Abstract
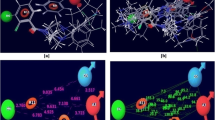
Chromodomain-helicase-DNA-binding protein 1-like is a chromodomain-containing protein in the SNF2-like family of ATPases. It has the capability to sustain proliferation in cell, encourage tumor growth and prevent apoptosis of cell. The goal of the current study is to build an in silico homology model further to identify the structural features that influence the inhibitory activity of chromodomain-helicase-DNA-binding protein 1-like protein grounded on a variety of 103 set of compounds. GOLD program is used to carry out molecular docking studies to ascertain the binding mode of structurally varied inhibitors of chromodomain-helicase-DNA-binding protein 1-like protein. Most active residues docked with chromodomain-helicase-DNA-binding protein 1-like protein are compound 20, 103 and 22 with their GOLD Scores 90.5, 81.01 and 79.2, respectively. These docked residues exhibited substantial interaction with active site residues of the protein. Ligand-protein binding is further elucidated with the extensive hydrogen bonding and other hydrophobic interactions. Chromodomain-helicase-DNA-binding protein 1-like protein belongs to Snf2 family of proteins with conserved evolutionary function. Another interesting aspect of this study is the presence of a conserved Snf2 N-terminal domain observed in chromodomain-helicase-DNA-binding protein 1-like protein. It controls the catalytic and the helicase activity which is crucial in regulating tumor progression. A hundred nanosecond molecular dynamics simulation of docked chromodomain-helicase-DNA-binding protein 1-like illustrated a stable binding pattern of ligand in the protein’s active site. Furthermore, trajectory analysis was performed to assess various characteristics of the docked system in terms of function of time. This study pinpoints potential novel inhibitors against chromodomain-helicase-DNA-binding protein 1-like protein which have not been reported previously but are involved in the overexpression in different cancers. This finding will help to design a prospective drug for varied number of cancers.









Similar content being viewed by others
References
Abro A, Azam SS (2016) Binding free energy based analysis of arsenic (+3 oxidation state) methyltransferase with S-adenosylmethionine. J Mol Liq 220:375–382
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22(2):195–201
Azam SS, Sarfaraz S, Abro A (2014) Comparative modeling and virtual screening for the identification of novel inhibitors for myo-inositol-1-phosphate synthase. Mol Biol Rep 23(9):4108–4137
Azam SS, Abro A, Raza S (2015) Binding pattern analysis and structural insight into the inhibition mechanism of Sterol 24-C methyltransferase by docking and molecular dynamics approach. J Biomol Struct Dyn 11:1–15
Azam SS, Saroosh A, Zaman N, Raza S (2013) Role of N-acetylserotonin O-methyltransferase in bipolar disorders and its dynamics. J Mol Liq 182:25–31
Berendsen HJ, Postma JV, van Gunsteren WF, DiNola AR, Haak JR (1984) Molecular dynamics with coupling to an external bath. J Chem Phys 81(8):3684–3690
Caruthers JM, McKay DB (2002) Helicase structure and mechanism. Curr Opin Struct Biol 12(1):123–133
Chen L, Hu L, Chan TH, Tsao GS, Xie D, Huo KK, Fu L, Ma S, Zheng BJ, Guan XY (2009) Chromodomain helicase/adenosine triphosphatase DNA binding protein 1-like (CHD1l) gene suppresses the nucleus‐to‐mitochondria translocation of nur77 to sustain hepatocellular carcinoma cell survival. Hepatology 50(1):122–129
Chen VB, Arendall WB, Headd JJ, Keedy DA, Immormino RM, Kapral GJ, Murray LW, Richardson JS, Richardson DC (2010) MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr Sect D 66(1):12–21
Clapier CR, Cairns BR (2009) The biology of chromatin remodeling complexes. Annu Rev Biochem 78:273–274
Colovos C, Yeates TO (2008) Verification of protein structures: patterns of nonbonded atomic interactions. Protein Sci 2:1511–1519
Danielson ML, Lill MA (2012) Predicting flexible loop regions that interact with ligands: the challenge of accurate scoring. Proteins Struct Funct Bioinform 80(1):246–260
Duan Y, Wu C, Chowdhury S, Lee MC, Xiong G, Zhang W, Yang R, Cieplak P, Luo R, Lee T, Caldwell J, Wang J, Kollman P (2003) A point‐charge force field for molecular mechanics simulations of proteins based on condensed‐phase quantum mechanical calculations. J Comput Chem 24(16):1999–2012
Eisen JA, Sweder KS, Hanawalt PC (1995) Evolution of the SNF2 family of proteins: subfamilies with distinct sequences and functions. Nucleic Acids Res 23(14):2715–2723
Eisenberg D, Lüthy R, Bowie JU (1997) VERIFY3D: assessment of protein models with three-dimensional profiles. Methods Enzymol 277:396–404
Eswar N, Eramian D, Webb B, Shen MY, Sali A (2008) Protein structure modeling with modeller. Structural proteomics. Humana Press, New York, p 145–159
Flaus A, Martin DM, Barton GJ, Owen-Hughes T (2006) Identification of multiple distinct Snf2 subfamilies with conserved structural motifs. Nucleic Acids Res 34(10):2887–2905
Gasteiger E, Hoogland C, Gattiker A, Wilkins MR, Appel RD, Bairoch A (2005) Protein identification and analysis tools on the ExPASy server. In: Walker John M (ed) The proteomics protocols handbook. Humana Press, New York, pp 571–607
Hauk G, McKnight JN, Nodelman IM, Bowman GD (2010) The chromodomains of the Chd1 chromatin remodeler regulate DNA access to the ATPase motor. Mol Cell 39(5):711–723
Jones G, Willett P, Glen RC (1995) Molecular recognition of receptor sites using a genetic algorithm with a description of desolvation. J Mol Biol 245:43–53
Karas GI, Kustatscher G, Buhecha HR, Allen MD, Pugieux C, Sait F, Bycroft M, Ladurner AG (2005) The macro domain is an ADP-ribose binding module. EMBO J 24:1911–1920
Khafizov K, Madrid-Aliste C, Almo SC, Fiser A (2014) Trends in structural coverage of the protein universe and the impact of the protein structure initiative. Proc Natl Acad Sci 111:3733–3738
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) PROCHECK: a program to check the stereochemical quality of protein structures. J Appl Cryst 26(2):283–291
Li Z, Wan H, Shi Y, Ouyang P (2004) Personal experience with four kinds of chemical structure drawing software: review on Chem Draw, Chem Window, ISIS/Draw, and Chem Sketch. J Chem Inf Comput Sci 44:1886–1890
Lusser A, Urwin DL, Kadonaga JT (2005) Distinct activities of CHD1 and ACF in ATP-dependent chromatin assembly. Nat Struct Mol Biol 12:160–166
Moonrin N, Songtawee N, Rattanabunyong S, Chunsrivirot S, Mokmak W, Tongsima S, Choowongkomon K (2015) Understanding the molecular basis of EGFR kinase domain/MIG-6 peptide recognition complex using computational analyses. BMC Bioinform 16(1):1
Parkin DM, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
Pearlman DA, Case DA, Caldwell JW, Ross WS, Cheatham III TE, DeBolt S, Ferguson D, Seibal G, Kollman P (1995) AMBER, a package of computer programs for applying molecular mechanics, normal mode analysis, molecular dynamics and free energy calculations to simulate the structural and energetic properties of molecules. Comput Phys Commun 91(1):1–41
Pettersen EF, Goddard TD, Huang CC, Couch GS, Greenblatt DM, Meng EC, Ferrin TE (2004) UCSF Chimera, a visualization system for exploratory research and analysis. J Comput Chem 25:1605–1612
Pieper U, Eswar N, Braberg H, Madhusudhan MS, Davis FP, Stuart AC, Mirkovic N, Rossi A, Marti‐Renom MA, Fiser A, Webb B (2004) MODBASE, a database of annotated comparative protein structure models, and associated resources. Nucleic Acids Res 32:D217–D222
Saleem A, Azam SS, Zarina S (2012) Docking and molecular dynamics simulation studies on glycation-induced conformational changes of human paraoxonase 1. Eur Biophys J 41(2):241–248
Salomon‐Ferrer R, Case DA, Walker RC (2013) An overview of the Amber biomolecular simulation package. Wiley Interdiscip Rev Comput Mol Sci 3(2):198–210
Schomburg I (2002) BRENDA, enzyme data and metabolic information. Nucleic Acids Res 30(1):47–49
Shamim A, Abbasi SW, Azam SS (2015) Structural and dynamical aspects of Streptococcus gordonii FabH through molecular docking and MD simulations. J Mol Graph Model 60:180–196
Simic R, Lindstrom DL, Tran HG, Roinick KL, Costa PJ, Johnson AD, Hartzog GA, Arndt KM (2003) Chromatin remodeling protein Chd1 interacts with transcription elongation factors and localizes to transcribed genes. EMBO J 22:1846–1856
Stockdale C, Flaus A, Ferreira H, Owen-Hughes T (2006) Analysis of nucleosome repositioning by yeast ISWI and Chd1 chromatin remodeling complexes. J Biol Chem 281:16279–16288
Teodoro ML, Phillips GN, Kavraki LE (2001) Molecular docking: a problem with thousands of degrees of freedom. In Robotics and Automation, 2001. Proceedings 2001 ICRA. IEEE international conference. vol 1, pp 960–965
Uddin R, Saeed K (2014) Identification and characterization of potential drug targets by subtractive genome analyses of methicillin resistant Staphylococcus aureus. Comput Biol Chem 48:55–63
Vaught A (1996) Graphing with Gnuplot and Xmgr: two graphing packages available under linux. Linux J 7
Verdonk ML, Berdini V, Hartshorn MJ, Mooij WT, Muray CW, Taylor RD, Watson P (2004) Virtual screening using protein ligand docking: avoiding artificial enrichment. J Chem Inf Comput Sci 44:793–806
Visualizer DS (2012) Release 3.5. Accelrys Inc, San Diego
Volkamer A, Kuhn D, Grombacher T, Rippmann F, Rarey M (2012) Combining global and local measures for structure-based druggability predictions. J Chem Inf Model 52:360–372
Wallace AC, Laskowski RA, Thornton JM (1995) LIGPLOT: a program to generate schematic diagrams of protein–ligand interactions. Protein Eng 8(2):127–134
Weiner PK, Kollman PA (1981) AMBER: assisted model building with energy refinement. A general program for modeling molecules and their interactions. J Comput Chem 2(3):287–303
Wiederstein M, Sippl MJ (2007) ProSA-web: interactive web service for the recognition of errors in three-dimensional structures of proteins. Nucleic Acids Res 35(Suppl 2):W407–W410
Wolber G, Langer T (2005) LigandScout: 3-D pharmacophores derived from protein-bound ligands and their use as virtual screening filters. J Chem Inf Model 45 1:160–169
Zhang Y (2008) I-TASSER server for protein 3D structure prediction. BMC Bioinform 9(1):40
Acknowledgments
Authors are highly grateful to the International Foundation of Science and Higher Education Commission, Islamabad, Pakistan for funding this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Iqbal, S., Shamim, A., Azam, S.S. et al. Identification of potent inhibitors for chromodomain-helicase- DNA-binding protein 1-like through moleculardocking studies. Med Chem Res 25, 2924–2939 (2016). https://doi.org/10.1007/s00044-016-1712-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-016-1712-x