Abstract
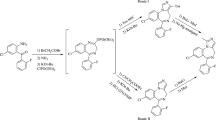
Condensation of various amines (Ia–c) with 4-isothiocyanato-4-methyl-2-pentanone (2a) at room temperature gave tetrahydropyrimidinethiones (3a–c) whereas condensation of 1a–e with 3-isothiocyanatobutanal at room temperature gave tetrahydropyrimidinethiones (4a–e). Amines 1a, d, f–i on condensation with 4-isothiocyanato-4-methyl-2-pentanone (2a) by heating under reflux for 8 h at pH ~4 (for 1a, d) and at RT (for 1f–i) gave dihydropyrimidinethiones 5a–b and 5c–f, respectively. Condensation of 1f with 3-isothiocyanatobutanal gave dihydropyimidinethione 6. When isothiocyanate (2a) and phenylenediamine derivatives 1g, h were heated under reflux at pH ~4, tricyclic compounds 7a, b were obtained in good yields. Condensation of 3-isothiocyanatobutanal with o-phenylenediamines 1h and 1j at pH ~ 5 by refluxing in methanol for 8 h gave products 8 and 9. All these compounds were screened for anti-inflammatory activity. Compounds 3c, 4c and 7a exhibited anti-inflammatory activity comparable to standard drug ibuprofen.


Similar content being viewed by others
References
Arai H, Matsumura T, Ishida H, Yamaura Y, Aratake S, Ohshima E, Yanagawa K, Miyama M, Suzuki K, Kawabe A, Nakanishi S, Kobayashi K, Sato T, Miki I, Ueno K, Fujii S, Iwase M (2004) PCT Int. Appl. WO 2003104230 Chem Abstr 140: 42193
Breault GA, Pease JE (2000) PCT Int. Appl. WO 2000012485 Chem Abstr 132: 194385
Devesa I, Alcaraz MJ, Riguera R, Ferrandiz ML, Ferrandiz ML (2004) A new pyrazolo pyrimidine derivative inhibitor of cyclooxygenase-2 with anti-angiogenic activity. Eur J Pharmacol 488:225–230
Hogenkamp D J, Nguyen P, Shao B (2001) PCT Int. Appl. WO 2001068612 Chem Abstr 135:257270
Kim DC, Lee YR, Yang B-S, Shin KJ, Kim DJ, Chung BY, Yoo KH (2003) Synthesis and biological evaluations of pyrazolo[3,4-d]pyrimidines as cyclin-dependent kinase 2 inhibitors. Eur J Med Chem 38:525–532
Lee JW, Lee BY, Kim CS, Lee SK, Lee SJ (2000) PCT Int. Appl. WO 2000029403 Chem Abstr 132: 347584
Mathes RA, Stewart FD, Swedish JF (1948) Synthesis of 2-pyrimidinethiols. J Am Chem Soc 70:1452–1453
Peretokin AV, Shutalev AD, Chupin VV, Mergenova AM, Ignatova LA, Malina YF, Unkoshii BV (1985) Formation of isomeric β-thhiocyanato carbonyl compounds in the reaction of thiocyanic acid with α,β-unsaturated aldehydes and ketones. Zh Org Khim 21:1004–1016
Sondhi SM, Dinodia M, Singh J, Rani R (2007) Heterocyclic compounds as anti-inflammatory agents. Curr Bioactive Comp 3:91–108
Sondhi SM, Rani R, Roy P, Agrawal SK, Saxena AK (2009a) Microwave-assisted synthesis of N-substituted cyclic imides and their evaluation for anticancer and anti-inflammatory activities. Bioorg Med Chem Lett 19:1534–1538. doi:10.1016/j.bmcl.2008.07.048
Sondhi SM, Singh J, Kumar A, Jamal H, Gupta PP (2009b) Synthesis of amidine and amide derivatives and their evaluation for anti-inflammatory and analgesic activities. Eur J Med Chem 44:1010–1015. doi:10.1016/j.ejmech.2008.06.029
Sondhi SM, Rani R, Gupta PP, Agrawal SK, Saxena AK (2009c) Synthesis, anticancer, and anti-inflammatory activity evaluation of methanesulfonamide and amidine derivatives of 3,4-diaryl-2-imino-4-thiazolines. Mol Divers 13:357–366
Sondhi SM, Dinodia M, Jain S, Kumar A (2009d) Synthesis of biologically active novel bis-Schiff’s bases, bishydrazone and bisguanidine derivatives. Indian J Chem Soc 48B:1128–1136
Winter CA, Risley EA, Nuss GW (1962) Carrageenin-induced edema in hind paw of rat as an assay for antiinflammatory drugs. Proc Soc Exp Biol Med 111:544–547
Wolf GA (ed) (1981) Burger’s medicinal chemistry part III. Wiley, NewYork, pp 1205–1273
Acknowledgments
We are thankful to technical staff of the Chemistry Department, I. I. T. Roorkee, for spectroscopic studies and elemental analysis. One of the authors Mr. Jaiveer Singh (SRF-NET) is thankful to CSIR, New Delhi, for financial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sondhi, S.M., Singh, J., Agrawal, S.K. et al. Synthesis of pyrimidine and condensed pyrimidine derivatives and their evaluation for anti-inflammatory activity. Med Chem Res 21, 91–99 (2012). https://doi.org/10.1007/s00044-010-9507-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-010-9507-y