Abstract
Purpose
Extent of lung contusion on initial computed tomography (CT) scan predicts the occurrence of acute respiratory distress syndrome (ARDS) in blunt chest trauma patients. We hypothesized that lung ultrasonography (LUS) on admission could also predict subsequent ARDS.
Methods
Forty-five blunt trauma patients were prospectively studied. Clinical examination, chest radiography, and LUS were performed on arrival at the emergency room. Lung contusion extent was quantified using a LUS score and compared to CT scan measurements. The ability of the LUS score to predict ARDS was tested using the area under the receiver operating characteristic curve (AUC-ROC). The diagnostic accuracy of LUS was compared to that of combined clinical examination and chest radiography for pneumothorax, lung contusion, and hemothorax, with thoracic CT scan as reference.
Results
Lung contusion extent assessed by LUS on admission was predictive of the occurrence of ARDS within 72 h (AUC-ROC = 0.78 [95 % CI 0.64–0.92]). The extent of lung contusion on LUS correlated well with CT scan measurements (Spearman’s coefficient = 0.82). A LUS score of 6 out of 16 was the best threshold to predict ARDS, with a 58 % [95 % CI 36–77] sensitivity and a 96 % [95 % CI 76–100] specificity. The diagnostic accuracy of LUS was higher than that of combined clinical examination and chest radiography: (AUC-ROC) 0.81 [95 % CI 0.50–1.00] vs. 0.74 [0.48–1.00] (p = 0.24) for pneumothorax, 0.88 [0.76–1.00] vs. 0.69 [0.47–0.92] (p < 0.05) for lung contusion, and 0.84 [0.59–1.00] vs. 0.73 [0.51–0.94] (p < 0.05) for hemothorax.
Conclusions
LUS on admission identifies patients at risk of developing ARDS after blunt trauma. In addition, LUS allows rapid and accurate diagnosis of common traumatic thoracic injuries.
Similar content being viewed by others
Introduction
Lung contusion is the most frequent thoracic injury in blunt chest trauma and it is associated with increased morbidity and mortality [1]. Direct damage of the lung tissue causes both local and systemic inflammatory responses that can lead to acute respiratory distress syndrome (ARDS) and multiple organ failure [2, 3]. The initial size of the lung contusion seems to play a key role in these mechanisms. Several CT scan studies have shown that initial lung contusion volume is predictive of the development of subsequent ARDS [4–7]. However, CT scan measurement of lung contusion volume requires 3-dimensional modelling that is not readily available. In addition, severely injured patients may have hemodynamic instability preventing transport to the CT suite. In this context, bedside ultrasonography appears to be the most appropriate diagnostic tool [8–11]. Recently, lung ultrasonography (LUS) has been proposed to evaluate common traumatic lung injuries and seems to be accurate [12]. However, the ability of LUS to assess the extent of lung contusion and its prognostic value have been poorly studied until now. To our knowledge, only the study by Rocco and colleagues addressed this issue in a small cohort [13]. They found a good correlation between LUS and thoracic CT scan for the initial extent of lung contusion, but did not assess its prognostic value.
We hypothesized that early assessment of lung contusion extent using LUS can predict the occurrence of ARDS in blunt trauma patients. We conducted a prospective observational study to evaluate the ability of LUS performed on admission of trauma patients to predict subsequent ARDS (primary outcome). In addition, we compared the performance of LUS with that of combined clinical examination and chest radiography (CXR) for diagnosing pneumothorax, lung contusion, and hemothorax (secondary outcomes).
Materials and methods
Patients
This prospective observational cohort study was conducted from May 2010 to July 2011 in the emergency room of the University Hospital of Angers, a level 1 trauma center. The institutional ethics committee approved the design of the study and waived requirement for informed consent from the patient (registration 2010/05).
In our hospital, severe trauma patients are referred to trained anesthesiologists in an emergency room located near the CT suite and the emergency operating theater (four operating rooms available 24/7). Patients with multiple blunt trauma were enrolled in the study if one of the physicians trained in LUS (i.e., with an experience of more than 30 LUS in trauma patients) was present. Demographic data, physiological parameters, and arterial blood gas measurements on admission were collected. Therapeutic decisions were left to the discretion of the physician in charge of the patient (different from the one who performed LUS).
Clinical examination and chest radiography
On arrival at the emergency room, the physician in charge of the patient performed a clinical examination, including bilateral inspection and auscultation of the chest. CXR was performed at the same time with a mobile x-ray machine (AMX-4, GE Healthcare, Waukesha, WI, USA). According to the clinical examination and CXR findings, the physician assessed the diagnosis probability of pneumothorax, lung contusion, and hemothorax for each lung using a four-point scale: 0 = sure of the absence of a lesion, 1 = doubt about the absence of a lesion, 2 = probable presence of a lesion, and 3 = sure of the presence of a lesion. A score of 2 or more was considered as a positive diagnosis [12].
Lung ultrasonography
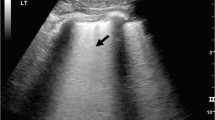
LUS was performed after completion of clinical examination by an anesthesiologist trained for LUS blinded to the clinical examination and CXR results. An M-turbo ultrasonograph with a 5–1 MHz probe (SonoSite, Bothell, WA, USA) was used. The chest wall was divided into eight areas, with the patient in the supine position (Fig. 1). In each of these areas, sonographic signs of pneumothorax, lung contusion, and hemothorax were investigated [8, 14]. The diagnostic criteria used are those of the international recommendations for LUS [8]. Briefly, pneumothorax was diagnosed in the presence of lung point(s) or absence of lung sliding together with absence of B lines and absence of lung pulse. Lung contusion was diagnosed by the presence of focal B lines (Fig. 1e in the Electronic supplementary material, ESM) and/or lung consolidation, and hemothorax was diagnosed as a dependent dark zone free of echo. The LUS operator recorded the diagnostic probability of each of these lesions using the same four-point scale as for the combination of clinical examination and CXR. LUS results could be given to the physician in charge if the patient had abnormal vital signs and required immediate treatment. To assess the extent of lung contusion, we defined a LUS score in each area: 0 = no contusion in the area, 1 = contusion in a part of the area, 2 = contusion in the whole area. By adding the scores of each of the eight areas, we obtained a total score ranging from 0 to 16 per patient.
Areas evaluated by lung ultrasonography on emergency room. Chest wall was separated into four quadrants on each side, according to the anterior axillary line (AAL) and the medio-thoracic line. Areas 1–4 on the right chest wall and areas 5–8 on the left chest wall. PSL parasternal line, PAL posterior axillary line
Thoracic CT scan
Thoracic acquisition was performed from the apex of the chest to the diaphragm using a 64-detector scanner (Optima CT660, GE Healthcare, Waukesha, WI, USA). Contiguous 1-mm-thick transverse views at lung window settings were systematically reconstructed and analyzed by a radiologist blinded to the results of other investigations (i.e., combination of clinical examination and CXR, and LUS). The time between admission to the emergency room and CT scan completion was recorded. Patients who underwent thoracic CT scan before being transferred to our hospital were included if the time between the CT scan and the admission was less than 120 min. In these cases, only the physician in charge of the patient knew the results of the previous CT scan, and two other physicians performed the combination of clinical examination and CXR, and LUS. Lung contusion on CT scan was described as apical-medial, apical-lateral, medial-basal, and/or lateral-basal in each lung. The following score was applied in each of these areas: 0 = no contusion in the area, 1 = contusion in a part of the area, 2 = contusion in the whole area. We obtained a CT scan score for the extent of lung contusion ranging from 0 to 16 per patient. The size of pneumothorax was classified as minuscule, anterior, or anterolateral, according to the classification described by Wolfman and colleagues [15]. The size of hemothorax was measured on the basis of the equation described by Hazlinger and colleagues [16]. All these trauma patients had a whole body CT scan together with this thoracic CT scan.
Outcomes
The occurrence of ARDS within the first 72 h after admission, the number of ventilator-free days (VFDs) by day 28, the ICU length of stay, and the in-hospital mortality were recorded. ARDS was defined according to the recent Berlin recommendation [17, 18]. We defined three cumulative categories of ARDS: severe (PaO2/FiO2 ≤ 100 mmHg), severe to moderate (PaO2/FiO2 ≤ 200 mmHg), and severe to mild (PaO2/FiO2 ≤ 300 mmHg).
Statistical analysis
Categorical variables were expressed as frequency and percentage; continuous data were presented as mean and standard deviation or median and interquartile range as appropriate. The predictive value of lung contusion extent assessed by early LUS and the development of ARDS within the 72 h after admission was tested using area under the receiver operating characteristic curve (AUC-ROC). The Youden index was used to calculate the best threshold value for predicting ARDS onset [19]. Quantification of lung contusion by LUS score was compared to that of CT scan using Spearman rank correlation analysis. Correlation was considered clinically relevant when the coefficient was 0.8 or greater. The diagnostic accuracy of the combination of clinical examination and CXR was compared to that of LUS for pneumothorax, lung contusion, and hemothorax, using CT scan as the reference and was expressed as sensitivity, specificity, likelihood ratios, and AUC-ROC. We compared the AUC of the different diagnostic tools using a test for dependent receiver operating characteristic curves (same sample); other comparisons were done using Wilcoxon or Fisher exact tests, as appropriated. Statistical analysis was performed using XLSTAT 2012 (Addinsoft). Statistical significance was defined as p < 0.05.
Results
Patients
During the study period, 219 patients were admitted to our emergency room for multiple blunt trauma. Among them, 50 patients had both a LUS performed on admission by one of the operators trained in LUS and a whole body CT scan. Five patients were not included, four because of an excessive delay between admission and CT scan (performed in another hospital), and one because they were less than 15 years old (Fig. 2 for flow chart). Table 1 shows the overall characteristics of the 45 patients included. The median [IQR] of the abbreviated injury scale for chest was 4 [3, 4]. LUS was successfully performed in all patients in less than 10 min. The median time between admission to the emergency room and thoracic CT scan was 39 [27–67] min.
Prediction of ARDS
Among the 41 patients alive at 72 h, 19 (46 %) developed an ARDS, including five severe, nine moderate, and five mild, according to the Berlin classification [17, 18]. The LUS score on admission was predictive of the occurrence of severe ARDS (AUC = 0.86 [0.77–0.96]), severe to moderate ARDS (AUC = 0.77 [0.61–0.92]), and severe to mild ARDS (AUC = 0.78 [0.64–0.92]). These results are reported on Fig. 3. A LUS score of 6 was identified as the best threshold value for predicting severe ARDS: sensitivity (Se) 80 %, specificity (Sp) 78 %, positive predictive value (PPV) 33 %, negative predictive value (NPV) 97 %; severe to moderate ARDS: Se 64 %, Sp 89 %, PPV 75 %, NPV 83 %; and severe to mild ARDS: Se 58 %, Sp 96 %, PPV 92 %, NPV 72 %. The extent of lung contusion assessed by LUS score on admission correlated with thoracic CT scan measurements, as shown by a Spearman’s coefficient of 0.82 (Fig. 2e, ESM).
ROC curves for the diagnosis of ARDS occurrence within 72 h, according to LUS score. ARDS acute respiratory distress syndrome, AUC-ROC area under the receiver operating characteristic curve. Cumulative categories of ARDS: severe (PaO2/FiO2 ≤ 100 mmHg), severe to moderate (PaO2/FiO2 ≤ 200 mmHg), severe to mild (PaO2/FiO2 ≤ 300 mmHg)
Main clinical outcomes
Overall mortality was 18 % (Table 1). PaO2/FiO2 ratios and tidal volumes on admission were not significantly different whether patients developed an ARDS or not (Table 1e, ESM). Patients with ARDS experienced significant longer ICU length of stay (median [IQR] 13 [5–17] vs. 1 [1, 2] days, p < 0.05), and higher mortality rate: 21 vs. 0 % (p < 0.05) (Table 1e, ESM).
Diagnostic accuracies for all thoracic injuries
Among the 90 lungs examined, CT scan revealed 30 pneumothorax, 60 lung contusions, and 25 hemothorax. The occurrence of these lesions was similar between right and left chest walls (respectively, 17 vs. 13 pneumothorax, p = 0.37; 30 vs. 30 lung contusions; 11 vs. 14 hemothorax, p = 0.48), which allowed us to analyze lung fields as separate entities. The AUC-ROC of LUS was significantly higher than the AUC-ROC of combined CE and CXR for lung contusion (p < 0.05) and hemothorax (p < 0.05) diagnosis (Table 2e, ESM; and Fig. 4). We found no significant difference between the two diagnostic modalities for pneumothorax diagnosis (p = 0.24). Twelve patients required chest drainage: three for hemothorax, four for pneumothoraces, and five for combined hemo- and pneumothoraces. All these lesions were identified by LUS.
Discussion
The present study showed that early LUS accurately predicts the occurrence of ARDS in blunt trauma patients. Lung contusion extent assessed by LUS at the bedside correlated well with CT scan findings. In addition, we found LUS superior to the combined CE and CXR for diagnosing lung contusion and hemothorax at the bedside.
As standard chest radiography underestimates lung contusion extension at the initial phase of trauma [13, 20], thoracic CT scan is the reference method [4–7]. However, lung contusion volume measurement by CT scan requires a specific computer-based algorithm allowing 3-dimensional modelling that is not routinely available in the emergency setting. Furthermore, severe trauma frequently causes circulatory failure in the initial phase that contraindicates the achievement of an early CT scan. In this context, LUS appears to be an attractive alternative [11], since it is feasible at the bedside with a good accuracy for diagnosing lung contusion [8, 12, 13, 21]. We confirmed here that LUS is accurate in assessing the extension of lung contusion. In addition, to our knowledge, this is the first study showing that LUS performed on admission can predict the occurrence of subsequent ARDS. We found that a LUS score of 6 or more out of 16 was predictive of ARDS. Whereas the specificity of this score was good, its sensitivity was relatively weak for predicting severe to moderate and severe to mild ARDS, respectively 64 and 58 %. There is thus a risk of “false positive” prediction. However, this can only benefit the patient by increasing doctor vigilance. This threshold corresponds to a volume of lung contusion greater than 30 % of the total lung volume. This result is consistent with the recent CT scan study by Becher et al. [4], finding a threshold of 24 % as a predictor of ARDS. This LUS score has a high positive likelihood ratio for the development of subsequent ARDS, and may thus have prognostic implications. Indeed, patients developing ARDS experienced longer duration of mechanical ventilation and ICU stay, and above all higher mortality. ARDS is indeed a frequent and devastating complication after severe blunt trauma [1]. As growing evidence supports the use of protective ventilation to prevent ARDS onset [22, 23], its early prediction becomes an important issue.
We found that LUS was superior to combined clinical examination and CXR for diagnosing lung contusion and hemothorax on admission of blunt trauma patients. These results are consistent with several previous studies [12, 13, 20, 24]. However, the sensitivity of LUS for diagnosing pneumothorax and hemothorax we found was weak. The first explanation for these quite low sensitivities is the high incidence of very small lesions in our study. Fourteen (33 %) pneumothorax were classified as “minuscule” and 10 (53 %) hemothorax were less than 100 mL. In comparison, none of the pneumothorax identified by CT scan on the study by Zhang and colleagues was minuscule, and only 5 (20 %) were in Soldati and colleagues’ study [20, 25]. Previous studies on hemothorax did not specify the percentage of small hemothorax on CT scan [13, 24]. Contrary to previous studies investigating only one type of lung lesion [20, 21, 24, 25], we chose not to exclude patients with subcutaneous emphysema, multiple trauma, or patients under mechanical ventilation. The presence of subcutaneous emphysema and/or pneumothorax could have prevented the diagnosis of underlying lesions. Hyacinthe and colleagues, who included consecutive patients without any selection, as in our study, also reported weak sensitivities for pneumothorax and hemothorax diagnosis [12]. Finally, it should be noted that all the pneumothorax and hemothorax requiring chest drainage in our study were diagnosed by LUS. Thus, the information provided by LUS seems sufficient to support appropriate clinical decision-making, as previously reported [9, 26].
Our study has methodological weaknesses. We did not assess interoperator variability because it was not feasible to mobilize two operators trained in LUS at the same time, in addition to the physician in charge of the patient. However, the study by Lichtenstein and colleagues reported low interoperator variability of LUS in intensive care patients [26]. We did not measure the delay between LUS and CT scan, but median CT scan delays from admission were quite low (39 [27–67] min), so that the delay between LUS and CT scan should be less than 90 min as requested in a previous study [12]. We did not use specific 3-dimensional modelling software for the measurement of lung contusion volume on CT scan, as it was not available in our hospital at the time of the study. Regarding the LUS examination, we made some assumptions: we assume that the findings on LUS represent lung contusion, but there are many causes of B lines; we also assume that pleural effusions were related to hemothorax, without sampling all of them. However, these assumptions are the same with the CT scan findings. Finally, we did not assess the posterior regions of the lungs, because trauma patients have to remain supine. We could thus have underestimated the volume of lung contusion by LUS examination.
In our view, these results could have clinical implications. LUS seems to provide relevant data. LUS could thus be added to the Extended Focused Assessment with Sonography for Trauma (EFAST) [27], not only for lung contusion diagnosis but also for the diagnosis of hemothorax and pneumothorax. The development of web-based assessment tools for the EFAST could help in this way [22, 27]. In addition, the early detection of blunt trauma patients at risk of ARDS could indicate closer surveillance of these patients. The impact of an early intervention such as protective ventilation in these patients could also be assessed. Indeed, the retrospective study by Ciesla and colleagues had already suggested that protective ventilation could contribute to the reduction of post-traumatic ARDS [22]. Recent data supports the generalization of this strategy prophylactically to almost all mechanical ventilated patients in intensive care [23].
Conclusion
Lung contusion extent assessed by LUS on admission identifies patients at risk of developing ARDS within 72 h after a severe blunt trauma when their LUS score is higher than 6. LUS is a rapid and accurate tool for diagnosing main traumatic lung injuries at the bedside. These results argue for systematic assessment of lung contusion extent by LUS in the initial phase of blunt trauma, as part of the EFAST.
References
Allen GS, Coates NE (1996) Pulmonary contusion: a collective review. Am Surg 62:895–900
Ciesla DJ, Moore EE, Johnson JL, Burch JM, Cothren CC, Sauaia A (2005) The role of the lung in postinjury multiple organ failure. Surgery 138:749–757
Raghavendran K, Notter RH, Davidson BA, Helinski JD, Kunkel SL, Knight PR (2009) Lung contusion: inflammatory mechanisms and interaction with other injuries. Shock 32:122–130
Becher RD, Colonna AL, Enniss TM, Weaver AA, Crane DK, Martin RS, Mowery NT, Miller PR, Stitzel JD, Hoth JJ (2012) An innovative approach to predict the development of adult respiratory distress syndrome in patients with blunt trauma. J Trauma Acute Care Surg 73:1229–1235
Miller PR, Croce MA, Bee TK, Qaisi WG, Smith CP, Collins GL, Fabian TC (2001) ARDS after pulmonary contusion: accurate measurement of contusion volume identifies high-risk patients. J Trauma 51:223–230
Strumwasser A, Chu E, Yeung L, Miraflor E, Sadjadi J, Victorino GP (2011) A novel CT volume index score correlates with outcomes in polytrauma patients with pulmonary contusion. J Surg Res 170:280–285
Wang S, Ruan Z, Zhang J, Jin W (2011) The value of pulmonary contusion volume measurement with three-dimensional computed tomography in predicting acute respiratory distress syndrome development. Ann Thorac Surg 92:1977–1983
Volpicelli G, Elbarbary M, Blaivas M, Lichtenstein DA, Mathis G, Kirkpatrick AW, Melniker L, Gargani L, Noble VE, Via G, Dean A, Tsung JW, Soldati G, Copetti R, Bouhemad B, Reissig A, Agricola E, Rouby JJ, Arbelot C, Liteplo A, Sargsyan A, Silva F, Hoppmann R, Breitkreutz R, Seibel A, Neri L, Storti E, Petrovic T (2012) International evidence-based recommendations for point-of-care lung ultrasound. Intensive Care Med 38:577–591
Xirouchaki N, Kondili E, Prinianakis G, Malliotakis P, Georgopoulos D (2014) Impact of lung ultrasound on clinical decision making in critically ill patients. Intensive Care Med 40:57–65
Mayo PH (2013) Critical care ultrasonography: the Italian approach. Intensive Care Med 39:1849–1850
Volpicelli G, Lamorte A, Tullio M, Cardinale L, Giraudo M, Stefanone V, Boero E, Nazerian P, Pozzi R, Frascisco MF (2013) Point-of-care multiorgan ultrasonography for the evaluation of undifferentiated hypotension in the emergency department. Intensive Care Med 39:1290–1298
Hyacinthe AC, Broux C, Francony G, Genty C, Bouzat P, Jacquot C, Albaladejo P, Ferretti GR, Bosson JL, Payen JF (2012) Diagnostic accuracy of ultrasonography in the acute assessment of common thoracic lesions after trauma. Chest 141:1177–1183
Rocco M, Carbone I, Morelli A, Bertoletti L, Rossi S, Vitale M, Montini L, Passariello R, Pietropaoli P (2008) Diagnostic accuracy of bedside ultrasonography in the ICU: feasibility of detecting pulmonary effusion and lung contusion in patients on respiratory support after severe blunt thoracic trauma. Acta Anaesthesiol Scand 52:776–784
Bouhemad B, Zhang M, Lu Q, Rouby JJ (2007) Clinical review: bedside lung ultrasound in critical care practice. Crit Care 11:205
Wolfman NT, Myers WS, Glauser SJ, Meredith JW, Chen MY (1998) Validity of CT classification on management of occult pneumothorax: a prospective study. AJR Am J Roentgenol 171:1317–1320
Hazlinger M, Ctvrtlik F, Langova K, Herman M (2014) Quantification of pleural effusion on CT by simple measurement. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 158:107–111
Ferguson ND, Fan E, Camporota L, Antonelli M, Anzueto A, Beale R, Brochard L, Brower R, Esteban A, Gattinoni L, Rhodes A, Slutsky AS, Vincent JL, Rubenfeld GD, Thompson BT, Ranieri VM (2012) The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med 38:1573–1582
Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, Fan E, Camporota L, Slutsky AS (2012) Acute respiratory distress syndrome: the Berlin definition. JAMA 307:2526–2533
Youden WJ (1950) Index for rating diagnostic tests. Cancer 3:32–35
Soldati G, Testa A, Sher S, Pignataro G, La Sala M, Silveri NG (2008) Occult traumatic pneumothorax: diagnostic accuracy of lung ultrasonography in the emergency department. Chest 133:204–211
Soldati G, Testa A, Silva FR, Carbone L, Portale G, Silveri NG (2006) Chest ultrasonography in lung contusion. Chest 130:533–538
Ciesla DJ, Moore EE, Johnson JL, Cothren CC, Banerjee A, Burch JM, Sauaia A (2006) Decreased progression of postinjury lung dysfunction to the acute respiratory distress syndrome and multiple organ failure. Surgery 140:640–648
Lellouche F, Lipes J (2013) Prophylactic protective ventilation: lower tidal volumes for all critically ill patients? Intensive Care Med 39:6–15
Brooks A, Davies B, Smethhurst M, Connolly J (2004) Emergency ultrasound in the acute assessment of haemothorax. Emerg Med J 21:44–46
Zhang M, Liu ZH, Yang JX, Gan JX, Xu SW, You XD, Jiang GY (2006) Rapid detection of pneumothorax by ultrasonography in patients with multiple trauma. Crit Care 10:R112
Lichtenstein D, Goldstein I, Mourgeon E, Cluzel P, Grenier P, Rouby JJ (2004) Comparative diagnostic performances of auscultation, chest radiography, and lung ultrasonography in acute respiratory distress syndrome. Anesthesiology 100:9–15
Nandipati KC, Allamaneni S, Kakarla R, Wong A, Richards N, Satterfield J, Turner JW, Sung KJ (2011) Extended focused assessment with sonography for trauma (EFAST) in the diagnosis of pneumothorax: experience at a community based level I trauma center. Injury 42:511–514
Acknowledgments
The authors thank Professor Jean-Jacques Rouby for his helpful advice about the manuscript. Support was provided solely from institutional and/or departmental sources. All authors are full-time employees of their respective institutions and did not receive any extra financial support to perform the present study.
Conflicts of interest
None of the authors has a conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Additional information
Take-home message: Lung ultrasonography score performed at admission for blunt trauma allows an accurate quantification of lung contusion. A score greater than 6 is predictive of the development of ARDS within 72 h.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Leblanc, D., Bouvet, C., Degiovanni, F. et al. Early lung ultrasonography predicts the occurrence of acute respiratory distress syndrome in blunt trauma patients. Intensive Care Med 40, 1468–1474 (2014). https://doi.org/10.1007/s00134-014-3382-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-014-3382-9