Abstract
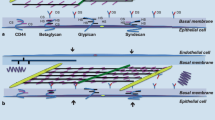
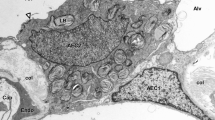
Pulmonary gas exchange critically depends upon the hydration state and the thinness of the interstitial tissue layer within the alveolo-capillary membrane. In the interstitium, fluid freely moving within the fibrous extracellular matrix (ECM) equilibrates with water chemically bound to hyaluronic acid and proteoglycans (PGs). The dynamic equilibrium between these two phases is set and maintained by the transendothelial fluid and solutes exchanges, by the convective outflows into the lymphatic system, and by the mechanical and hydrophilic properties of the solid elements of the ECM. The fibrous ECM components, in particular the chondroitin sulfate proteoglycan (CS-PG) and the heparan-sulfate proteoglycan (HS-PG) families, play a major role in the maintenance of tissue fluid homeostasis. In fact, they provide: (a) a perivascular and interstitial highly restrictive sieve with respect to plasma proteins, thus modulating both interstitial protein concentration and transendothelial fluid filtration; (b) a mechanical support to lymphatic vessels sustaining and modulating their draining function, and (c) a rigid three-dimensional low-compliant scaffold opposing fluid accumulation into the interstitial space. Fragmentation of PG induced by increased plasma volume, by degradation through proteolytic or inflammatory agents, by exposure to inspiratory gas mixture with modified oxygen fraction, or by increased tissue strain/stress invariably results in the progressive loosening of PG intermolecular bonds with other ECM components. The loss of the PGs regulatory functions compromises the protective role of the tissue solid matrix progressively leading to interstitial and eventually severe lung edema.



Similar content being viewed by others
References
Weibel ER, Bachofen H (1997) The fiber scaffold of lung parenchyma. In: Crystal RG, West JB (eds) The lung, vol 1. Scientific Foundations, Raven Press, New York, pp 1139–1146
Skold CM, Blaschke E, Eklund A (1996) Transient increases in albumin and hyaluronan in broncoalveolar lavage fluid after quitting smoking: possible signs of reparative mechanisms. Respir Med 90:523–529
Li Y, Rahmanian M, Widstrom C, Lepperdinger G, Frost GI, Heldin P (2000) Irradiation induced expression of hyaluronan (HA) synthase 2 and hyaluronidase 2 genes in rat lung tissue accompanies active turnover of HA and induction of types I and III collagen gene expression. Am J Resp Cell Mol Biol 23:411–418
Cantor JO, Shteyngart B, Cerreta JM, Lui M, Armand G, Turino GM (2000) The effect of hyaluronan on elastic fibers injury in vitro and elastase-induced airspace enlargement in vivo. Exp Biol Med 225:65–71
Hardingham T, Fosang AJ (1992) Proteoglycans: many forms and many functions. FASEB J 6:861–870
Roberts CR, Wight TN, Hascall VC (1997) Proteoglycans. In: Crystal RG, West JB, Weibel ER, Barnes PJ (eds) The lung: scientific foundations, 2nd edn. Lippincott-Raven, Philadelphia, pp 757–767
Iozzo RV, Murdoch AD (1996) Proteoglycans of the extracellular environment: clues from the gene and protein side offer novel perspective in molecular diversity and function. FASEB J 10:598–614
Crouch EC, Martin GR, Brody JS, Laurie GW (1997) Basement membrane. In: Crystal RG, West JB, Weibel ER, Barnes PJ (eds) The lung: scientific foundations, 2nd edn. Lippincott-Raven, Philadelphia, pp 757–767
Yurchenko PD, Schittny JC (1990) Molecular architecture of basement membrane. FASEB J 4:1577–1590
Juul SE, Kinsella MG, Wight TN, Hodson WA (1993) Alterations in non-human primates (M. nemestrina) lung proteoglycans during normal development and acute hyaline membrane disease. Am J Resp Cell Mol Biol 8:299–310
Farquhar MG (1981) The glomerular basement membrane. A selective macromolecular filter. In: Hay E (ed) Cell biology of extracellular matrix. Plenum Press, New York, pp 335–378
Zhao J, Sime PJ, Bringas P Jr, Gauldie J, Warburton D (1999) Adenovirus-mediated decorin gene transfer prevents TGF-beta-induced inhibition of lung morphogenesis. Am J Physiol Lung Mol Cell Physiol 277:L412–L422
Miserocchi G, Negrini D, Gonano C (1990) Direct measurements of interstitial pulmonary pressure in in situ lung with intact pleural space. J Appl Physiol 69:2168–2174
Miserocchi G, Negrini D, Gonano C (1991) Parenchymal stress affects interstitial and pleural pressure in in situ lung. J Appl Physiol 71:1967–1972
Miserocchi G, Nakamura T, Mariani E, Negrini D (1981) Pleural liquid pressure over the interlobar, mediastinal and diaphragmatic surfaces of the lung. Respir Physiol 46:61–69
Miserocchi G, Kelly S, Negrini D (1988) Pleural and extrapleural interstitial liquid pressure measured by cannulas and micropipettes. J Appl Physiol 65:555–562
Negrini D, Cappelli C, Morini M, Miserocchi G (1987) Gravity dependent distribution of parietal subpleural interstitial pressure. J Appl Physiol 63:1912–1918
Bert J, Pearce R (1984) The interstitium and microvascular exchange. In: Michel CC (ed) Handbook of physiology. The American Physiological Society, Bethesda Maryland, pp 521–547
Laurent T (1964) The interaction between polysaccariddes and other macromolecules. 9. The exclusion of molecules from hyaluronic acid gels and solutions. J Biochem 93:106–112
Wiig H, Tenstad O (2001) Interstitial exclusion of positively and negatively charged IgG in rat skin and muscle. Am J Physiol 280:H1505–H1512
Negrini D, Tenstad O, Wiig H (2003) Interstitial exclusion of albumin in rabbit lung measured with the continuous method in combination with the wick technique. Microcirculation 10:153–165
Negrini D, Tenstad O, Wiig H (2003) Interstitial exclusion of albumin in rabbit during development of pulmonary edema. J Physiol 548.3:907–917
Negrini D, Passi A, Bertin K, Bosi F, Wiig H (2001) Isolation of pulmonary interstitial fluid in rabbits by a modified wick technique. Am J Physiol Lung Cell Mol Physiol 280:L1057–L1065
Aukland K, Reed RK (1993) Interstitial-lymphatic mechanisms in the control of extracellular fluid volume. Physiol Rev 73:1–78
Negrini D, Gonano C, Miserocchi G (1992) Microvascular pressure profile in intact in situ lung. J Appl Physiol 72:332–339
Taylor AE, Granger DN (1984) Exchange of macromolecules across the microcirculation. In: Renkin EM, Michel CC (eds) Handbook of physiology. The cardiovascular system. The microcirculation. American Physiological Society, Bethesda, Maryland, pp 467–520
Negrini D, Fabbro M del (1999) Subatmospheric pressure in the rabbit pleural lymphatic network J Physiol 520:761–769
Negrini D, Moriondo A; Mukenge S (2004) Transmural pressure during cardiogenic pressure oscillations in rodent diaphragmatic lymphatic vessels. Lymphatic Res Biol 2:69–81
Moriondo A, Mukenge S, Negrini D (2006) Transmural pressure in rat initial subpleural lymphatics during spontaneous or mechanical ventilation. Am J Physiol Heart Circ Physiol 289:H263–H269
Miserocchi G, Negrini D, Fabbro M del, Venturoli D (1993) Pulmonary interstitial pressure in intact in situ lung: the transition to interstitial edema. J Appl Physiol 74:1171–1177
Negrini D, Passi A, Luca G de, Miserocchi G (1996) Pulmonary interstitial pressure and proteoglycans during development of pulmonary edema. Am J Physiol Heart Circ Physiol 39: 270:H2000–H2007
Negrini D, Passi A, Luca G de, Miserocchi G (1998) Proteoglycan involvement during development of lesional pulmonary edema. Am J Physiol Lung Cell Mol Physiol 274:L203–L211
Passi A, Negrini D, Albertini R, De Luca G, Miserocchi G (1998) Involvement of lung interstitial proteoglycans in development of hydraulic- and elastase-induced edema. Am J Physiol Lung Cell Mol Physiol 275:L631–L635
Miserocchi G, Passi A, Negrini D, Luca G de, Fabbro M del (2001) Pulmonary interstitial pressure and tissue matrix structure in acute hypoxia. Am J Physiol Lung Cell Mol Physiol 280:L881–L887
Negrini D, Tenstad O, Passi A, Wiig H (2006) Differential degradation of matrix proteoglycans and edema development in rabbit lung. Am J Physiol Lung Cell Mol Physiol 290:L470–L477
Venkatesan N, Ebihara T, Roughley PJ, Ludwig M (2000) Alterations in large and small proteoglycans in bleomycin-induced pulmonary fibrosis in rats. Am J Resp Crit Care Med 161:2066–2073
Venkatesan N, Roughley PJ, Ludwig M (2002) Proteoglycan expression in bleomycin lung fibroblasts: role of transforming grow factor-β1 and interferon-γ. Am J Physiol Lung Cell Mol Physiol 283:L806–L814
Pittet JF, Griffiths MJD, Geiser T, Kaminski N, Dalton SL, Huang X, Brown LAS, Gotwals PJ, Koteliansky VE, Matthay MA, Shepparf D (2001) TGF-β is a critical mediator of acute lung injury. J Clin Invest 107:1537–1544
Ohnishi K, Takagi M, Kurokawa Y, Satomi S, Konttinen YT (1998) Matrix metalloproteinase-mediated extracellular matrix protein degradation in human pulmonary emphysema. Lab Invest 78:1077–1087
Woessner JF (1991) Matrix metalloproteinases and their inhibitors in connective tissue remodelling. FASEB J 5:2145–2154
Parks WC, Shapiro SD (2001) Matrix proteinases in lung biology. Respir Res 2:10–19
Passi A, Negrini D, Albertini R, Miserocchi G, Luca G de (1999) The sensitivity of versican from rabbit lung to gelatinase A (MMP-2) and B (MMP-9) and its involvement in the development of hydraulic lung edema. FEBS Lett 456:3–6
McElvaney NG, Crystal RG (1997) Proteases and lung injury. In: Crystal RG, West JB, Weibel ER, Barnes PJ (eds) The lung: scientific foundations, 2nd edn. Lippincott-Raven, Philadelphia, pp 2205–2218
Brew K, Dinakarpadian D, Nagase H (2000) Tissue inhibitors of metalloproteinases: evolution, structure and function. Biochem Biophys Acta 1477:267–283
McElvaney NG, Crystal RG (1997) Antiproteases and lung defense. In: Crystal RG, West JB, Weibel ER, Barnes PJ (eds) The lung: scientific foundations, 2nd edn. Lippincott-Raven Philadelphia, pp 2219–2235
Vender RL (1996) Therapeutic potential of neutrophil-elastase inhibition in pulmonary disease. J Invest Med 44:531–539
Finlay GA, O'Driscoll LR, Russel LR, D'Arcy EM, Masterson JB, FitzGerald MX, O'Connor CM (1997) Matrix metalloproteinase expression and production by macrophages in emphysema. Am J Resp Crit Care Med 156:240–247
Ohnishi K, Takagi M, Kurokawa Y, Satomi S, Konttiken YT (1998) Matrix metalloproteasis-mediated extracellular matrix protein degradation in human pulmonary emphysema. Lab Invest 78:1077–1087
Fukuda Y, Ishazaki M, Kudoh S, Kitaichi M, Yamanaka M (1998) Localization of matrix metalloproteinases -1, -2, -9 and tissue inhibitor of matalloproteinases-2 in lung disease. Lab Invest 78:687–689
Pardo A, Borrios R, Maldonado V, Melendez J, Perez J, Ruiz V, Segura Valdez L, Sznaider JI, Selman M (1998) Gelatinases A and B are upregulated in rat lungs by subacute hyperoxia: pathogenetic implications. Am J Pathol 153:833–844
Negrini D, Moriondo A, Passi A, Viola M, Marcozzi C, Pelosi P, Severgnini P, Ottani V, Quaranta M (2006) Pulmonary extracellular matrix fragmentation induced by mechanical ventilation. #4476. J Biomech 39:599
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is discussed in the editorial available at: http://dx.doi.org/10.1007/s00134-007-0965-8.
Rights and permissions
About this article
Cite this article
Negrini, D., Passi, A. & Moriondo, A. The role of proteoglycans in pulmonaryedema development. Intensive Care Med 34, 610–618 (2008). https://doi.org/10.1007/s00134-007-0962-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-007-0962-y