Abstract
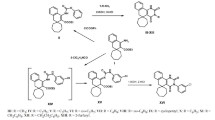
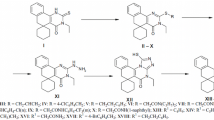
A series of novel 3-(4-chlorophenyl)-2-(2-(4-substituted)-2-oxoethylthio)quinazolin-4(3H)-one was synthesized by the reaction of 2-(3-(4-chlorophenyl)-4-oxo-3,4-dihydroquinazolin-2-ylthio)acetyl chloride with various amines. The starting material, (3-(4-chlorophenyl)-4-oxo-3,4-dihydroquinazolin-2-ylthio)acetyl chloride was synthesized from 4-chloroaniline by a multistep synthesis. All the title compounds were tested for their in vivo H1-antihistaminic activity on conscious guinea pigs at the dose level of 10 mg/kg using chlorpheniramine maleate as the reference standard. The results of the biological activity indicate that the test compounds protected the animals from histamine-induced bronchospasm significantly. Compound 3-(4-chlorophenyl)-2-(2-(4-methylpiperazin-1-yl)-2-oxoethylthio)quinazolin-4(3H)-one (1) emerged as the most potent compound of the series (71.13 % protection) when compared to the reference standard chlorpheniramine maleate (70.09 % protection). Interestingly, compound A1 shows negligible sedation (12 %) compared to chlorpheniramine maleate (32 %). Therefore, compound A1 can serve as the lead molecule for further development.


Similar content being viewed by others
References
Alagarsamy V (2004) Synthesis and pharmacological investigation of some novel 2-methyl-3-(substituted methylamino)-(3H)-quinazolin-4-ones as histamine H1-receptor blockers. Pharmazie 59:753–755
Alagarsamy V, Venkatesaperumal R, Vijayakumar S, Angayarkanni T, Pounammal P, Senthilganesh S, Kandeeban S (2002) Synthesis and pharmacological investigation of some novel 2-phenyl-3-(substituted methyl amino) quinazolin-4(3H)-ones as H1-receptor blockers. Pharmazie 57:306–307
Alagarsamy V, Solomon VR, Murugan M (2007) Synthesis and pharmacological investigation of novel 4-benzyl-1-substituted-4H-[1,2,4]triazolo[4,3-a]quinazolin-5-ones as new class of H1-antihistaminic agents. Bioorg Med Chem 15:4009–4015
Carr AA, Meyer DR (1982) Synthesis of terfenadine. Arzneimittelforschung 32:1157–1159
Chairungsrilerd N, Furukawa K, Ohta T, Nozoe S, Ohizumi Y (1996) Pharmacological properties of alpha-mangostin, a novel histamine H1 receptor antagonist. Eur J Pharmacol 314:351–356
Devsingh S, Bhagawanraju M, Bahekar RH, Rajan KS, Raghuram rao A (2001) Synthesis and H1- Antihistaminic activity of few quinazolinones. Ind J Chem 40B:813–816
Dews PB (1953) The measurement of the influence of drugs on voluntary activity in mice. Br J Pharmacol 8:46–48
Ellis EF, Adkinson NF, Yungingen JW, Busso WW. (1985). H1-receptor antagonists. Musby-year book, Inc., New York, p. 856–891
Hopp RJ, Bewtra A, Nair NM, Townley RG (1985) The effect of age on methacholine response. J Allergy Clin Immunol 76:609–613
Kuhn WL, Van Maanen EF (1961) Central nervous system effects of thalidomide. J Pharmacol Exp Ther 134:60–68
Raghuramrao A, Chandrasekhar V, Mallareddy V (1986) Synthesis of heteryl-a-[(2-alkoxyethyl)oxy)dialkylaminoalkyl acetamides as possible antihistaminic agents. Indian Drugs 24:40
Raju VSK, Bhagawanraju M, Bahekar RH, Rajan KS, Raghuram rao A (1999) Synthesis and H1- Antihistaminic activity of new quinazolinones. Indian Drugs 36:759
Simons FE, Simons KJ (1994) The pharmacology and use of H1-receptor-antagonist drugs. N Engl J Med 330:1663–1670
Van Arman CG, Miller LM, O’Malley MP (1961) SC-10049: a catecholamine bronchodilator and hyperglycemic agent. J Pharmacol Exp Ther 133:90–97
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alagarsamy, V., Narendhar, B., Sulthana, M.T. et al. Design and synthesis of 3-(4-chlorophenyl)-2-(2-(4-substituted)-2-oxoethylthio)quinazolin-4(3H)-one as antihistamine agents. Med Chem Res 23, 4692–4699 (2014). https://doi.org/10.1007/s00044-014-1034-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-014-1034-9