Abstract
Aim
The aim of this study was to evaluate acute and late radiotherapy-associated hepatotoxicity in consideration of dose–volume effects and liver function in childhood and adolescence.
Patients and methods
Since 2001, irradiated children and adolescents in Germany have been prospectively documented in the “Register of Treatment-Associated Late Effects After Radiotherapy of Malignant Diseases in Childhood and Adolescence (RiSK)” using standardized forms. Toxicity was graded according to the Radiation Therapy Oncology Group (RTOG) criteria.
Results
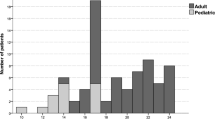
Until April 2012, 1,392 children and adolescents from 62 radiotherapy centers were recruited. In all, 216 patients underwent irradiation of the liver (median age 9 years, range 1–18 years, 70 patients with total-body irradiation, TBI). For 75 % of patients without TBI, information on acute toxicity of the liver was available: 24 patients had acute toxicity of grade 1–4 (grade 1, 2, and 4, in 20, 3, and 1 patient, respectively), including five patients receiving simultaneous hepatotoxic chemotherapy. Information on late toxicity was documented in 465 forms from 216 patients, with a median follow-up of 2 years. A maximum grade of toxicity of ≥ 0 occurred in 18 patients over time (with grade 1, 2, and 3 toxicity occurring in 15, 2, and 1 patient, respectively), including three patients (17 %) with TBI. One of them received simultaneous hepatotoxic chemotherapy. In multivariable analysis, volume–dose correlations showed no statistically noticeable effect on acute or chronic toxicity.
Conclusion
Only low hepatotoxicity developed in children after irradiation of various abdominal and thoracic tumors. Due to the low radiation doses to the liver (median liver dose = 5 Gy) and the low toxicities that were consecutively observed, dose–volume curves for liver toxicity could not be established. These findings reflect the cautious attitude of radiation oncologists in terms of attributable liver doses in the treatment of the investigated tumor entities. It offers the option of increasing these conservative doses if tumor control is necessary.
Zusammenfassung
Zielsetzung
Ziel dieser Auswertung war die Bewertung der akuten und späten bestrahlungsassoziierten Hepatotoxizität im Kindes- und Jugendalter unter Berücksichtigung der Dosis-Volumen-Effekte und der Leberfunktion.
Patienten und Methoden
Seit 2001 werden in Deutschland bestrahlte Kinder und Jugendliche prospektiv im "Register zur Erfassung von Spätfolgen nach Strahlentherapie im Kindes- und Jugendalter (RiSK)" mit Hilfe standardisierter Fragebögen dokumentiert. Die Toxizität wurde nach den Kriterien der Radiation Therapy Oncology Group (RTOG) graduiert.
Ergebnisse
Bis April 2012 wurden 1392 Kinder und Jugendliche aus 62 Strahlentherapiezentren erfasst. Davon wurden 216 Patienten an der Leber bestrahlt (medianes Alter 9 Jahre, Spanne 1–18 Jahre, 70 Ganzkörperbestrahlungen [TBI]). Von 75 % der Patienten ohne TBI lagen uns Angaben zur akuten Toxizität der Leber vor: 24 Patienten mit Grad 1–4 (Grad 1, 2 und 4 traten bei jeweils 20, 3 und1 Patienten auf), darunter 5 Patienten mit simultaner hepatotoxischer Chemotherapie. Eine maximale Spättoxizität ≥ 0 (465 Fragebögen von 216 Patienten, medianes Follow-up 2 Jahre, maximaler Grad der Toxizität im zeitlichen Verlauf) trat bei 18 Patienten auf (15 mit Grad 1, 2 mit Grad 2 und 1 Patient mit Grad 3), darunter waren 3 Patienten (17 %) mit TBI. Eine simultane Chemotherapie erhielten 28 % der Patienten. In der multivariaten Analyse zeigten die Volumen-Dosis-Beziehungen keine statistisch auffälligen Effekte hinsichtlich akuter oder chronischer Toxizität.
Fazit
Nach der Bestrahlung verschiedener abdomineller und thorakaler Tumoren entwickelten Kinder eine nur geringe Lebertoxizität. Aufgrund der niedrigen Strahlendosen an der Leber (mediane Leberdosis : 5 Gy) und der konsekutiv geringen beobachteten Toxizitäten konnten Toxizitätskurven der Dosis-Leber-Volumen nicht etabliert werden. Diese Befunde spiegeln die vorsichtige Einstellung der Strahlentherapeuten hinsichtlich der zu verantwortenden Leberdosen bei der Behandlung der untersuchten Tumorentitäten wieder und eröffnen die Option auf vorsichtige Dosiserhöhungen an der Leber in Fällen, in denen dies aus Gründen der Tumorkontrolle erforderlich ist.

Similar content being viewed by others
References
Castellino S, Muir A, Shah A et al (2010) Hepato-biliary late effects in survivors of childhood and adolescent cancer: a report from the Children's Oncology Group. Pediatr Blood Cancer 54:663–669
Mertens AC, Liu Q, Neglia JP et al (2008) Cause-specific late mortality among 5-year survivors of childhood cancer: the Childhood Cancer Survivor Study. J Natl Cancer Inst 100:1368–1379
Mounessi FS, Lehrich P, Haverkamp U et al (2013) Pelvic Ewing sarcomas. Three-dimensional conformal vs. intensity-modulated radiotherapy. Strahlenther Onkol 189:308–314
Andolino DL, Johnson CS, Maluccio M et al (2011) Stereotactic body radiotherapy for primary hepatocellular carcinoma. Int J Radiat Oncol Biol Phys 81:e447–453
Dorr W, Kallfels S, Herrmann T (2013) Late bone and soft tissue sequelae of childhood radiotherapy. Relevance of treatment age and radiation dose in 146 children treated between 1970 and 1997. Strahlenther Onkol 189:529–534
Bölling T, Schuck A, Rube C et al (2006) Therapy-associated late effects after irradiation of malignant diseases in childhood and adolescence. Feasibility analyses of a prospective multicenter register study. Strahlenther Onkol 182:443–449
Bölling T, Schuck A, Pape H et al (2008) Study protocol of the German “Registry for the detection of late sequelae after radiotherapy in childhood and adolescence”; (RiSK). Radiat Oncol 3:10
Bölling T, Ernst I, Pape H et al (2011) Dose-volume analysis of radiation nephropathy in children: preliminary report of the risk consortium. Int J Radiat Oncol Biol Phys 80:840–844
Bölling T, Geisenheiser A, Pape H et al (2011) Hypothyroidism after head-and-neck radiotherapy in children and adolescents: preliminary results of the “Registry for the Evaluation of Side Effects After Radiotherapy in Childhood and Adolescence”; (RiSK). Int J Radiat Oncol Biol Phys 81:e787–791
Cox JD, Stetz J, Pajak TF (1995) Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J Radiat Oncol Biol Phys 31:1341–1346
Schmoll HJ, Höffken K, Possinger K et al (2006) Toxizität antineoplastischer Substanzen. In: Schmoll HJ, Höffken K, Possinger K (eds) Kompendium Internistische Onkologie: Standards in Diagnostik und Therapie. Springer, Berlin, pp 867–896
Cheng JC, Wu JK, Huang CM et al (2002) Radiation-induced liver disease after radiotherapy for hepatocellular carcinoma: clinical manifestation and dosimetric description. Radiother Oncol 63:41–45
Guha C, Kavanagh BD (2011) Hepatic radiation toxicity: avoidance and amelioration. Semin Radiat Oncol 21:256–263
Marks LB, Yorke ED, Jackson A et al (2010) Use of normal tissue complication probability models in the clinic. Int J Radiat Oncol Biol Phys 76:10–19
Selo N, Bölling T, Ernst I et al (2010) Acute toxicity profile of radiotherapy in 690 children and adolescents: RiSK data. Radiother Oncol 97:119–126
Zabel-du Bois A, Milker-Zabel S, Bruns F et al (2014) Evaluation of time, attendance of medical staff and resources for radiotherapy in pediatric and adolescent patients: the DEGRO-QUIRO trial. Strahlenther Onkol 190:582–590
Malik IA, Moriconi F, Sheikh N et al (2010) Single-dose gamma-irradiation induces up-regulation of chemokine gene expression and recruitment of granulocytes into the portal area but not into other regions of rat hepatic tissue. Am J Pathol 176:1801–1815
Rave-Frank M, Malik IA, Christiansen H et al (2013) Rat model of fractionated (2 Gy/day) 60 Gy irradiation of the liver: long-term effects. Radiat Environ Biophys 52:321–338
Bölling T, Willich N, Ernst I (2010) Late effects of abdominal irradiation in children: a review of the literature. Anticancer Res 30:227–231
Emami B, Lyman J, Brown A et al (1991) Tolerance of normal tissue to therapeutic irradiation. Int J Radiat Oncol Biol Phys 21:109–122
Dawson LA, Ten Haken RK (2005) Partial volume tolerance of the liver to radiation. Semin Radiat Oncol 15:279–283
Flandin I, Hartmann O, Michon J et al (2006) Impact of TBI on late effects in children treated by megatherapy for Stage IV neuroblastoma. A study of the French Society of Pediatric oncology. Int J Radiat Oncol Biol Phys 64:1424–1431
Chou RH, Wong GB, Kramer JH et al (1996) Toxicities of total-body irradiation for pediatric bone marrow transplantation. Int J Radiat Oncol Biol Phys 34:843–851
Kun LE, Camitta BM (1978) Hepatopathy following irradiation and adriamycin. Cancer 42:81–84
Bhanot P, Cushing B, Philippart A et al (1979) Hepatic irradiation and adriamycin cardiotoxicity. J Pediatr 95:561–563
Muller K, Schlamann A, Guckenberger M et al (2014) Craniospinal irradiation with concurrent temozolomide for primary metastatic pediatric high-grade or diffuse intrinsic pontine gliomas. A first report from the GPOH-HIT-HGG Study Group. Strahlenther Onkol 190:377–381
Muller K, Schlamann A, Seidel C et al (2013) Craniospinal irradiation with concurrent temozolomide and nimotuzumab in a child with primary metastatic diffuse intrinsic pontine glioma. A compassionate use treatment. Strahlenther Onkol 189:693–696
Acknowledgments
We gratefully acknowledge the financial support of RiSK by the German Children’s Cancer Foundation, Bonn. Research of the last author, DS, was supported by the MHH Equal Opportunities Office, Hannover. Furthermore, we would like to thank everyone involved in the documentation of patients in various clinics, the RiSK office for its support, and Silke and David Mainwaring for proofreading.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
P. Rösler, H. Christiansen, R-D. Kortmann, C. Martini, C. Matuschek, F. Meyer, C. Rübe, T. Langer, R. Koch, H.T. Eich, N. Willich, and D. Steinmann state that there are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Rösler, P., Christiansen, H., Kortmann, RD. et al. Hepatotoxicity after liver irradiation in children and adolescents. Strahlenther Onkol 191, 413–420 (2015). https://doi.org/10.1007/s00066-014-0796-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-014-0796-9