Abstract
Aim/hypothesis
We examined whether the renin inhibitor, aliskiren, provides similar or greater protection than ACE inhibition from non-proliferative diabetic retinopathy and from the proliferative neoangiogenesis of oxygen-induced retinopathy.
Methods
Transgenic (mRen-2)27 rats, which overexpress mouse renin and angiotensin in extra-renal tissues, were studied. For diabetic studies, non-diabetic, diabetic (streptozotocin, 55 mg/kg), diabetic + aliskiren (10 mg kg−1 day−1, pump), or diabetic + lisinopril (10 mg kg−1 day−1, drinking water) rats were evaluated over 16 weeks. For oxygen-induced retinopathy studies, rats were exposed to 80% oxygen (22 h/day) from postnatal days 0 to 11, and then room air from postnatal days 12 to 18. Aliskiren (10 or 30 mg kg−1 day−1, pump) or lisinopril (10 mg kg−1 day−1, drinking water) was administered during retinopathy development between postnatal days 12 and 18.
Results
Systolic BP in diabetic (mRen-2)27 rats was reduced with 10 mg kg−1 day−1 aliskiren, but only lisinopril normalised systolic blood pressure. In diabetic (mRen-2)27 rats, 10 mg kg−1 day−1 aliskiren and lisinopril reduced retinal acellular capillaries and leucostasis to non-diabetic levels. In oxygen-induced retinopathy, neoangiogenesis and retinal inflammation (leucostasis, ED-1 immunolabelling) were partially reduced by 10 mg kg−1 day−1 aliskiren and normalised by 30 mg kg−1 day−1 aliskiren, whereas lisinopril normalised neoangiogenesis and reduced leucostasis and ED-1 immunolabelling. Aliskiren and lisinopril normalised retinal vascular endothelial growth factor expression; however, only aliskiren reduced intercellular adhesion molecule-1 to control levels.
Conclusions/interpretation
Aliskiren provided similar or greater retinal protection than ACE inhibition and may be a potential treatment for diabetic retinopathy.
Similar content being viewed by others
Introduction
Growth factor, metabolic and haemodynamic pathways have been identified as participating in the development of diabetic retinopathy [1]. One such pathway is the renin–angiotensin system (RAS) as there is evidence that a local RAS including renin exists within the retina [2–4], and that prorenin is elevated in the plasma and vitreous humour of patients with proliferative diabetic retinopathy [5, 6]. Furthermore, experimental studies indicate that ACE inhibition and type 1 angiotensin receptor blockade (ARB) are protective in models of retinopathy [7–11]. These findings have, to some extent, been translated into clinical trials such as the DIabetic REtinopathy Candesartan Trial (DIRECT), which reported that ARB reduced the incidence of retinopathy in type 1 diabetic patients and increased regression of retinopathy in type 2 diabetic patients [12, 13]. ACE inhibition and ARB have also been reported to reduce the progression of diabetic retinopathy in normoalbuminuric and normotensive patients [14]. On the other hand, other randomised controlled clinical trials have reported ACE inhibition to be either equally effective as other blood-pressure-lowering strategies [15, 16], or to have no benefit [17]. The reasons for these variable findings are not fully understood, but possible explanations include uncertainty as to whether all ACE inhibitors and ARBs reach therapeutic levels sufficient to suppress the local RAS in the eyes of patients with diabetic retinopathy. In addition, the RAS may not be completely inhibited with ACE inhibition and ARB because of compensatory feedback mechanisms that result in increased renin production and release.
Aliskiren is a potent inhibitor of human and mouse renin, with the IC50 for human renin being 0.6 nmol/1, mouse renin 4.5 nmol/1 and rat renin 90 nmol/l [18]. For this reason, pre-clinical studies of aliskiren predominately use mouse models of disease or the transgenic (mRen-2)27 (Ren-2) rat [18, 19]. The Ren-2 rat was developed by the introduction of the mouse Ren-2 gene (also known as Ren2) into the genome of the Sprague–Dawley rat, and exhibits fulminant hypertension and overactivity of the RAS in extra-renal tissues [20]. With streptozotocin, the Ren-2 rat develops accelerated diabetic retinopathy and other complications, and serves as a model of type 1 diabetes [8, 21]. To our knowledge, the effects of aliskiren on experimental diabetic retinopathy have not been evaluated. The aim of this study was to determine if aliskiren is as effective as ACE inhibition in reducing vascular pathology in Ren-2 rats with diabetic retinopathy. As diabetic rodents do not progress to the angiogenic phase of proliferative diabetic retinopathy, we also compared the effects of aliskiren and ACE inhibition in Ren-2 rats with oxygen-induced retinopathy (OIR), a model of marked neoangiogenesis and inflammation in neonatal animals [22].
Methods
Animals
Procedures complied with the Alfred Medical Research and Education Precinct Animal Ethics Committee, Melbourne, VIC, Australia. Animals had free access to normal chow and drinking water and were housed in a stable environment (22 ± 1°C, 12 h light–dark cycle).
Diabetes in Ren-2 rats
Female homozygous transgenic Ren-2 rats weighing 180–210 g were randomised to the following groups: non-diabetic control; diabetic control; diabetic + aliskiren; and diabetic + lisinopril. To model type 1 diabetes, rats were fasted overnight and then administered streptozotocin (55 mg/kg, tail vein, Sigma-Aldrich, Castle Hill, NSW, Australia) [8, 11]. Non-diabetic rats received 0.1 mol/l citrate buffer (tail vein). Each week, rats were weighed and blood glucose levels measured (Accu-check Advantage II monitor; Roche Diagnostics, Castle Hill, NSW, Australia). Only rats with blood glucose >15 mmol/l were considered diabetic and received insulin every 2 days (2–4 U i.p.; Humulin NPH, Eli Lilly, Indianapolis, IN, USA). Aliskiren and lisinopril were administered from 2 days after the induction of diabetes and continued for 16 weeks. Aliskiren (Novartis, East Hanover, NJ, USA) was administered at 10 mg kg−1 day−1 (in 0.9% NaCl, wt/vol.) at a flow rate of 2.5 μl/h by minipump (Alzet; 2ML4, Cupertino, CA, USA) inserted subcutaneously in the flank. Pumps were changed every 4 weeks. Lisinopril (Novartis) was administered in drinking water at 10 mg kg−1 day−1. Every 4 weeks, systolic blood pressure (BP) was determined in preheated conscious rats via tail-cuff plethysmography [8]. Comparisons were with female non-diabetic Sprague–Dawley rats.
OIR in Ren-2 rats
On postnatal day (PD) 0, homozygous Ren-2 rat pups and mothers were randomised to: sham control; sham + aliskiren (10 mg kg−1 day−1); sham + lisinopril (10 mg kg−1 day−1); OIR control; OIR + aliskiren (10 mg kg−1 day−1); OIR + aliskiren (30 mg kg−1 day−1); or OIR + lisinopril (10 mg kg−1 day−1). From PD0 to PD11, pups were exposed to 80% oxygen (wt/wt) for 22 h/day and room air for 2 h/day, a process that suppresses retinal physiological angiogenesis [7, 9]. On PD12, pups were placed in room air until PD18, which induces an increase in angiogenic and inflammatory factors, and neoangiogenesis [7, 9]. Shams were Ren-2 rats exposed to room air from birth until PD18. Aliskiren was delivered to pups between PD12 to PD18 by minipump (1007D, Alzet) at 0.5 μl/h (100 μl volume) inserted subcutaneously in the flank. Lisinopril was administered between PD12 to PD18 in drinking water to dams.
OIR in C57Bl/6 mice
To determine if aliskiren reduced neoangiogenesis in normotensive animals, OIR was induced in C57Bl/6 mice [23]. On PD7, pups and their mothers were randomised to sham control, OIR control or OIR + aliskiren (30 mg kg−1 day−1). From PD7 to PD11, pups were exposed to 75% oxygen, and on PD12 were placed in room air until PD18. Shams were mice exposed to room air from birth until PD18. As minipumps are not available for neonatal mice, aliskiren was delivered by daily i.p. injection between PD12 to PD18. Prior to tissue collection, all animals were anaesthetised with pentobarbital sodium (170 mg/ml, Virbac, Peakhurst, NSW, Australia).
Acellular capillaries in diabetic Ren-2 rats
Acellular capillaries were counted in trypsin digests of retina as previously described [24]. Separate quantification was performed for central retina (0 to 1,666 ± 166 μm from the optic disc), mid retina (1,667 to 3,050 ± 154 μm) and peripheral retina (3,051 to 4,333 ± 128 μm). Approximately 80 (×40 objective) non-overlapping photomicrographs were taken from each retina (15 central, 30 mid, 35 peripheral regions) with a Spot digital camera (SciTech, Preston, VIC, Australia) attached to an Olympus BX51 microscope (Olympus, Tokyo, Japan). Results are expressed as the number of acellular capillaries per mm2 in the central, mid and peripheral retina.
Neoangiogenesis in OIR
Eyes were processed into paraffin, serially sectioned at 3 μm, and blood vessel profiles (BVPs) quantified in four non-overlapping fields of retina/section, with six sections/retina at least 60 μm apart evaluated per rat [9]. A BVP was defined as an endothelial cell or a blood vessel with a lumen. Intraretinal BVPs (ganglion cell layer [GCL] and inner plexiform layer [IPL]) and preretinal BVPs (protruding into the vitreous cavity while attached to the retinal surface) were counted separately [9].
Wholemounts of mouse retina were prepared as described previously [3, 25]. Briefly, eyes were fixed in 4% paraformaldehyde (wt/vol.) for 30 min and retina dissected in 0.1 mol/l PBS, pH 7.4, and flatmounted. Retinas were stained with FITC-conjugated Griffonia (Bandeiraea) simplicifolia BS-I lectin (1:100, Sigma-Aldrich) in 1% wt/vol. Triton X-100 (Sigma-Aldrich) in 0.1 mol/l PBS overnight. Retinas were washed with PBS for 6 × 15 min and mounted with media (DakoCytomation, Glostrup, Denmark). Images were taken at ×100 magnification using an AxioCam MRc 6.1.0.0 digital camera attached to a Zeiss Axio X1 microscope (Carl Zeiss, Germany). Retinal montages were constructed using the tiling tool in AxioObserver software (version 5.3, Carl Zeiss). Neovascular tufts were defined as clumps of blood vessels positively labelled for BS-I lectin. Using the ‘Select colour range’ tool in Adobe Photoshop Elements 6 (Adobe Systems Incorporated, USA), neovascular tufts labelled for BS-I lectin were selected over the entire retina and expressed as the percentage of BS-I lectin labelled neovascular tufts/retina. To quantify avascular retina, BS-I lectin negative areas of retina were traced in the mid-central retina. Results were expressed as the percentage of avascular area/retina (square centimetres).
Histopathology
Eyes were processed into paraffin, serially sectioned at 3 μm, and every tenth section from each eye (approximately 30 sections/animal) stained with haematoxylin and eosin (H&E) and evaluated for histopathology as described previously [7, 8].
Leucostasis in diabetes and OIR in Ren-2 rats
Rhodamine–concanavalin A labelled adherent leucocytes were counted in blood vessels of central, mid and peripheral retina of retinal flatmounts and expressed as adherent leucocytes/retina [9]. In the diabetic study, separate quantification was performed for central retina (0 to 1,816 ± 74 μm from the optic disc), mid retina (1,817 to 3,266 ± 34 μm) and peripheral retina (3,267 to 4,667 ± 68 μm). In the OIR study, separate quantification was performed for central retina (0 to 1,316 ± 151 μm from the optic disc), mid retina (1,317 to 2,466 ± 72 μm) and peripheral retina (2,467 to 3,533 ± 42 μm).
Immunohistochemistry for ED-1 (CD68) in OIR in Ren-2 rats
Paraffin sections, 3 μm, were incubated overnight at 4°C with 1:50 ED-1 antibody (mouse anti-rat CD68, MorphoSys UK, Oxford, UK) [9]; negative controls were sections without ED-1 antibody. Four non-overlapping fields/sections were photographed at ×200 magnification and Analytical Imaging Analysis Software (Imaging Research, St Catharines, ON, Canada) was used to outline the GCL and IPL. Hue and saturation were adjusted to detect the same intensity of immunolabelling in each field of retina. Results were expressed as immunolabelling/proportional field of inner retina. Six sections at least 60 μm apart were quantified from each retina from at least six rats per group.
Real-time PCR in OIR in Ren-2 rats
Total RNA from retina was isolated, subjected to DNase treatment and reverse transcribed as described previously [9]. Vascular endothelial growth factor (Vegf [also known as Vegfa]) and intercellular adhesion molecule-1 (Icam-1 [also known as Icam1]) mRNA and 18S ribosomal RNA were amplified and quantitative real-time PCR conducted [9]. Fold differences in gene expression relative to sham controls were calculated using the \( {2^{{ - \Delta \Delta {{\text{C}}_{\text{t}}}}}} \) method.
ELISA in OIR in Ren-2 rats
Retinas were homogenised in 400 μl PBS containing protease inhibitors (Sigma-Aldrich), the lysate was centrifuged and VEGF and ICAM-1 levels measured in the supernatant fraction using ELISA kits (R&D Systems, Minneapolis, MN, USA). VEGF and ICAM-1 protein levels were normalised to total protein concentration of each supernatant fraction as determined by a Bradford protein assay (Bio-Rad, Gladesville, NSW, Australia).
Measurement of plasma active renin in diabetes and OIR in Ren-2 rats
Blood was collected from the heart into tubes on ice containing heparin. Samples were centrifuged and the plasma stored at –20°C. The enzymatic activity of active renin in the plasma was determined by incubation with rat angiotensinogen and measurement of angiotensin I generation by RIA as described by Campbell et al. [26], except that the antibody-capture method for the renin assay was used to avoid overestimation of renin inhibition by aliskiren [27].
Statistics
Analyses were performed using either a one-way ANOVA (parametric) followed by appropriate post-hoc analyses correcting for the number of comparisons, or a Kruskal–Wallis test followed by individual Mann–Whitney U tests (non-parametric). Analyses of neovascular tufts and avascular area were performed using an unpaired Student’s t test. Investigators were masked to the treatment groups.
Results
Body weight, systolic BP and blood glucose
Diabetic Ren-2 rats had reduced weight gain compared with non-diabetic control rats, and body weight was not altered by either aliskiren or lisinopril (Table 1). Systolic BP in Ren-2 rats was unaffected by diabetes. In diabetic Ren-2 rats, both aliskiren and lisinopril reduced systolic BP, but only lisinopril normalised BP to the level of normotensive non-diabetic Sprague–Dawley control rats. In diabetic Ren-2 rats, blood glucose was elevated compared with non-diabetic control, and was unaffected by either aliskiren or lisinopril. In OIR Ren-2 rats, there was no effect of treatments on body weight (Table 1). In OIR mice, body weight was reduced compared with sham controls. In OIR mice, aliskiren partially restored the reduction in body weight (Table 1).
Aliskiren reduced retinal leucostasis in diabetic Ren-2 rats
Leucostasis was increased in the central, mid and peripheral retina of diabetic Ren-2 rats compared with non-diabetic Ren-2 controls (Fig. 1). In diabetic Ren-2 rats, both aliskiren and lisinopril reduced leucostasis in the central, mid and peripheral retina to a similar extent and to the level of non-diabetic Ren-2 rats.
Leucostasis in representative retinal flatmounts from diabetic Ren-2 rats. Adherent leucocytes were labelled with rhodamine–concanavalin A. Scale bar, 35 μm. a Non-diabetic. b Diabetic. c D + aliskiren. d D + lisinopril. Leucostasis (arrows) was increased in the central, mid and peripheral retina of diabetic Ren-2 rats compared with non-diabetic controls. In diabetic Ren-2 rats, both aliskiren and lisinopril reduced leucostasis in all regions compared with control levels. e–g Graphs showing the number of leucocytes (mean ± SEM) in the central (e), mid (f) and peripheral (g) retina. *p < 0.01 compared with all groups; † p < 0.01 compared with diabetic control; n = 7–11 rats/group. Ali, aliskiren; D, diabetic; Lis, lisinopril; ND, non-diabetic
Aliskiren reduced retinal acellular capillaries in diabetic Ren-2 rats
Acellular capillaries were increased in the mid and peripheral, but not the central, retina of diabetic Ren-2 rats compared with non-diabetic Ren-2 controls (Fig. 2). In diabetic Ren-2 rats, both aliskiren and lisinopril reduced acellular capillaries in the mid and peripheral retina to a similar extent and to the level of non-diabetic Ren-2 rats, but had no effect on the number of acellular capillaries in the central retina. In H&E sections, there was no apparent effect of treatments on retinal cellular integrity.
Acellular capillaries in representative trypsin digests of retina from diabetic Ren-2 rats. Retina stained with periodic acid–Schiff’s reagent. Scale bar, 60 μm. a Non-diabetic. b Diabetic. c D + aliskiren. d D + lisinopril. Neither diabetes nor treatments affected acellular capillary number in the central retina. Acellular capillaries (arrow) were increased in the mid and peripheral retina of diabetic Ren-2 rats compared with non-diabetic controls. In diabetic Ren-2 rats, both aliskiren and lisinopril reduced acellular capillaries in the mid and peripheral retina to control levels. e–f Graphs showing mean number of acellular capillaries per mm2 of retina (mean ± SEM) in the central (e), mid (f) and peripheral (g) retina. *p < 0.05 compared with all groups; † p < 0.05 compared with diabetic controls; n = 7–9 rats/group. Ali, aliskiren; D, diabetic; Lis, lisinopril; ND, non-diabetic
Aliskiren reduced neoangiogenesis in Ren-2 rats with OIR
In sham controls, intraretinal blood vessel profiles (BVPs) were detected in the GCL and IPL, whereas preretinal BVPs attached to the retinal surface and protruding into the vitreous were not detected. In shams, neither aliskiren nor lisinopril affected the number of intraretinal BVPs, and preretinal BVPs were not detected (Fig. 3). OIR was associated with an increase in both intraretinal and preretinal BVPs compared with controls (Fig. 3). In OIR, aliskiren (10 mg kg−1 day−1) reduced intraretinal and preretinal BVPs compared with OIR controls but not to sham levels, whereas aliskiren (30 mg kg−1 day−1) and lisinopril reduced intraretinal BVPs and preretinal BVPs to sham levels. Qualitative analysis of every tenth retinal section stained with H&E revealed similar results, and there was no apparent effect of aliskiren or lisinopril on cellular integrity.
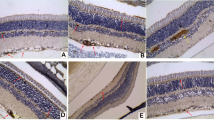
BVPs in representative 3 μm paraffin sections of retina from Ren-2 rats with OIR. Intraretinal BVPs (single arrows) were in the GCL and IPL. Preretinal BVPs (double arrows) were attached to the retinal surface and protruding into the vitreous. Sections stained with H&E. Scale bar, 25 μm. a Sham untreated control. b Sham + aliskiren 10 mg kg−1 day−1. c Sham + lisinopril. d, e OIR untreated control. f, g OIR + aliskiren 10 mg kg−1 day−1. h OIR + aliskiren 30 mg kg−1 day−1. i OIR + lisinopril. In sham controls, intraretinal but not preretinal BVPs were detected (a). In shams, neither aliskiren (b) nor lisinopril (c) altered the number of intraretinal BVPs, and preretinal BVPs were not detected. In OIR controls, both intraretinal (d) and preretinal (e) BVPs were increased compared with all sham groups. In OIR, both intraretinal (f) and preretinal (g) BVPs were slightly reduced with aliskiren 10 mg kg−1 day−1. In OIR, aliskiren 30 mg kg−1 day−1 (h) and lisinopril (i) reduced intraretinal BVPs to the level of sham control, and preretinal BVPs were not detected. j Graph showing mean number of intraretinal and preretinal BVPs per retinal field (mean ± SEM). *p < 0.001 for intraretinal and preretinal BVPs compared with all sham groups; † p < 0.05 for intraretinal and preretinal BVPs compared with all sham groups and OIR untreated control; ‡ p < 0.005 for intraretinal BVPs compared with OIR untreated control and OIR + aliskiren (10 mg); n = 6–9 rats/group. White bars, sham groups and intraretinal BVPs; black bars, OIR groups and intraretinal BVPs; grey bars, preretinal BVPs (only detected in OIR control and OIR + aliskiren 10 mg kg−1 day−1). Ali, aliskiren; Con, control; Lis, lisinopril
Aliskiren reduced neoangiogenesis in C57Bl/6 mice with OIR
Mice with OIR exhibited an avascular central retina adjacent to the optic disc and neovascular tufts in the mid and peripheral retina (Fig. 4) [23]. In mice with OIR, aliskiren (30 mg kg−1 day−1) reduced neovascular tufts by 93% and avascular retina by 35% (Fig. 4).
Wholemounts of retina from C57Bl/6 mice with OIR and immunolabelled with FITC-conjugated lectin. Scale bar, 1 mm. a Sham control. b OIR control. c OIR + aliskiren 30 mg kg−1 day−1. Graphs showing mean percentages of neovascular tufts (d) and avascular area per retina (e) (mean ± SEM). Sham controls did not have neovascular tufts or avascular areas of retina. In OIR, neovascular tufts (arrows) were detected in the mid and peripheral area of the retina, and the central retina adjacent to the optic disc (starred) was avascular. In OIR mice treated with aliskiren, neovascular tufts were reduced by 93%, and the avascular area was reduced by 35% compared with OIR control. * p < 0.005 compared with OIR control; n = 6–10 mice/group. Ali, aliskiren; Con, control
Aliskiren reduced retinal leucostasis in Ren-2 rats with OIR
Neither aliskiren nor lisinopril affected retinal leucostasis in shams (Fig. 5). OIR was associated with an increase in leucostasis in the central, mid and peripheral retina compared with sham controls (Fig. 5). In OIR, aliskiren (10 mg kg−1 day−1) did not reduce retinal leucostasis compared with untreated OIR control rats. However, aliskiren (30 mg kg−1 day−1) reduced leucostasis of OIR rats in the central, mid and peripheral retina to the level of sham controls. Lisinopril reduced leucostasis in the central, mid and peripheral retina of OIR rats, but not to the level of sham control or OIR rats treated with 30 mg kg−1 day−1 aliskiren.
Leucostasis in representative retinal flatmounts from Ren-2 rats with OIR. Adherent leucocytes labelled with rhodamine–concanavalin A. Scale bar, 25 μm. a Sham untreated control. b Sham + aliskiren 10 mg kg−1 day−1. c Sham + lisinopril. d OIR untreated control. e OIR + aliskiren 10 mg kg−1 day−1. f OIR + aliskiren 30 mg kg−1 day−1. g OIR + lisinopril. In shams, treatments did not alter leucostasis. In OIR controls, leucostasis (arrows) was increased in the central, mid and peripheral retina compared with all sham groups. In OIR, leucostasis was unaffected by aliskiren 10 mg kg−1 day−1 but reduced by aliskiren 30 mg kg−1 day−1 in all regions to sham control levels. In OIR, lisinopril reduced leucostasis in all regions but not to the level of sham control or OIR + aliskiren 30 mg kg−1 day−1. h–j Graphs showing number of leucocytes (mean ± SEM) in the central (h), mid (i) and peripheral (j) retina. *p < 0.005 compared with all sham groups; † p < 0.005 compared with OIR control and OIR + aliskiren 10 mg kg−1 day−1; ‡ p < 0.05 compared with all sham and OIR groups; n = 6–12 rats/group. White bars, sham groups; black bars, OIR groups. Ali, aliskiren; Con, control; Lis, lisinopril
Aliskiren reduced ED-1 immunolabelling in the inner retina of Ren-2 rats with OIR
Neither aliskiren nor lisinopril affected ED-1 immunolabelling in the inner retina of sham rats (Fig. 6). OIR was associated with an increase in ED-1 immunolabelling in the inner retina compared with sham controls (Fig. 6). In OIR, aliskiren (10 mg kg−1 day−1) reduced ED-1 immunolabelling compared with untreated OIR controls, but not to sham levels, whereas aliskiren (30 mg kg−1 day−1) reduced ED-1 immunolabelling to the level of sham control. Lisinopril reduced ED-1 immunolabelling of OIR rats, but not to the level of sham control or OIR rats treated with 30 mg kg−1 day−1 aliskiren (Fig. 6).
ED-1 immunolabelling of representative 3 μm paraffin sections of the inner retina from Ren-2 rats with OIR. Inner retina comprises the GCL and IPL. Sections were counterstained with haematoxylin. Scale bar, 30 μm. a Sham untreated control. b Sham + aliskiren 10 mg kg−1 day−1. c Sham + lisinopril. d OIR untreated control. e OIR + aliskiren 10 mg kg−1 day−1. f OIR + aliskiren 30 mg kg−1 day−1. g OIR + lisinopril. In shams, treatments did not alter ED-1 immunolabelling (arrows). In OIR controls, ED-1 immunolabelling (arrows) was increased compared with all sham groups. In OIR, ED-1 immunolabelling was reduced with aliskiren 10 mg kg−1 day−1 and normalised to sham levels with aliskiren 30 mg kg−1 day−1. In OIR, lisinopril reduced ED-1 immunolabelling but not to the level of sham control or OIR + aliskiren 30 mg kg−1 day−1. h Graph showing ED-1 immunolabelling per proportional field of inner retina (mean ± SEM). *p < 0.01 compared with all sham groups; † p < 0.05 compared with sham groups, OIR control and OIR + aliskiren 30 mg kg−1 day−1; ‡ p < 0.01 compared with OIR control; n = 6–12 rats/group. White bars, sham groups; black bars, OIR groups. Ali, aliskiren; Con, control; Lis, lisinopril
Aliskiren reduced retinal Vegf and Icam-1 mRNA and protein levels in Ren-2 rats with OIR
Neither aliskiren nor lisinopril affected Vegf or Icam-1 mRNA levels in the retinas of sham rats (Fig. 7). OIR was associated with an increase in retinal Vegf and Icam-1 mRNA levels compared with sham groups (Fig. 7). In OIR, 10 mg kg−1 day−1 aliskiren did not alter retinal Vegf and Icam-1 mRNA levels, whereas 30 mg kg−1 day−1 aliskiren reduced retinal Vegf and Icam-1 mRNA to the levels of sham control (Fig. 7). In OIR, lisinopril reduced retinal Vegf mRNA to control levels, but had no effect on Icam-1 mRNA levels. OIR was associated with increased retinal VEGF and ICAM-1 protein levels compared with sham control (Fig. 7). Aliskiren (30 mg kg−1 day−1) reduced retinal VEGF and ICAM-1 protein levels in OIR rats to the level of sham control rats (Fig. 7).
Vegf and Icam-1 mRNA and VEGF and ICAM-1 protein levels in retina from Ren-2 rats with OIR. In sham control rats, neither aliskiren nor lisinopril altered Vegf mRNA levels. a Vegf mRNA: in OIR, Vegf mRNA levels were increased compared with sham control. In OIR controls, Vegf mRNA levels were unaffected by aliskiren at 10 mg kg−1 day−1, but reduced to the level of sham control with 30 mg kg−1 day−1. In OIR, lisinopril reduced Vegf mRNA levels to sham control levels. Values are mean ± SEM. *p < 0.001 compared with all sham groups; † p < 0.05 compared with OIR control; n = 6–8 rats/group. b Icam-1 mRNA: in sham rats, neither aliskiren nor lisinopril altered Icam-1 mRNA levels. In OIR, Icam-1 mRNA levels were increased compared with sham control. In OIR, Icam-1 mRNA levels were unaffected by aliskiren at 10 mg kg−1 day−1, but reduced to the level of sham control with 30 mg kg−1 day−1. In OIR, lisinopril had no effect on Icam-1 mRNA levels. Values are mean ± SEM. *p < 0.05 compared with sham control; † p < 0.05 compared with OIR control; n = 6–8 rats/group. c, d In OIR, VEGF (c) and ICAM-1 (d) protein levels were increased compared with sham control. In OIR, aliskiren 30 mg kg−1 day−1 reduced VEGF and ICAM-1 to the level of sham control. Values are mean ± SEM. *p < 0.05 compared with sham control; † p < 0.05 compared with OIR control; n = 6–8 rats/group. White bars, sham groups; black bars, OIR groups. Ali, aliskiren; Con, control; Lis, lisinopril
Plasma active renin levels in diabetic Ren-2 rats and Ren-2 rats with OIR
Plasma active renin levels of diabetic Ren-2 rats were 45% lower than in non-diabetic Ren-2 rats (Fig. 8). Aliskiren (10 mg kg−1 day−1) increased plasma active renin levels 2.3-fold, whereas lisinopril (10 mg kg−1 day−1) increased plasma active renin levels by 55-fold in diabetic Ren-2 rats. In sham Ren-2 rats, aliskiren (10 mg kg−1 day−1) increased plasma active renin levels by 3.2-fold and lisinopril (10 mg kg−1 day−1) increased plasma active renin levels by 32-fold. By contrast, Ren-2 rats with OIR had plasma active renin levels 28-fold higher than in sham Ren-2 rats, and 10 and 30 mg kg−1 day−1 aliskiren produced similar 63–64% reductions in plasma active renin levels, whereas lisinopril (10 mg kg−1 day−1) did not alter plasma active renin levels in Ren-2 rats with OIR (Fig. 8).
Active renin levels in plasma from Ren-2 rats with diabetes or OIR. Values are expressed as picomoles angiotensin I generated per millilitre per hour. a Diabetic study: in diabetic rats, plasma active renin levels were decreased by 45% compared with non-diabetic control. In diabetic rats, plasma active renin levels were increased 2.3-fold with aliskiren 10 mg kg−1 day−1 and increased 55-fold with lisinopril compared with diabetic controls. Values are mean ± SEM. *p < 0.05 compared with non-diabetic control; † p < 0.005 compared with diabetic control; n = 6 rats/group. b OIR study: in sham rats, plasma active renin levels were increased 3.2-fold with aliskiren 10 mg kg−1 day−1 and 32-fold with lisinopril compared with sham control. In OIR, plasma active renin levels were increased 28-fold compared with sham control. In OIR rats aliskiren at either 10 mg kg−1 day−1 mg or 30 mg kg−1 day−1 reduced plasma active renin levels compared with OIR control, although not to the level of sham control. In OIR, lisinopril had no effect on plasma active renin levels. Values are mean ± SEM. * p < 0.0005 compared with sham control; † p < 0.0005 compared with sham groups; ‡ p < 0.05 compared with OIR control and sham control; n = 8 rats/group. Ali, aliskiren; Ang I, angiotensin I; Con, control; D, diabetic; Lis, lisinopril; ND, non-diabetic
Discussion
To our knowledge this is the first report that aliskiren improves vasculopathy in models of retinal disease. In Ren-2 rats with diabetes, aliskiren normalised acellular capillary formation and leucostasis, and in Ren-2 rats with OIR, aliskiren reduced neoangiogenesis, inflammation and the upregulation of retinal Vegf and Icam-1 expression. In general, these effects of aliskiren were similar to those of ACE inhibition and to our previous reports of the effects of ARB [7–9, 11], demonstrating the benefits of renin inhibition in models of retinopathy.
Inflammation may contribute to the development of microvascular pathology in diabetic retinopathy and OIR [28, 29]. In the present study, aliskiren attenuated the intense pro-inflammatory profile in retina of Ren-2 rats with diabetes or OIR, with a reduction in leucostasis, ICAM-1 production and ED-1 immunolabelling to control levels. There is evidence that aliskiren reduced the macrophage content of atherosclerotic plaques [30], leucocyte adhesion in femoral arteries [31] and the number of CD11b+ and CD11c+ antigen presenting cells in a model of multiple sclerosis [32]. The results of the present study are consistent with these findings, and highlight the potent anti-inflammatory properties of aliskiren. Neoangiogenesis is a feature of both diabetic retinopathy and OIR, resulting in vascular leakage, haemorrhage and compromised vision. In diabetic retinopathy, neoangiogenesis may be preceded by the presence of acellular capillaries, in which pericytes and endothelial cells are absent. Our finding that acellular capillaries develop in the mid and peripheral retina of diabetic Ren-2 rats is consistent with our previous studies [24], and with observations in diabetic patients [33]. As rodents do not develop retinal neoangiogenesis, even after years of diabetes [34], we also studied OIR [22]. Our finding that in Ren-2 rats with OIR the highest dose of aliskiren (30 mg) reduced neoangiogenesis and retinal VEGF and ICAM-1 to control levels indicates that aliskiren attenuates retinal vasculopathy. These effects of aliskiren are consistent with our previous reports for ACE inhibition and ARB in rats with OIR [7, 9].
Our findings in diabetic Ren-2 rats with retinopathy are consistent with studies in diabetic nephropathy where aliskiren given at a dose of 10 mg kg−1 day−1 normalised renal pathology and had similar effects to ACE inhibition [19]. However, in Ren-2 rats with OIR, although 10 mg kg−1 day−1 aliskiren attenuated retinal neoangiogenesis and ED-1 immunolabelling, it did not reduce this pathology to control levels, or lower retinal Vegf and Icam-1 mRNA levels. In contrast, the higher dose of 30 mg−1 kg−1 day aliskiren substantially reduced all aspects of retinal vasculopathy including retinal Vegf and Icam-1 mRNA levels. One reason why a higher dose of aliskiren was required may be the more extensive and rapid nature of the vasculopathy that occurs in OIR compared with diabetic retinopathy. Interestingly, in Ren-2 rats with OIR, although lisinopril normalised retinal neoangiogenesis and Vegf mRNA, it did not reduce retinal leucostasis, ED-1 immunolabelling or Icam-1 mRNA to control levels. These findings suggest that higher doses of lisinopril may be required to completely attenuate the intense retinal inflammation that occurs in Ren-2 rats with OIR.
A possible limitation of our study was the use of Ren-2 rats as these animals overproduce renin and angiotensin II in extrarenal tissues [20], a situation that may not necessarily occur in individuals with diabetic retinopathy. On the other hand, diabetic Ren-2 rats have high levels of plasma prorenin and are hypertensive, features that occur in diabetic individuals [5, 6, 35], such as the high-risk patients in which aliskiren is currently being evaluated [36]. Both hypertension and increased angiotensin II levels may contribute to the retinal lesions that develop in the Ren-2 rats. However, in our study of diabetic Ren-2 rats, although aliskiren did not completely reduce systolic BP to control levels, it still normalised retinal acellular capillaries and leucostasis. These results suggest that the beneficial effects of aliskiren in the retina may be partially independent of BP reduction and are consistent with reports that aliskiren conferred renal [19] and cardiac [37] protection without normalising BP. These findings are consistent with our previous studies in diabetic Ren-2 rats where we compared ARB with the β-blocker, atenolol. Both treatments normalised BP in diabetic Ren-2 rats; however, only ARB reduced retinal vasculopathy and improved the electroretinogram [24, 38]. Similar comparisons in OIR may not necessarily be informative given that β-blockers have anti-proliferative effects in OIR [39], and whether this occurs by a BP-dependent mechanism is uncertain as BP cannot be measured in P18 rodents. We therefore examined this question in normotensive mice with OIR, and our finding that aliskiren reduced neoangiogenesis and avascular retina in this model indicates that the beneficial effects of aliskiren on vasculopathy were not necessarily related to its anti-hypertensive properties.
Diabetic retinopathy and OIR involve damage not only to the microvasculature, but also to neurons and glia, resulting in compromised retinal function [40, 41]. ACE inhibition and ARB improve aspects of neuronal and glial damage in both situations [42], such as ameliorating losses in the electroretinogram [25, 38, 43] and neuronal and glial cell degeneration [44–46]. Our identification of renin in retinal Müller cells [2] and ganglion cells [3] is suggestive that aliskiren may influence these cell populations. A limitation of our study and a future direction is the assessment of the potential of aliskiren to ameliorate neuronal and glial pathology in diabetic retinopathy and OIR.
The lower plasma active renin level in diabetic Ren-2 rats, in comparison with non-diabetic Ren-2 rats, was in agreement with our report of lower plasma angiotensin II and angiotensin I levels in diabetic Ren-2 rats, although we did not find a reduction in plasma active renin levels in our previous study [47]. The Ren-2 rat has elevated levels of mouse renin and angiotensin peptides in plasma and tissues [48] that suppress renal expression of rat renin. Inhibition of angiotensin II formation, such as by ACE inhibition, reverses the suppression of renal expression of rat renin [49], thereby accounting for the increase in plasma active renin levels in sham and diabetic Ren-2 rats administered lisinopril. Moreover, aliskiren similarly increases renal expression of rat renin [18]. However, aliskiren is a much more potent inhibitor of mouse renin (IC50 4.5 nmol/l) than rat renin (IC50 90 nmol/l) [18], and increased rat renin expression together with its lesser susceptibility to inhibition by aliskiren may account for the increase in plasma active renin levels in Ren-2 rats administered aliskiren.
Ren-2 rats with OIR had marked elevation of plasma renin levels because of increased expression of the mouse renin transgene and the partial suppression of plasma active renin levels by aliskiren in OIR rats represented the net effect of inhibition of mouse renin activity and increased renal expression of rat renin. Moreover, lisinopril may have failed to reduce angiotensin II formation sufficiently to cause a measurable increase in renal expression of rat renin in OIR rats. For both the diabetic Ren-2 rats and the Ren-2 rats with OIR, it is likely that aliskiren produced much greater inhibition of renin activity in the retina than in plasma, given that the markedly elevated renin levels in the eye of Ren-2 rats represents local production of mouse renin [7]. Further studies are required to determine whether the effects of aliskiren in the retina are due to modulation of local renin.
In conclusion, aliskiren has benefits in models of atherosclerosis [30], diabetic nephropathy [18, 19] and cardiac disease [37, 50]. We extended these benefits to a protective role for aliskiren in experimental models of retinal vasculopathy. These findings indicate that aliskiren may have the potential to be a treatment for diabetic retinopathy.
Abbreviations
- ARB:
-
Angiotensin receptor blockade
- BP:
-
Blood pressure
- BVPs:
-
Blood vessel profiles
- GCL:
-
Ganglion cell layer
- ICAM-1:
-
Intercellular adhesion molecule-1
- IPL:
-
Inner plexiform layer
- OIR:
-
Oxygen-induced retinopathy
- PD:
-
Postnatal day
- RAS:
-
Renin–angiotensin system
- Ren-2:
-
(mRen-2)27
- VEGF:
-
Vascular endothelial growth factor
References
Simo R, Hernandez C (2009) Advances in the medical treatment of diabetic retinopathy. Diabetes Care 32:1556–1562
Berka JL, Stubbs AJ, Wang DZ et al (1995) Renin-containing Muller cells of the retina display endocrine features. Invest Ophthalmol Vis Sci 36:1450–1458
Sarlos S, Wilkinson-Berka JL (2005) The renin–angiotensin system and the developing retinal vasculature. Invest Ophthalmol Vis Sci 46:1069–1077
Wagner J, Jan Danser AH, Derkx FH et al (1996) Demonstration of renin mRNA, angiotensinogen mRNA, and angiotensin converting enzyme mRNA expression in the human eye: evidence for an intraocular renin–angiotensin system. Br J Ophthalmol 80:159–163
Luetscher JA, Kraemer FB, Wilson DM, Schwartz HC, Bryer-Ash M (1985) Increased plasma inactive renin in diabetes mellitus. A marker of microvascular complications. N Engl J Med 312:1412–1417
Danser AH, van den Dorpel MA, Deinum J et al (1989) Renin, prorenin, and immunoreactive renin in vitreous fluid from eyes with and without diabetic retinopathy. J Clin Endocrinol Metab 68:160–167
Moravski CJ, Kelly DJ, Cooper ME et al (2000) Retinal neovascularization is prevented by blockade of the renin–angiotensin system. Hypertension 36:1099–1104
Moravski CJ, Skinner SL, Stubbs AJ et al (2003) The renin–angiotensin system influences ocular endothelial cell proliferation in diabetes: transgenic and interventional studies. Am J Pathol 162:151–160
Wilkinson-Berka JL, Tan G, Jaworski K, Miller AG (2009) Identification of a retinal aldosterone system and the protective effects of mineralocorticoid receptor antagonism on retinal vascular pathology. Circ Res 104:124–133
Zhang JZ, Xi X, Gao L, Kern TS (2007) Captopril inhibits capillary degeneration in the early stages of diabetic retinopathy. Curr Eye Res 32:883–889
Miller AG, Tan G, Binger KJ et al (2010) Candesartan attenuates diabetic retinal vascular pathology by restoring glyoxalase-I function. Diabetes 59:3208–3215
Chaturvedi N, Porta M, Klein R et al (2008) Effect of candesartan on prevention (DIRECT-Prevent 1) and progression (DIRECT-Protect 1) of retinopathy in type 1 diabetes: randomised, placebo-controlled trials. Lancet 372:1394–1402
Sjolie AK, Klein R, Porta M et al (2008) Effect of candesartan on progression and regression of retinopathy in type 2 diabetes (DIRECT-Protect 2): a randomised placebo-controlled trial. Lancet 372:1385–1393
Mauer M, Zinman B, Gardiner R et al (2009) Renal and retinal effects of enalapril and losartan in type 1 diabetes. N Engl J Med 361:40–51
United Kingdom Prospective Diabetes Study Group (1998) Efficacy of atenolol and captopril in reducing risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 39. UK Prospective Diabetes Study Group. BMJ 317:713–720
Estacio RO, Jeffers BW, Gifford N, Schrier RW (2000) Effect of blood pressure control on diabetic microvascular complications in patients with hypertension and type 2 diabetes. Diabetes Care 23(Suppl 2):B54–B64
Patel A, MacMahon S, Chalmers J et al (2007) Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial. Lancet 370:829–840
Feldman DL, Jin L, Xuan H et al (2008) Effects of aliskiren on blood pressure, albuminuria, and (pro)renin receptor expression in diabetic TG(mRen-2)27 rats. Hypertension 52:130–136
Kelly DJ, Zhang Y, Moe G, Naik G, Gilbert RE (2007) Aliskiren, a novel renin inhibitor, is renoprotective in a model of advanced diabetic nephropathy in rats. Diabetologia 50:2398–2404
Mullins JJ, Peters J, Ganten D (1990) Fulminant hypertension in transgenic rats harbouring the mouse Ren-2 gene. Nature 344:541–544
Kelly DJ, Skinner SL, Gilbert RE, Cox AJ, Cooper ME, Wilkinson-Berka JL (2000) Effects of endothelin or angiotensin II receptor blockade on diabetes in the transgenic (mRen-2)27 rat. Kidney Int 57:1882–1894
Penn JS, Tolman BL, Henry MM (1994) Oxygen-induced retinopathy in the rat: relationship of retinal nonperfusion to subsequent neovascularization. Invest Ophthalmol Vis Sci 35:3429–3435
Smith LE, Wesolowski E, McLellan A et al (1994) Oxygen-induced retinopathy in the mouse. Invest Ophthalmol Vis Sci 35:101–111
Wilkinson-Berka JL, Tan G, Jaworski K, Ninkovic S (2007) Valsartan but not atenolol improves vascular pathology in diabetic Ren-2 rat retina. Am J Hypertens 20:423–430
Wilkinson-Berka JL, Heine R, Tan G et al (2010) RILLKKMPSV influences the vasculature, neurons and glia, and (pro)renin receptor expression in the retina. Hypertension 55:1454–1460
Campbell DJ, Lawrence AC, Towrie A, Kladis A, Valentijn AJ (1991) Differential regulation of angiotensin peptide levels in plasma and kidney of the rat. Hypertension 18:763–773
Campbell DJ (2008) Interpretation of plasma renin concentration in patients receiving aliskiren therapy. Hypertension 51:15–18
Kern TS, Miller CM, Du Y et al (2007) Topical administration of nepafenac inhibits diabetes-induced retinal microvascular disease and underlying abnormalities of retinal metabolism and physiology. Diabetes 56:373–379
Ishida S, Yamashiro K, Usui T et al (2003) Leukocytes mediate retinal vascular remodeling during development and vaso-obliteration in disease. Nat Med 9:781–788
Nussberger J, Aubert JF, Bouzourene K, Pellegrin M, Hayoz D, Mazzolai L (2008) Renin inhibition by aliskiren prevents atherosclerosis progression: comparison with irbesartan, atenolol, and amlodipine. Hypertension 51:1306–1311
Ino J, Kojima C, Osaka M, Nitta K, Yoshida M (2009) Dynamic observation of mechanically-injured mouse femoral artery reveals an antiinflammatory effect of renin inhibitor. Arterioscler Thromb Vasc Biol 29:1858–1863
Stegbauer J, Lee DH, Seubert S et al (2009) Role of the renin–angiotensin system in autoimmune inflammation of the central nervous system. Proc Natl Acad Sci USA 106:14942–14947
Cardillo Piccolino F, Zingirian M, Mosci C (1987) Classification of proliferative diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 225:245–250
Engerman RL, Kern TS (1995) Retinopathy in animal models of diabetes. Diabetes Metab Rev 11:109–120
Matthews DR, Stratton IM, Aldington SJ, Holman RR, Kohner EM (2004) Risks of progression of retinopathy and vision loss related to tight blood pressure control in type 2 diabetes mellitus: UKPDS 69. Arch Ophthalmol 122:1631–1640
Parving HH, Persson F, Lewis JB, Lewis EJ, Hollenberg NK (2008) Aliskiren combined with losartan in type 2 diabetes and nephropathy. N Engl J Med 358:2433–2446
Westermann D, Riad A, Lettau O et al (2008) Renin inhibition improves cardiac function and remodeling after myocardial infarction independent of blood pressure. Hypertension 52:1068–1075
Phipps JA, Wilkinson-Berka JL, Fletcher EL (2007) Retinal dysfunction in diabetic ren-2 rats is ameliorated by treatment with valsartan but not atenolol. Invest Ophthalmol Vis Sci 48:927–934
Ristori C, Filippi L, Dal Monte M et al (2011) Role of the adrenergic system in a mouse model of oxygen-induced retinopathy: antiangiogenic effects of beta-adrenoreceptor blockade. Invest Ophthalmol Vis Sci 52:155–170
Lieth E, Barber AJ, Xu B et al (1998) Glial reactivity and impaired glutamate metabolism in short-term experimental diabetic retinopathy. Penn State Retina Research Group. Diabetes 47:815–820
Liu K, Akula JD, Hansen RM, Moskowitz A, Kleinman MS, Fulton AB (2006) Development of the electroretinographic oscillatory potentials in normal and ROP rats. Invest Ophthalmol Vis Sci 47:5447–5452
Fletcher EL, Phipps JA, Ward MM, Vessey KA, Wilkinson-Berka JL (2010) The renin–angiotensin system in retinal health and disease: its influence on neurons, glia and the vasculature. Prog Retin Eye Res 29:284–311
Kurihara T, Ozawa Y, Nagai N et al (2008) Angiotensin II type 1 receptor signaling contributes to synaptophysin degradation and neuronal dysfunction in the diabetic retina. Diabetes 57:2191–2198
Silva KC, Rosales MA, Biswas SK, Lopes de Faria JB, Lopes de Faria JM (2009) Diabetic retinal neurodegeneration is associated with mitochondrial oxidative stress and is improved by an angiotensin receptor blocker in a model combining hypertension and diabetes. Diabetes 58:1382–1390
Downie LE, Pianta MJ, Vingrys AJ, Wilkinson-Berka JL, Fletcher EL (2008) AT1 receptor inhibition prevents astrocyte degeneration and restores vascular growth in oxygen-induced retinopathy. Glia 56:1076–1090
Downie LE, Hatzopoulos KM, Pianta MJ et al (2010) Angiotensin type-1 receptor inhibition is neuroprotective to amacrine cells in a rat model of retinopathy of prematurity. J Comp Neurol 518:41–63
Campbell DJ, Kelly DJ, Wilkinson-Berka JL, Cooper ME, Skinner SL (1999) Increased bradykinin and “normal” angiotensin peptide levels in diabetic Sprague–Dawley and transgenic (mRen-2)27 rats. Kidney Int 56:211–221
Campbell DJ, Rong P, Kladis A, Rees B, Ganten D, Skinner SL (1995) Angiotensin and bradykinin peptides in the TGR(mRen-2)27 rat. Hypertension 25:1014–1020
Tokita Y, Franco-Saenz R, Mulrow PJ (1995) Reversal of the suppressed kidney renin level in the hypertensive transgenic rat TGR(mRen-2)27 by angiotensin converting enzyme inhibition. Am J Hypertens 8:1031–1039
Pilz B, Shagdarsuren E, Wellner M et al (2005) Aliskiren, a human renin inhibitor, ameliorates cardiac and renal damage in double-transgenic rats. Hypertension 46:569–576
Acknowledgements
This study was supported by National Health and Medical Research Council of Australia (NHMRC) Grant #491058. JW-B and DJC are NHMRC Senior Research Fellows. AGM is a Juvenile Diabetes Research Foundation Post-Doctoral Scholar. The authors thank David R. Berka for technical assistance.
Contribution statement
J.W.-B., D.J.C. and A.G.M. contributed to the conception and design, analysis and interpretation of data described in the study. G.T., K.J.B., L.S., K.McM., D.D. and G.P. contributed to the analysis and interpretation of data. All authors contributed to the drafting or revising of the manuscript and supported the final approval of the published version of the manuscript.
Duality of interest
The authors declare that, in part, the Novartis Corporation, USA, supported this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wilkinson-Berka, J.L., Tan, G., Binger, K.J. et al. Aliskiren reduces vascular pathology in diabetic retinopathy and oxygen-induced retinopathy in the transgenic (mRen-2)27 rat. Diabetologia 54, 2724–2735 (2011). https://doi.org/10.1007/s00125-011-2239-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-011-2239-9