for candidaemia and invasive candidosis: a phase III randomised double-blind trial. Lancet 369:1519–1527. https://doi.org/10.1016/S0140-6736(07)60605-9
Scudeller L, Viscoli C, Menichetti F et al (2014) An Italian consensus for invasive candidiasis management (ITALIC). Infection 42:263–279. https://doi.org/10.1007/s15010-013-0558-0
Kett DH, Shorr AF, Reboli AC et al (2011) Anidulafungin compared with fluconazole in severely ill patients with candidemia and other forms of invasive candidiasis: support for the 2009 IDSA treatment guidelines for candidiasis. Crit Care 15:R253. https://doi.org/10.1186/cc10514
Gonçalves-Pereira J, Póvoa P (2011) Echinocandins—first line in invasive candidiasis: how strong is this “strong” evidence? Crit Care 15:461. https://doi.org/10.1186/cc10580 (author reply 461)
Andes DR, Safdar N, Baddley JW et al (2012) Impact of treatment strategy on outcomes in patients with candidemia and other forms of invasive candidiasis: a patient-level quantitative review of randomized trials. Clin Infect Dis 54:1110–1122. https://doi.org/10.1093/cid/cis021
Murri R, Scoppettuolo G, Ventura G et al (2016) Initial antifungal strategy does not correlate with mortality in patients with candidemia. Eur J Clin Microbiol Infect Dis 35:187–193. https://doi.org/10.1007/s10096-015-2527-2
Chen Q, Lin M-H, Chen M-L et al (2012) Efficacy and safety of micafungin for invasive candida infections: a meta-analysis of randomized controlled trials. Chin Med J (Engl) 125:345–351
Wang J-F, Xue Y, Zhu X-B, Fan H (2015) Efficacy and safety of echinocandins versus triazoles for the prophylaxis and treatment of fungal infections: a meta-analysis of RCTs. Eur J Clin Microbiol Infect Dis 34:651–659. https://doi.org/10.1007/s10096-014-2287-4
Garnacho-Montero J, Díaz-Martín A, Cantón-Bulnes L et al (2018) Initial antifungal strategy reduces mortality in critically ill patients with candidemia: a propensity score-adjusted analysis of a multicenter study. Crit Care Med 46:384–393. https://doi.org/10.1097/CCM.0000000000002867
Garey KW, Pai MP, Suda KJ et al (2007) Inadequacy of fluconazole dosing in patients with candidemia based on Infectious Diseases Society of America (IDSA) guidelines. Pharmacoepidemiol Drug Saf 16:919–927. https://doi.org/10.1002/pds.1365
Yang Q, Wang T, Xie J et al (2016) Pharmacokinetic/pharmacodynamic adequacy of echinocandins against Candida spp. in intensive care unit patients and general patient populations. Int J Antimicrob Agents 47:397–402. https://doi.org/10.1016/j.ijantimicag.2016.02.004
Gustot T, Ter Heine R, Brauns E et al (2018) Caspofungin dosage adjustments are not required for patients with Child-Pugh B or C cirrhosis. J Antimicrob Chemother 73:2493–2496. https://doi.org/10.1093/jac/dky189
Di Bonaventura G, Spedicato I, Picciani C et al (2004) In vitro pharmacodynamic characteristics of amphotericin B, caspofungin, fluconazole, and voriconazole against bloodstream isolates of infrequent Candida species from patients with hematologic malignancies. Antimicrob Agents Chemother 48:4453–4456. https://doi.org/10.1128/AAC.48.11.4453-4456.2004
Lewis RE (2011) Current concepts in antifungal pharmacology. Mayo Clin Proc 86:805–817. https://doi.org/10.4065/mcp.2011.0247
Johansen HK, Gotzsche PC (2000) Amphotericin B lipid soluble formulations vs amphotericin B in cancer patients with neutropenia. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.cd000969
Keane S, Geoghegan P, Povoa P et al (2018) Systematic review on the first line treatment of amphotericin B in critically ill adults with candidemia or invasive candidiasis. Expert Rev Anti Infect Ther. https://doi.org/10.1080/14787210.2018.1528872
Silva S, Rodrigues C, Araújo D et al (2017) Candida species biofilms’ antifungal resistance. J Fungi 3:8. https://doi.org/10.3390/jof3010008
Muakkassa FK, Ghannoum M (2016) Updates on therapeutic strategies against Candida (and Aspergillus) biofilm related infections. Springer, Cham, pp 95–103
Chaves F, Garnacho-Montero J, Del Pozo JL et al (2018) Diagnosis and treatment of catheter-related bloodstream infection: clinical guidelines of the Spanish society of infectious diseases and clinical microbiology and (SEIMC) and the Spanish society of Spanish society of intensive and critical care medicine A. Med Intens 42:5–36. https://doi.org/10.1016/j.medin.2017.09.012
Vazquez J, Reboli AC, Pappas PG et al (2014) Evaluation of an early step-down strategy from intravenous anidulafungin to oral azole therapy for the treatment of candidemia and other forms of invasive candidiasis: results from an open-label trial. BMC Infect Dis 14:97. https://doi.org/10.1186/1471-2334-14-97
Bailly S, Leroy O, Montravers P et al (2015) Antifungal de-escalation was not associated with adverse outcome in critically ill patients treated for invasive candidiasis: post hoc analyses of the AmarCAND2 study data. Intensive Care Med 41:1931–1940. https://doi.org/10.1007/s00134-015-4053-1
Guarascio AJ, Slain D, McKnight R et al (2013) A matched-control evaluation of an antifungal bundle in the intensive care unit at a university teaching hospital. Int J Clin Pharm 35:145–148. https://doi.org/10.1007/s11096-012-9712-5
Martínez-Jiménez MC, Muñoz P, Valerio M et al (2015) Combination of Candida biomarkers in patients receiving empirical antifungal therapy in a Spanish tertiary hospital: a potential role in reducing the duration of treatment. J Antimicrob Chemother 70:3107–3115. https://doi.org/10.1093/jac/dkv241
Rouzé A, Loridant S, Poissy J et al (2017) Biomarker-based strategy for early discontinuation of empirical antifungal treatment in critically ill patients: a randomized controlled trial. Intensive Care Med 43:1668–1677. https://doi.org/10.1007/s00134-017-4932-8
Bassetti M, Righi E, Ansaldi F et al (2015) A multicenter multinational study of abdominal candidiasis: epidemiology, outcomes and predictors of mortality. Intensive Care Med 41:1601–1610. https://doi.org/10.1007/s00134-015-3866-2
Kauffman CA (2015) Complications of candidemia in ICU patients: endophthalmitis, osteomyelitis, endocarditis. Semin Respir Crit Care Med 36:641–649. https://doi.org/10.1055/s-0035-1562891
Fernández-Cruz A, Cruz Menárguez M, Muñoz P et al (2015) The search for endocarditis in patients with candidemia: a systematic recommendation for echocardiography? A prospective cohort. Eur J Clin Microbiol Infect Dis 34:1543–1549. https://doi.org/10.1007/s10096-015-2384-z
Rodríguez-Adrián LJ, King RT, Tamayo-Derat LG et al (2003) Retinal lesions as clues to disseminated bacterial and candidal infections: frequency, natural history, and etiology. Med (Baltim) 82:187–202. https://doi.org/10.1097/01.md.0000076008.64510.f1
De Waele JJ, Vogelaers D, Blot S, Colardyn F (2003) Fungal infections in patients with severe acute pancreatitis and the use of prophylactic therapy. Clin Infect Dis 37:208–213. https://doi.org/10.1086/375603
Tsuruta R, Mizuno H, Kaneko T et al (2007) Preemptive therapy in nonneutropenic patients with Candida infection using the Japanese guidelines. Ann Pharmacother 41:1137–1143. https://doi.org/10.1345/aph.1K010
Hanson KE, Pfeiffer CD, Lease ED et al (2012) β-d-glucan surveillance with preemptive anidulafungin for invasive candidiasis in intensive care unit patients: a randomized pilot study. PLoS One 7:e42282. https://doi.org/10.1371/journal.pone.0042282
Ostrosky-Zeichner L, Shoham S, Vazquez J et al (2014) MSG-01: a randomized, double-blind, placebo-controlled trial of caspofungin prophylaxis followed by preemptive therapy for invasive candidiasis in high-risk adults in the critical care setting. Clin Infect Dis 58:1219–1226. https://doi.org/10.1093/cid/ciu074
Prattes J, Hoenigl M, Rabensteiner J et al (2014) Serum 1,3-beta-d-glucan for antifungal treatment stratification at the intensive care unit and the influence of surgery. Mycoses 57:679–686. https://doi.org/10.1111/myc.12221
Knitsch W, Vincent J-L, Utzolino S et al (2015) A randomized, placebo-controlled trial of preemptive antifungal therapy for the prevention of invasive candidiasis following gastrointestinal surgery for intra-abdominal infections. Clin Infect Dis 61:1671–1678. https://doi.org/10.1093/cid/civ707
Kollef M, Micek S, Hampton N et al (2012) Septic shock attributed to Candida infection: importance of empiric therapy and source control. Clin Infect Dis 54:1739–1746. https://doi.org/10.1093/cid/cis305
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
COIs declared by the authors: IML: Lectures: Thermofisher, Polyphor, J&J, Virogates, MSD. Advisory board: Fresenius Kabi, MaaT Pharma, Bayer, Gilead, Clinigen, Biotest, Accelerate. JGM has received speaker honoraria from Astellas and MSD. MB has participated in the past five years in advisory boards and/or received speaker honoraria from Achaogen, Angelini, Astellas, AstraZeneca, Bayer, Basilea, Cidara, Gilead, Menarini, MSD, Nabriva, Paratek, Pfizer, Roche, The Medicine Company, Shionogi, Tetraphase, VenatoRx, and Vifor. SSK has received honoraria for participating in advisory boards or as a speaker for Merck, Pfizer, Hikma, Pasteur Aventis, Gilead. JDW has consulted for Accelerate, AtoxBio, Bayer Healthcare, Cubist, MSD, Pfizer (honoraria were paid to his institution). PM: Personal fees and non-financial support from Astellas, Astrazeneca, Basilea, Bayer, Cubist, Menarini, MSD, Parexel, Pfizer, Tetraphase, and The Medicines Company unrelated to the submitted work. GD: Advisory boards and/or received speaker honoraria from Astellas, Bayer, Cidara, Gilead, MSD, Nabriva, Paratek, Pfizer, Tetraphase, Cipla India, Glenmark India, Infectopharm Germany. MCE has received grant support from Astellas Pharma, bioMerieux, Gilead Sciences, Merck Sharp & Dohme, Pfizer, Schering Plough, CIDARA, Amplyx, F2G, Scynexis, Soria Melguizo SA, and Ferrer International. He is a founding Partner of the start-up Micología Molecular SL.
Ethical approval
An approval by an ethics committee was not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Martin-Loeches, I., Antonelli, M., Cuenca-Estrella, M. et al. ESICM/ESCMID task force on practical management of invasive candidiasis in critically ill patients. Intensive Care Med 45, 789–805 (2019). https://doi.org/10.1007/s00134-019-05599-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-019-05599-w