Abstract
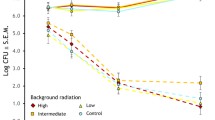
Microorganisms have shaped the evolution of a variety of defense mechanisms against pathogenic infections. Radioactivity modifies bacterial communities and, therefore, bird hosts breeding in contaminated areas are expected to adapt to the new bacterial environment. We tested this hypothesis in populations of barn swallows (Hirundo rustica) from a gradient of background radiation levels at Chernobyl and uncontaminated controls from Denmark. Investment in defenses against keratinolytic bacteria was measured from feather structure (i.e., susceptibility to degradation) and uropygial secretions. We studied degradability of tail feathers from areas varying in contamination in laboratory experiments using incubation of feathers with a feather-degrading bacterium, Bacillus licheniformis, followed by measurement of the amount of keratin digested. The size of uropygial glands and secretion amounts were quantified, followed by antimicrobial tests against B. licheniformis and quantification of wear of feathers. Feathers of males, but not of females, from highly contaminated areas degraded at a lower rate than those from medium and low contamination areas. However, feathers of both sexes from the Danish populations showed little evidence of degradation. Individual barn swallows from the more contaminated areas of Ukraine produced the largest uropygial secretions with higher antimicrobial activity, although wear of feathers did not differ among males from different populations. In Denmark, swallows produced smaller quantities of uropygial secretion with lower antimicrobial activity, which was similar to swallow populations from uncontaminated areas in Ukraine. Therefore, barn swallows breeding in contaminated areas invested more in all defenses against keratinolytic bacteria than in uncontaminated areas of Ukraine and Denmark, although they had similar levels of feather wear. Strong natural selection exerted by radioactivity may have selected for individuals with higher defense capacity against bacterial infections during the 30 years since the Chernobyl disaster.

Similar content being viewed by others
References
Balbontín J, Møller AP (2015) Environmental conditions during early life accelerate the rate of senescence in a short-lived passerine bird. Ecology 96:948–959
Barbosa A, Merino S, Cuervo JJ, De Lope F, Møller AP (2003) Feather damage of long tails in barn swallows Hirundo rustica. Ardea 91:85–90
Bisson IA, Marra PP, Burtt EH, Sikaroodi M, Gillevet PM (2007) A molecular comparison of plumage and soil bacteria across biogeographic, ecological, and taxonomic scales. Microb Ecol 54:65–81
Bonisoli-Alquati A, Voris A, Mousseau TA, Møller AP, Saino N, Wyatt MD (2010) DNA damage in barn swallows (Hirundo rustica) from the Chernobyl region detected by use of the comet assay. Comp Biochem Phys C 151:271–277
Burtt EH (1986) An analysis of physical, physiological, and optical aspects of avian coloration with emphasis on wood-warblers. Ornithol Monographs 38
Burtt EH, Ichida JM (1999) Occurrence of feather-degrading bacilli in the plumage of birds. Auk 116:364–372
Burtt EH, Schroeder MR, Smith LA, Sroka JE, Mcgraw KJ (2011) Colourful parrot feathers resist bacterial degradation. Biol Lett 7:214–216
Cuervo JJ, De Ayala RM (2014) Effects of experimental tail shortening on the phenotypic condition of barn swallows Hirundo rustica: implications for tail-length evolution. J Avian Biol 45:345–353
Czirjak GA, Møller AP, Mousseau TA, Heeb P (2010) Microorganisms associated with feathers of barn swallows in radioactively contaminated areas around Chernobyl. Microb Ecol 60:373–380
Giraudau M, Czirjak GA, Duval C, Bretagnolle V, Gutierrez C, Guillon N, Heeb P (2013) Effect of preen oil on plumage bacteria: an experimental test with the mallard. Behav Process 92:1–5
Goldstein G, Flory KR, Browne BA, Majid S, Ichida JM, Burtt EH (2004) Bacterial degradation of black and white feathers. Auk 121:656–659
Gunderson AR, Frame AM, Swaddle JP, Forsyth MH (2008) Resistance of melanized feathers to bacterial degradation: is it really so black and white? J Avian Biol 39:539–545
Hasegawa M, Arai E, Watanabe M, Nakamura M (2010) Mating advantage of multiple male ornaments in the barn swallow Hirundo rustica gutturalis. Ornithol Sci 9:141–148
Jones HE, West HM, Chamberlain PM, Parekh NR, Beresford NA, Crout NMJ (2004) Effects of gamma irradiation on Holcus lanatus (Yorkshire fog grass) and associated soil microorganisms. J Environ Radioactiv 74:57–71
Lucas FS, Broennimann O, Febbraro I, Heeb P (2003) High diversity among feather-degrading bacteria from a dry meadow soil. Microb Ecol 45:282–290
Martín-Platero AM, Valdivia E, Ruiz-Rodríguez M, Soler JJ, Martín-Vivaldi M, Maqueda M, Martínez-Bueno M (2006) Characterization of antimicrobial substances produced by Enterococcus faecalis MRR 10–3, isolated from the uropygial gland of the hoopoe (Upupa epops). Appl Environ Microbiol 72:4245–4249
Martín-Vivaldi M, Peña A, Peralta-Sánchez JM, Sánchez L, Ananou S, Ruiz-Rodríguez M, Soler JJ (2010) Antimicrobial chemicals in hoopoe preen secretions are produced by symbiotic bacteria. Proc R Soc B-Biol Sci 277:123–130
Martín-Vivaldi M, Ruiz-Rodríguez M, Soler JJ, Peralta-Sánchez JM, Méndez M, Valdivia E, Martín-Platero AM, Martínez-Bueno M (2009) Seasonal, sexual and developmental differences in hoopoe Upupa epops preen gland morphology and secretions: evidence for a role of bacteria. J Avian Biol 40:191–205
McFall-Ngai M, Hadfield MG, Bosch TCG, Carey HV, Domazet-Loso T, Douglas AE, Dubilier N, Eberl G, Fukami T, Gilbert SF, Hentschel U, King N, Kjelleberg S, Knoll AH, Kremer N, Mazmanian SK, Metcalf JL, Nealson K, Pierce NE, Rawls JE, Reid A, Ruby EG, Rumpho M, Sanders JG, Tautz D, Wernegreen JJ (2013) Animals in a bacterial world, a new imperative for the life sciences. PNAS 110:3229–3236
McNamara NP, Griffiths RI, Tabouret A, Beresford NA, Bailey MJ, Whiteley AS (2007) Appl Soil Ecol 37:1–9
Mironenko NV, Alekhina IA, Zhdanova NN, Bulat SA (2000) Intraspecific variation in gamma-radiation resistance and genomic structure in the filamentous fungus Alternaria alternate: a case study of strains inhabiting Chernobyl reactor nr 4. Ecotoxicol Environ Saf 45:177–187
Møller AP (1993) Morphology and sexual selection in the barn swallow Hirundo rustica in Chernobyl, Ukraine. Proc R Soc B-Biol Sci 252:51–57
Møller AP (1994) Sexual selection and the barn swallow. Oxford University Press, Oxford
Møller AP, Barbosa A, Cuervo JJ, de Lope F, Merino S, Saino N (1998) Sexual selection and tail streamers in the barn swallow. Proc R Soc B-Biol Sci 265:409–414
Møller AP, Barnier F, Mousseau TA (2012a) Ecosystems effects 25 years after Chernobyl: pollinators, fruit set and recruitment. Oecologia 170:1155–1165
Møller AP, Bonisoli-Alquati A, Rudolfsen G, Mousseau TA (2012b) Elevated mortality among birds in Chernobyl as judged from skewed age and sex ratios. PLoS ONE 7, e35223
Møller AP, Bonisoli-Alquati A, Mousseau TA (2013) High frequency of albinism and tumours in free-living birds around Chernobyl. Mutat Res-Gen Toxicol Environ 757:52–59
Møller AP, Czirjak GA, Heeb P (2009) Feather micro-organisms and uropygial antimicrobial defences in a colonial passerine bird. Funct Ecol 23:1097–1102
Møller AP, Hobson KA, Mousseau TA, Peklo AM (2006) Chernobyl as a population sink for barn swallows: tracking dispersal using stable isotope profiles. Ecol Appl 16:1696–1705
Møller AP, Mousseau TA (2006) Biological consequences of Chernobyl: 20 years on. Trends Ecol Evol 21:200–207
Møller AP, Mousseau TA (2007) Species richness and abundance of forest birds in relation to radiation at Chernobyl. Biol Lett 3:483–486
Møller AP, Mousseau TA (2013) Assessing effects of radiation on abundance of mammals and predator–prey interactions in Chernobyl using tracks in the snow. Ecol Indic 26:112–116
Møller AP, Mousseau TA (2016) Are organisms adapting to ionizing radiation at Chernobyl? Trends Ecol Evol 31:281–289
Møller AP, Mousseau TA, de Lope F, Saino N (2007) Elevated frequency of abnormalities in barn swallows from Chernobyl. Biol Lett 3:414–417
Møller AP, Mousseau TA, Milinevsky G, Peklo A, Pysanets E, Szép T (2005) Condition, reproduction and survival of barn swallows from Chernobyl. J Anim Ecol 74:1102–1111
Ragon M, Restoux G, Moreira D, Møller AP, Lopez-Garcia P (2011) Sunlight exposed biofilm microbial communities are naturally resistant to Chernobyl ionizing-radiation levels. PLoS ONE 6, e21764
Reneerkens J, Versteegh MA, Schneider AM, Piersma T, Burtt EH (2008) Seasonally changing preen-wax composition: Red Knots’ (Calidris canutus) flexible defense against feather-degrading bacteria? Auk 125:285–290
Romanovskaya VA, Sokolov IG, Rokitko PV, Chernaya NA (1998) Effect of radioactive contamination on soil bacteria in the 10-km zone around the Chernobyl nuclear power plant. Microbiol 67:226–231
Romanovskaya VA, Rokitko PV, Mikheev AN, Gushcha NI, Malashenko YR, Chernaya NA (2002) The effect of γ-radiation and desiccation on the viability of the soil bacteria isolated from the alienated zone around the Chernobyl Nuclear Power Plant. Microbiol 71:608–613
Ruiz de Castañeda R, Burtt EH, González-Braojos S, Moreno J (2012) Bacterial degradability of an intrafeather unmelanized ornament: a role for feather-degrading bacteria in sexual selection? Biol J Linn Soc 105:409–419
Ruiz-de-Castañeda R, Burtt EH, González-Braojos S, Moreno J (2015) Bacterial degradability of white patches on primary feathers is associated with breeding date and parental effort in a migratory bird. Ibis 157:871–876
Ruiz-González MX, Czirják GA, Genevaux P, Møller AP, Mousseau TA, Heeb P (2016) Resistance of feather-associated bacteria to intermediate levels of ionizing radiation near Chernobyl. Sci Rep 6:22969
Ruiz-Rodríguez M, Valdivia E, Soler JJ, Martín-Vivaldi M, Martin-Platero AM, Martinez-Bueno M (2009) Symbiotic bacteria living in the hoopoe’s uropygial gland prevent feather degradation. J Exp Biol 212:3621–3626
Ruiz-Rodríguez M, Martínez-Bueno M, Martín-Vivaldi M, Valdivia E, Soler JJ (2013) Bacteriocins with a broader antimicrobial spectrum prevail in enterococcal symbionts isolated from the hoopoe’s uropygial gland. FEMS Microbiol Ecol 85:495–502
Ruiz-Rodríguez M, Tomás G, Martin-Gálvez D, Ruiz-Castellano C, Soler JJ (2015) Bacteria and the evolution of honest signals. The case of ornamental throat feathers in spotless starlings. Funct Ecol 29:701–709
Sangali S, Brandelli A (2000) Feather keratin hydrolysis by a Vibrio sp strain kr2. J Appl Microbiol 89:735–743
Shawkey MD, Pillai SR, Hill GE (2003) Chemical warfare? Effects of uropygial oil on feather-degrading bacteria. J Avian Biol 34:345–349
Shawkey MD, Mills KL, Dale C, Hill GE (2005) Microbial diversity of wild bird feathers revealed through culture-based and culture-independent techniques. Microb Ecol 50:40–47
Shawkey MD, Pillai SR, Hill GE (2009) Do feather-degrading bacteria affect sexually selected plumage color? Naturwissenschaften 96:123–128
Watanabe K, Hayano K (1995) Seasonal variation of soil protease activities and their relation to proteolytic bacteria and Bacillus spp. in paddy field soil. Soil Biol Biochem 27:197–203
Zhdanova NN, Zakharchenko VA, Vember VV, Nakonechnaya LT (2000) Fungi from Chernobyl: mycobiota of the inner regions of the containment structures of the damaged nuclear reactor. Mycol Res 104:1421–1426
Acknowledgments
M. Ruiz-Rodríguez had a Postdoc from the program “Andalucía Talent Hub” (Agencia Andaluza del Conocimiento, Junta de Andalucía). Funding was provided in part from the Samuel Freeman Charitable Trust, the US Fulbright Program, the CNRS (France), the American Council of Learned Societies, the University of South Carolina College of Arts and Sciences, and the Spanish Ministerio de Economía y Competitividad (European funds (FEDER)) (CGL2013-48193-C3-1-P). We also thank two anonymous referees for comments that have improved the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Alexandre Roulin
Rights and permissions
About this article
Cite this article
Ruiz-Rodríguez, M., Møller, A.P., Mousseau, T.A. et al. Defenses against keratinolytic bacteria in birds living in radioactively contaminated areas. Sci Nat 103, 71 (2016). https://doi.org/10.1007/s00114-016-1397-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-016-1397-5