Abstract
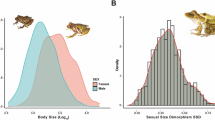
Recent studies on climate responses in ectothermic (cold-blooded) vertebrates have been few in number and focussed on phenology rather than morphology. According to Bergmann’s rule, endothermic (warm-blooded) vertebrates from cooler climates tend to be larger than congeners from warmer regions. Although amphibians are ectothermic vertebrates, weather and climatic conditions may also impact on their morphology, and thereby affect their survival rates and population dynamics. In this paper, we show, in a unique long-term study during the period 1963–2003 in an agricultural landscape in western Poland, that the body length of two water frog parental species (males of both Rana ridibunda and R. lessonae) increased significantly. However, their hybridogenetic hybrid R. esculenta did not show similar changes. A significant relationship with a large-scale climatic factor, the winter North Atlantic Oscillation index, was found positive for R. ridibunda males and R. lessonae females, and negative for R. esculenta females. Our findings, the first for amphibians, are consistent with other studies reporting that recent climate change has affected the morphology of animals. However, we also show that changes in amphibian phenotype linked to climate may vary independently between (even very similar) species.

Similar content being viewed by others
References
Abt G, Reyer HU (1993) Mate choice and fitness in a hybrid frog: Rana esculenta females prefer Rana lessonae males over their own. Behav Ecol Sociobiol 32:221–228
Berger L (1966) Biometrical studies on the population of green frogs from the environs of Poznañ. Ann Zool 23:303–324
Berger L (1973a) Sexual maturity of males within forms of Rana esculenta complex. Zool Pol 22:177–188
Berger L (1973b) Systematics and hybridization in European green frogs of Rana esculenta complex. J Herpetol 7:1–10
Berger L, Rybacki M (1998) Composition and ecology of water frog populations in agricultural landscape in Wielkopolska (central Poland). Biol Bull Pozn 35:103–111
Biek R, Funk CW, Maxell BA, Mills LS (2002) What is missing in amphibian decline research: insights from ecological sensitivity analysis. Conserv Biol 16:728–734
Bull CM, Burzacott D (2002) Changes in climate and in the timing of pairing of the Australian lizard, Tiliqua rugosa: a 15-year study. J Zool 256:383–387
Carey C, Alexander MA (2003) Climate change and amphibian declines: is there a link? Diver Distrib 9:111–121
Denton JS, Beebee TJC (1993) Density-related features of natterjack toad (Bufo calamita) populations in Britain. J Zool 229:105–119
Gotthard K (2001) Growth strategies of ectothermic animals in temperate environments. In: Atkinson D, Thorndyke M (eds) Animal developmental ecology. BIOS Scientific Publishers, Oxford, pp 287–304
Grant PR, Grant BR (2002) Unpredictable evolution in a 30-year study of Darwin’s finches. Science 296:707–711
Kiesecker JM, Blaustein AR, Belden LK (2001) Complex causes of amphibian population declines. Nature 410:681–684
Laugen AT, Laurila A, Jönsson KI, Söderman F, Merilä J (2005) Do common frogs (Rana temporaria) follow Bergmann’s rule? Evol Ecol Res 7:717–731
Lodė T (2001) Character convergence in advertisement call and mate choice in two genetically distinct water frog hybridogenetic lineages (Rana kl. esculenta, Rana kl. grafi). J Zool Syst Evol Res 39:91–97
Møller AP, Merilä J (2004) Analysis and interpretation of long-term studies investigating responses to climate change. Adv Ecol Res 35:111–130
Negovetic S, Anholt BR, Semelitsch RD, Reyer H-U (2001) Specific responses of sexual and hybridogenetic European waterfrog tadpoles to temperature. Ecology 82:766–774
Post E, Stenseth NC, Langvatn R, Fromentin J-M (1997) Global climate change and phenotypic variation among red deer cohorts. Proc R Soc Lond B 264:1317–1324
Rybacki M, Berger L (1994) Distribution and ecology of water frogs in Poland. Zool Pol 39:293–303
Schmidt-Nielsen K (1984) Scaling: why is animal size so important. Cambridge University Press, Cambridge, NY
Som C, Anholt BR, Reyer H-U (2000) The effect of assortative mating on the coexistence of a hybridogenetic waterfrog and its sexual host. Am Nat 156:34–46
Stenseth NC, Ottersen G, Hurrell JW, Mysterud A, Lima M, Chan, K-S, Yoccoz NG, Adlandsvik B (2003) Studying climate effects on ecology through the use of climate indices: the North Atlantic Oscillation, El Nino Southern Oscillation and beyond. Proc R Soc Lond B 270:2087–2096
Tryjanowski P, Rybacki M, Sparks T (2003) Changes in the first spawning dates of common frogs and common toads in western Poland in 1978–2002. Ann Zool Fenn 40:459–464
Uzzel T, Günther R, Berger L (1977) Rana ridibunda and Rana esculenta: a leaky hybridogenetic system (Amphibia, Salientia). Proc Acad Nat Sci Philadelphia 128:147–171
Walther G-R, Post E, Convey P, Menzel A, Parmesan C, Beebee TJC, Fromentin J-M, Hoegh-Guldberg O, Bairlein F (2002) Ecological responses to recent climate change. Nature 416:389–395
Yom-Tov Y (2001) Global warming and body mass decline in Israeli passerine birds. Proc R Soc Lond B 268:947–952
Acknowledgements
During the study, work was supported by different grants from CPBP and KBN. We are grateful to Y. Yom-Tov and M. Hromada for discussions on ideas in the manuscript and to two anonymous reviewers for improving an earlier version. PT’s sabbatical at Monks Wood was supported by the Foundation for Polish Science.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tryjanowski, P., Sparks, T., Rybacki, M. et al. Is body size of the water frog Rana esculenta complex responding to climate change?. Naturwissenschaften 93, 110–113 (2006). https://doi.org/10.1007/s00114-006-0085-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00114-006-0085-2